Abstract
Rationale
Alcohol use disorder (AUD) involves dysregulation of innate immune signaling in brain. Toll-like receptor 3 (TLR3), an innate immune receptor that is upregulated in post-mortem human alcoholics, leads to induction of interferon (IFN) signaling. IFNs have been linked to depressive-like symptoms and therefore may play a role in addiction pathology. Astrocyte-neuronal signaling may contribute to maladaptation of neuronal circuits.
Objectives
In this manuscript, we examine ethanol (EtOH) induction of IFN signaling in neuronal, astrocyte, and microglial cell lines and assess astrocyte-neuronal interactions.
Methods
U373 astrocytes, SH-SY5Y neurons, and BV2 microglia were treated with EtOH and analyzed for autocrine/paracrine IFN signaling.
Results
EtOH induced TLR3, IFNβ, and IFNγ in SH-SY5Y neurons and U373 astrocytes, but not in BV2 microglia. The IFN response gene TRAIL was also strongly upregulated by TLR3 agonist Poly(I:C) and EtOH in U373 astrocytes. TRAIL blockage via neutralizing antibody prevented induction of IFNs in SH-SY5Y neurons but not in U373 astrocytes. Blocking TRAIL in conditioned media from EtOH-treated astrocytes prevented induction of IFNs in SH-SY5Y neurons. Finally, an in vivo model of chronic 10-day binge EtOH exposure in C57BL6/J mice, as well as single acute treatment with Poly(I:C), showed increased TRAIL +IR cells in both orbitofrontal and entorhinal cortex.
Conclusions
This study establishes a role of astrocyte to neuron TRAIL release in EtOH-induced IFN responses. This may contribute to alcohol associated negative affect and suggest potential therapeutic benefit of TRAIL inhibition in AUD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alcohol use disorder (AUD) features prominent dysregulation of innate immune signaling in brain (Crews et al. 2017; Mayfield et al. 2013). AUD is also associated with progressively increasing negative-affect or depressive symptoms that are thought to promote the development and worsening of the disease (Boden and Fergusson 2011). Innate immune signaling, particularly interferons (IFNs), has been associated with the development of depression in humans and in vivo (Borsini et al. 2017; Callaghan et al. 2018; Fritz et al. 2018; Mina et al. 2015; Pinto and Andrade 2016). IFNγ was recently found to be upregulated in postmortem human alcoholic cortex (Johnson et al. 2015), and chronic ethanol (EtOH) induces IFNγ in vivo (Duncan et al. 2016; Pascual et al. 2015). Negative affect is a key aspect in the cycle of addiction, which is thought to drive self-administration (Koob 2015; Koob and Volkow 2010; Volkow et al. 2016). The induction of IFNs by EtOH, as well as the association of IFNs with negative affect, suggests a potential contribution for IFNs in the negative affective stage of addiction pathology. However, little is known about the mechanism of EtOH-induction of IFNs.
Toll-like receptors (TLRs), which are an important part of the innate immune system that respond to viral and bacterial components, also recognize endogenous agonists to promote sterile inflammation. TLRs are upregulated in post-mortem human alcoholic brain (for review, see Crews et al. 2017) as well as in rodent models (Lippai et al. 2013). Endosomal TLRs initiate IFN responses ultimately by induction of interferon regulatory factor (IRF) transcription factors. TLR3 is an endosomal TRIF-dependent TLR that results in IRF3 activation and IFN gene induction. TLR3 has been linked to alcohol drinking behavior in rodents (Jang et al. 2016), and 10-day binge EtOH treatment in mice not only upregulates TLR3 but also potentiates cytokine responses following systemic treatment with TLR3 agonist Poly(I:C) (Qin and Crews 2012). TLR3 is induced by EtOH in the prefrontal cortex and nucleus accumbens following alcohol self-administration (McCarthy et al. 2017b). We have shown previously that TLR3 is upregulated in postmortem human alcoholic brain, as well as in SH-SY5Y neurons in vitro (Lawrimore and Crews 2017). Thus, it is critical to understand the role of TLR3 and IFN induction in response to EtOH.
In peripheral macrophages, it has been well established that TLR3 activation leads to induction of type I (e.g., IFNβ) and type II (IFNγ) IFNs (Sen and Sarkar 2005) as well as a host of interferon response genes (IRGs) (Levy and Darnell Jr 2002). These include antiviral proteins, such as TNF-related apoptosis-inducing ligand (TRAIL/TNFSF10/APO2L/CD253). TRAIL can cause either cell death in TRAIL cell death-sensitive cells or IFN and other cytokine induction. This has been observed in multiple cell types (Croft and Siegel 2017; Henry and Martin 2017; Kumar-Sinha et al. 2002; Wilson et al. 2009). For instance, TRAIL does not induce cell death in T cells (LeBlanc and Ashkenazi 2003), though neuronal lines can be sensitized to TRAIL-mediated cell death by NGF (Ruggeri et al. 2016). However, IFN and TRAIL signaling, as well as how EtOH impacts these pathways, is still poorly understood among different brain cell types. We hypothesized that TRAIL signaling would regulate IFN induction in neurons.
Multiple studies have reported that EtOH sensitizes microglia, inducing a hyper-ramified morphology associated with increases in cytokine secretion. Although microglia are brain-specific innate immune monocytes, we have previously found that neurons also play a role in EtOH-induced innate immune signaling that is unique from that of microglia (Lawrimore and Crews 2017). Astrocytes also have immunological properties and have been shown to play an important role in EtOH-induced inflammation (Adermark and Bowers 2016), although the effect of EtOH is poorly understood. Innate immune signaling in brain shares the autocrine (cell-to-self signaling) and paracrine (cell-to-cell signaling) mechanisms found among systemic innate immune cells. For example, neuronal ligand CX3CL1 (fractalkine) signals to microglia through CX3CR1 altering immune signaling, synaptic plasticity (Sheridan et al. 2014), as well as amyloid β-complement processing in Alzheimer’s disease models (Lian et al. 2015). Although it is known that EtOH can alter astrocyte activation, little is known about astrocyte innate immune signaling and how it might impact neurons through paracrine signaling pathways in brain.
In this manuscript, we utilized neuronal, microglial, and astrocytic cell lines (SH-SY5Y, BV2, and U373, respectively) to examine EtOH-induced IFN signaling in each cell type. These cell lines have been used extensively in the study of their respective cell type (Cheung et al. 2009; Henn et al. 2009; Imaizumi et al. 2013; Korecka et al. 2013; Rosenberger et al. 2016) and importantly, enabled us to conduct experiments without possibility of contamination of other cell types that is common in primary cultures. We also examine astrocyte to neuronal signaling in the context of neuronal IFN induction. Interestingly, although BV2 microglia do respond to EtOH (Lawrimore and Crews 2017), the response did not include induction of TLR3 or IFNs. In contrast, in SH-5Y5Y neurons and U373 astrocytes, EtOH treatment resulted in a strong induction of TLR3, IFNβ, and IFNγ in both cell types. TRAIL was also strongly upregulated by both EtOH and TLR3 agonist Poly(I:C) in U373 astrocytes. TRAIL neutralizing antibody prevented EtOH induction of IFN in SH-SY5Y neurons, suggesting that TRAIL released by EtOH stimulates autocrine induction of IFN in neurons. Similarly, TRAIL neutralizing antibody blocked IFN induction in neurons by EtOH-conditioned U373-astrocyte media consistent with astrocyte TRAIL-mediated paracrine induction of IFN in neurons. Furthermore, mice treated systemically with TLR3 agonist Poly(I:C) or EtOH show increases in cortical TRAIL positive immunoreactive cells. These studies link EtOH to novel IFN-TRAIL innate immune signaling between neurons and astrocytes, which may contribute to the pathology of AUD.
Materials and methods
Cell lines and treatment reagents
BV2 microglia cells were acquired from ICLC (Genoa, Italy, #ATL03001). BV2 were cultured using Dubecco’s modified Eagle serum (DMEM, Life Technologies, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS, Life Technologies), 1× GlutaMAX (Life Technologies), and 1× antibiotic-antimycotic (Life Technologies). For all cell experiments (excluding MTT assay), polystyrene 6-well plates (Corning, Corning, NY, #3516) containing a culture area of 9.5 cm2 per well were used. BV2 were plated at 1.5 × 10^5 cells per well, and 16 h prior to treatments, media was aspirated, and then 2% FBS was added.
SH-SY5Y neuroblastoma cells were acquired from ATCC (Manassas, VA, #CRL-2266). SH-SY5Y were cultured using DMEM/F-12 + GlutaMAX (Life Technologies), 10% FBS, and 1× antibiotic-antimycotic. Prior to treatments, SH-SY5Y were plated in 6-well plates (3.75 × 10^5 per well), and 24 h after plating, cells were differentiated using 10-μM retinoic acid (RA, Sigma-Aldrich, St. Louis, MO, #R2625) for 4 days in Neurobasal media (Life Technologies) containing 2% B27 supplement (Life Technologies), 0.5-mM GlutaMAX, and 1× antibiotic-antimycotic, which induces a more mature neuronal phenotype (Kovalevich and Langford 2013). Media were refreshed 16 h prior to treatments.
U373-MG (U373) astroglioma cells were acquired from UNC Cell Culture Facility. U373 were cultured using Alpha MEM (Life Technologies), 10% FBS, and 1× antibiotic-antimycotic. U373 were plated in 6-well plates at 3.25 × 10^5 cells per well, and then 16 h prior to treatments, media was changed to 2% FBS.
All cell types were maintained in a humidified 5% CO2 incubator at 37 °C. Cells were treated at 90% confluency. Reagents used included EtOH (timepoint and concentration dependent on experiment; see results), Poly(I:C) (50 μg/mL, Amersham/GE Healthcare, Pittsburgh, PA, #27-4732-01), LPS (100 ng/mL, Sigma-Aldrich, #L2630), Imiquimod (1 μg/mL, Invivogen, #tlrl-imq), TRAIL (200 ng/mL, Millipore, #GF092), and TRAIL neutralizing antibody (2 μg/mL, BD Biosciences, #550912).
Conditioned media transfer
SH-SY5Y were cultured and differentiated with retinoic-acid as described in the previous section. U373 were cultured then treated with EtOH (100 mM, 24 h). Following treatment, media were collected, transferred to empty 6-well plates, and the EtOH was allowed to evaporate overnight as previously described (Walter and Crews 2017). Media were removed from SH-SY5Y, and the conditioned media from U373 were then transferred to each respective group. Immediately following media transfer, the TRAIL neutralizing antibody (2 μg/mL) was added to the appropriate groups. Cell lysates were harvested 24 h later for mRNA analysis using RT-PCR.
Mouse treatment
Male C57BL/6 mice (8 weeks old) were treated with either EtOH [5 g/kg, 25% w/v, intragastric (i.g.)] or water (i.g.) daily for 10 days, or were treated with a single dose of LPS [0.5 mg/kg, intraperitoneal (i.p.)], Poly(I:C) (13 mg/kg, i.p.), or saline (i.p.) as previously described (Qin and Crews 2012; Qin et al. 2008). Mice were sacrificed 24 h following the last dose. Following each treatment, mice were anesthetized using sodium pentobarbital, then transcardially perfused with 0.1-M PBS, followed by 4.0% paraformaldehyde in PBS. Brains were removed and sent to Neuroscience Associates (Knoxville, TN) for histological sectioning. Mice were housed in a temperature- (20 °C) and humidity-controlled vivarium on a 12-h/12-h light/dark cycle and provided ad libitum access to food and water. Experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of North Carolina at Chapel Hill and conducted in accordance with National Institutes of Health regulations for the care and use of animals in research.
Real-time PCR
Following treatments, cells were rinsed using ice cold PBS, then lysed using TRIzol (Invitrogen, Carlsbad, CA). Cells were homogenized using a microtube homogenizer system (Wilmad-Labglass, Vineland, NJ). Tubes were allowed to sit at room temperature (RT) for 5 min, then chloroform was added to each tube followed by mixing on table rocker for 10 min at RT. Tubes were spun at 13,800 ×g for 15 min at 4 °C, and the aqueous (RNA containing) phase was transferred to a new tube. An equal volume of 100% isopropranol was added to each tube, followed by mixing on a table rocker for 10 min, then spun at 13,800 ×g for 15 min at 4 °C. The RNA pellet was rinsed twice with cold 75% EtOH, allowed to air dry for 10 min at RT, then re-suspended in DEPEC-treated water. RNA concentration was determined using a Nanodrop (Thermo Scientific, Waltham, MA) and was reverse-transcribed to cDNA. The SYBR green PCR master mix (Life Technologies) was used for real-time (RT) PCR analysis. The relative differences in expression between groups were expressed using cycle time (Ct) values normalized with β-actin, and relative differences between control and treatment groups were calculated and expressed as relative increases setting control as 100%. Primers used are listed in Table 1.
Enzyme-linked immunosorbent assays (ELISAs)
Following treatments, 1 mL of media was collected and spun down at 500 ×g for 10 min to remove cell debris. Supernatant was transferred to a new tube and stored at − 20 until analysis. Media were analyzed using human TRAIL/TNFSF10 DuoSet ELISA (R&D Systems, #DY375) per the manufacturer’s instructions using undiluted media samples.
MTT assay
SH-SY5Y were plated on a polystyrene 96 well plate (Corning, #3596) at 1 × 10^5 cells per well and differentiated using retinoic acid as described in the previous section. Cells were treated with varying concentrations of TRAIL (1–1000 ng/mL) as well as TRAIL neutralizing antibody (2 μg/mL) that has been demonstrated to block TRAIL-induced apoptosis (Cantarella et al. 2003) in the appropriate groups in triplicate. Twenty-four hours following treatment, the MTT assay (Abcam, #ab211091) was used to assess cell death per the manufacturer’s instructions.
Immunohistochemistry
Neuroscience Associates sectioned brains into coronal slices (35 μm) and were returned to the lab for immunohistochemical staining as previously described (Vetreno and Crews 2012; Vetreno et al. 2013). Briefly, sections were washed with PBS followed by a hydrogen peroxide (0.6%) rinse, then antigen retrieval using Citra solution at 70 °C for 1 h. Sections were then blocked using rabbit serum, followed by an overnight incubation in goat anti-TRAIL (1:50, R&D #AF1121). The next day, sections were incubated with rabbit anti-goat IgG (1:200) at RT for 1 h. Sections were then incubated in ABC solution at RT for 1 h, followed by visualization using nickel-enhanced 3,3′-diaminobenzidinne (DAB). Sections were quantified for immunopositive cells using Nikon software, setting threshold for cell size and intensity that was applied unbiased to all sections.
Statistical analysis
Where appropriate, t tests or one-way ANOVAs with post hoc Tukey’s/Dunnet’s test were used. The particular test used for each data set is indicated in the appropriate figure legends. A p value of less than 0.05 was considered significant. Data is reported as mean ± standard error of the mean (SEM). All data analysis was conducted using Prism (Graphpad, La Jolla, CA).
Results
EtOH induces TLR3 and IFNs in U373 astrocytes and SH-SY5Y neurons, but not BV2 microglia
Previous studies have found EtOH increases expression of TLR3 in mouse (Qin and Crews 2012), rats (Vetreno and Crews 2012), as well as in post-mortem human alcoholic brain (Crews et al. 2013). Furthermore, EtOH upregulates TLR3 in SH-SY5Y neurons, but not in BV2 microglia (Lawrimore and Crews 2017). To determine EtOH-induced cell type-specific responses in this pathway, expression of TLR3, IFNβ, and IFNγ was examined following EtOH treatment in U373 astrocytes, SH-SY5Y neurons, and BV2 microglia. EtOH increased expression of TLR3 (2.2-fold, p < 0.05), IFNβ (2.9-fold, p < 0.05), and IFNγ (2.6-fold, p < 0.01) in U373 astrocytes (Fig. 1a, b). Furthermore, in SH-SY5Y, EtOH increased expression of TLR3 (5.4-fold, p < 0.01), IFNβ (2.7-fold, p < 0.001), and IFNγ (5-fold, p < 0.001) (Fig. 1c, d). However, BV2 microglia did not show induction of TLR3 or IFNs in response to EtOH (Fig. 1e, f). U373 astrocytes were found to be quite sensitive to the effects of EtOH. We found that 15 mM of EtOH, which is below the legal driving limit in the USA, upregulated IFNβ (3-fold, p < 0.05) and TLR3 (2.6-fold, p < 0.01) at 24 h (Fig. 2). Furthermore, multiple IFN signaling genes, cytokines, and TNF superfamily genes were upregulated in a concentration and time-dependent manner in U373 astrocytes (see Tables 2 and 3). Also, we found that multiple markers of “A1” astrocyte activation (Liddelow et al. 2017), such as AMIGO2, GFAP, and FKBP5 were all upregulated by EtOH (100 mM, 24 h; data not shown). In SH-SY5Y, we further found that EtOH upregulated IFN receptors and multiple components of the TNF superfamily signaling genes (Table 4). Thus, EtOH induces TLR3 and IFN expression in U373 astrocytes and SH-SY5Y neurons, but not BV2 microglia.
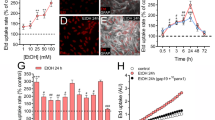
Ethanol increases TLR3, IFNs in U373 astrocytes and SH-SY5Y neurons, but not BV2 microglia. U373 astrocytes, SH-SY5Y neurons, and BV2 microglia were treated with EtOH (100 mM) for 24 h. Cell lysates were collected, and mRNA expression was measured using RT-PCR. Data is expressed as %CON (mean ± SEM), followed by analysis using two-tailed t tests. a, b EtOH increased expression of TLR3 (221 ± 12%), IFNβ (293 ± 54%), and IFNγ (258 ± 38%) in U373. c, d EtOH increased expression of TLR3 (536 ± 93%), IFNβ (273 ± 18%), and IFNγ (504 ± 75%) in SH-SY5Y. e, f EtOH did not increase either TLR3, IFNβ, or IFNγ in BV2. n = 5–6 per group; *p < 0.05, **p < 0.01, ***p < 0.001 vs. CON
Ethanol increases IFNβ and TLR3 at low concentrations in U373 astrocytes. U373 astrocytes were treated with varying concentrations of EtOH (15, 50, or 100 mM) for 24 h. Cell lysates were collected, and mRNA expression was measured using RT-PCR. Data is expressed as %CON (mean ± SEM). a EtOH increased expression of IFNβ mRNA at 15 (302 ± 59%), 50 (282 ± 57%), and 100 mM (293 ± 54%). One-way ANOVA [F(3,17) = 3.52, p = 0.0379] followed by Dunnet’s post hoc. b EtOH increased expression of TLR3 mRNA at 15 (259 ± 18%), 50 (258 ± 48%), and 100 mM (217 ± 35%). One-way ANOVA [F(3,17) = 5.84, p = 0.0068] followed by Dunnet’s post hoc. *p < 0.05, **p < 0.01 vs. CON
Stimulation with TLR3 agonist Poly(I:C) increases IFN expression and IFN response gene TRAIL in U373 astrocytes
Astrocytes show clear induction of TLR3 and IFNs in response to EtOH. We previously assessed the impact of TLR3 activation in SH-SY5Y neurons and BV2 microglia (Lawrimore and Crews 2017). In order to now investigate TLR3 signaling in U373 astrocytes, cells were treated with TLR3 agonist Poly(I:C) (50 μg/mL, 24 h). Poly(I:C) strongly upregulated both IFNβ (63-fold, p < 0.0001), IFNγ (2.4-fold, p < 0.05), and IFN regulated gene TNFSF10, also known as TRAIL (121-fold, p < 0.0001) (Fig. 3a). Furthermore, Poly(I:C) increased TRAIL secretion into the media (1.6-fold, p < 0.001) (Fig. 3b). In contrast, stimulation of TLR4 with LPS showed weaker responses, and stimulation of TLR7 with imiquimod showed no significant induction in the immune genes assessed (see Table 5). The concentrations of LPS (Shin et al. 2014) and imiquimod (Gay et al. 2013) tested cause significant immune gene induction in microglia. Thus, U373 astrocytes show induction of IFNs and TRAIL in response to TLR3.
Stimulation with TLR3 agonist Poly(I:C) increases IFN expression and IFN response gene TRAIL in U373 astrocytes. U373 astrocytes were treated with TLR3 agonist Poly(I:C) (50 μg/mL) for 24 h. Cell lysates were collected for mRNA analysis using RT-PCR, with data expressed as %CON (mean ± SEM). Cell media was collected for analysis of released TRAIL using ELISA. Statistical analysis conducted using two-tailed t-tests. a Poly(I:C) increased expression of IFNβ (6283 ± 1008%), IFNγ (238 ± 26%), and TRAIL (12,151 ± 689%). b Poly(I:C) increased release of TRAIL (158 ± 4.1%) in the media. c Schematic summarizing Poly(I:C) effects on IFN/TRAIL signaling in U373 astrocytes. n = 5–6 per group; ***p < 0.001, ****p < 0.0001 vs. CON
EtOH increases TRAIL expression and release in U373 astrocytes
TRAIL is a type II transmembrane protein that is cleaved and released where it acts on multiple receptors. TRAIL is linked to IFN signaling, being both induced by IFNs as well as induction of IFNs, dependent upon cell type (Kumar-Sinha et al. 2002). Since we found that EtOH increases TLR3 in U373 and that TLR3 stimulation increases TRAIL in U373, we next examined whether EtOH could also induce TRAIL in U373 astrocytes. We treated U373 astrocytes with EtOH and examined both TRAIL mRNA and release in the media. We found that EtOH increased TRAIL mRNA expression at 15 mM (1.7-fold p < 0.05), 50 mM (2-fold, p < 0.01), and 100 mM (1.7-fold, p < 0.05) (Fig. 4a, as well as released TRAIL as low as 15 mM (1.7-fold, p < 0.05) (Fig. 4b). EtOH rapidly released TRAIL protein into the media (Fig. 4e) that was accompanied by a delayed slow increase in mRNA even at 15 mM (Fig. 4d). Thus, EtOH promotes induction of TRAIL release and expression in U373 astrocytes.
Ethanol increases TRAIL expression and release in U373 astrocytes. U373 astrocytes were treated with EtOH at varying concentrations and timepoints. Cell lysates were collected for mRNA analysis using RT-PCR, and cell media was collected for analysis of released TRAIL using an ELISA. RT-PCR data is expressed as %CON (mean ± SEM). a EtOH upregulated TRAIL mRNA at 15 (173 ± 17%), 50 (207 ± 41%), and 100 mM (166 ± 23%). One-way ANOVA [F(3,29) = 5.0, p = 0.0065] followed by Dunnet’s post hoc. b EtOH increased TRAIL release in the media at 15 (174 ± 28%) and 100 mM (162 ± 27%). One-way ANOVA [F(3,38) = 5.21, p = 0.0041] followed by Dunnet’s post hoc. c Schematic illustrating EtOH increases TRAIL mRNA in U373 astrocytes. d EtOH increased TRAIL mRNA at 24 (469 ± 132%) h. One-way ANOVA [F(3,18) = 7.88, p = 0.0014] followed by Dunnet’s post hoc. e EtOH increased TRAIL release in the media at 1 (174 ± 36%), 6 (162 ± 28%), and 24 (174 ± 28%) h. One-way ANOVA [F(3,35) = 7.32, p = 0.0006] followed by Dunnet’s post hoc. f Schematic illustrating EtOH increases TRAIL release in U373 astrocytes. *p < 0.05, **p < 0.01, ***p < 0.001 vs. CON
EtOH induces IFNs in human SH-SY5Y neurons via TRAIL, but not in U373 astrocytes
Little is known about TRAIL signaling in brain, although in breast carcinoma cells (Kumar-Sinha et al. 2002), TRAIL induces IFNs. To determine if EtOH-induced TRAIL is linked to IFN induction, we used a TRAIL neutralizing antibody (αTRAIL) to block TRAIL. The TRAIL neutralizing antibody was added to the media 30 min prior to EtOH treatment of U373 astrocytes. While EtOH increased expression of IFNβ (2.6-fold, p < 0.05) and IFNγ (5.6-fold, p < 0.001), these increases were not blocked by the TRAIL neutralizing antibody (Fig. 5a–c). These data suggest that EtOH-induced IFN expression in astrocytes is not dependent on autocrine TRAIL signaling in U373 astrocytes and is more likely to be related to TLR3 or other EtOH-induced signaling. We found that both EtOH and TRAIL induced IFNs in SH-SY5Y neurons (Fig. 6a–f). Addition of the TRAIL neutralizing antibody 30 min prior to EtOH treatment of SH-SY5Y neurons did prevent EtOH-induced IFNβ and IFNγ gene induction (Fig. 6a–c). Similar to the effects of EtOH, the TRAIL neutralizing antibody blocked IFNβ and IFNγ induction in response to recombinant TRAIL (200 ng/mL) (Fig. 6d–f). Since TRAIL signaling has been shown to have neurodegenerative effects under certain conditions (Aktas et al. 2005; Cantarella et al. 2003), we examined whether TRAIL induces cell death in SH-SY5Y. TRAIL did not induce cell death at any of the concentrations examined (Fig. 6g). EtOH similarly does not induce cell death in SH-SY5Y neurons (Lawrimore and Crews 2017). Thus, these data suggest that EtOH induces IFN expression via TRAIL signaling in SH-SY5Y neurons in the absence of cell death.
Ethanol induced interferon expression in U373 astrocytes is not blocked by TRAIL neutralizing antibody. U373 astrocytes were treated with EtOH (100 mM). Thirty minutes prior to EtOH, TRAIL neutralizing antibody (αTRAIL; 2 μg/mL) was added to the specified groups. Twenty-four hours later, cell lysates were collected for mRNA analysis using RT-PCR, with data represented as %CON (mean ± SEM). a IFNβ was increased by EtOH (257 ± 15%) which was not blocked by αTRAIL (370 ± 65%). One-way ANOVA [F(3,12) = 10.92, p = 0.001] followed by Dunnet’s post hoc. b IFNγ was increased by EtOH (559 ± 98%), which was not blocked by αTRAIL (447 ± 47%). One-way ANOVA [F(3,10) = 23.62, p < 0.0001] followed by Dunnet’s post hoc. c Schematic illustrating that EtOH increases TRAIL U373 astrocytes, and that TRAIL neutralization does not affect EtOH-induced IFN induction. *p < 0.05, ***p < 0.001 vs. CON
Ethanol and TRAIL-induced interferons is blocked by TRAIL neutralizing antibody in SH-SY5Y neurons. a–c SH-SY5Y neurons were treated with EtOH (100 mM). Thirty minutes prior to EtOH, TRAIL neutralizing antibody (αTRAIL; 2 μg/mL) was added to the specified groups. Twenty-four hours later, cell lysates were collected for mRNA analysis using RT-PCR, with data represented as %CON (mean ± SEM). a IFNβ was increased by EtOH (433 ± 64%), which was blocked by αTRAIL (221 ± 39%). One-way ANOVA [F(3,17) = 13.62, p < 0.0001] followed by Tukey’s post hoc. b IFNγ was increased by EtOH (539 ± 146%), which was blocked by αTRAIL (278 ± 82%). One-way ANOVA [F(3,17) = 5.83, p = 0.0063] followed by Tukey’s post hoc. c Schematic illustrating EtOH increases IFNs in SH-SY5Y neurons that is blocked by TRAIL neutralizing antibody. d–f SH-SY5Y neurons were treated with TRAIL (200 ng/mL). Thirty minutes prior to TRAIL, TRAIL neutralizing antibody (αTRAIL; 2 μg/mL) was added to the specified groups. Twenty-four hours later, cell lysates were collected for mRNA analysis using RT-PCR. d IFNβ was increased by TRAIL (275 ± 58%) and was blocked by αTRAIL (106 ± 29%). One-way ANOVA [F(3,16) = 5.31, p = 0.0099] followed by Tukey’s post hoc. e IFNγ was increased by TRAIL (188 ± 17%) and was blocked by αTRAIL (80 ± 22%). One-way ANOVA [F(3,12) = 5.92, p = 0.010] followed by Tukey’s post hoc. f Schematic illustrating that TRAIL increases IFNs in SH-SY5Y neurons that is blocked by TRAIL neutralizing antibody. g MTT assay conducted in SH-SY5Y neuron with varying TRAIL concentrations (0–1000 ng/mL), with either αTRAIL (2 μg/mL) or a volume-equivalent of vehicle (PBS). No significant loss of viability was observed at any TRAIL concentration. Two-way ANOVA, no main effect of TRAIL, [F(7,32) = 1.43, p = 0.22]. *p < 0.05, **p < 0.01, ***p < 0.001 vs. CON; #p < 0.05 vs. EtOH; @p < 0.05, @@p < 0.01 vs. TRAIL
Since astrocytes secrete TRAIL in response to EtOH, but do not show IFN responses in response to TRAIL, we hypothesized that neurons might respond to TRAIL from astrocytes in a paracrine fashion. We treated U373 astrocytes with EtOH for 24 h then collected the media. To examine EtOH-induced signaling molecules separate from the direct effects of EtOH, we allowed the EtOH to evaporate overnight (Walter and Crews 2017). U373-astrocyte-derived EtOH conditioned media (EtOH-CM) was collected and then transferred to SH-SY5Y neurons with or without the addition of the TRAIL neutralizing antibody. We found that EtOH-CM induced both IFNβ (2.2-fold, p < 0.05) and IFNγ (1.8-fold, p < 0.05) in SH-SY5Y neurons. Interestingly, this induction of IFN was abolished by the TRAIL neutralizing antibody (Fig. 7b, c). These data suggest that EtOH releases TRAIL from U373 human astrocytes that induce IFN expression in SH-SY5Y neurons. Thus, EtOH causes both autocrine and paracrine TRAIL-mediated IFN induction in neurons. EtOH can directly cause IFN induction in neurons. Further, astrocytes communicate with neurons via TRAIL release in response to EtOH, leading to IFN induction.
Ethanol-induced TRAIL from U373 astrocytes induces interferons in SH-SY5Y neurons. a Schematic diagraming treatment outline. U373 astrocytes were treated with EtOH (100 mM, 24 h) followed by media removal. EtOH was allowed to evaporate from the media before transferring to SH-SY5Y neurons. TRAIL neutralizing antibody (αTRAIL) was added immediately following media transfer to the specified groups. Twenty-four hours later, cell lysates were collected for mRNA analysis using RT-PCR. b IFNβ was increased by ethanol-conditioned media (EtOH-CM; 215 ± 19%), which was blocked by αTRAIL (120 ± 20%). One-way ANOVA [F(3,15) = 6.67, p = 0.0044] followed by Tukey’s post hoc. c IFNγ was increased by EtOH-CM (178 ± 15%), which was blocked by αTRAIL (97 ± 11%). One-way ANOVA [F(3,17) = 3.15, p = 0.0421] followed by Tukey’s post hoc. d Schematic outlining signaling pathway between EtOH treated U373 and induction of interferons in SH-SY5Y neurons. *p < 0.05 vs. CON; #p < 0.05 vs. EtOH-CM
EtOH and Poly(I:C) induce TRAIL in mouse brain
To determine if EtOH and/or the TLR3 agonist Poly(I:C) alters brain TRAIL expression in vivo, we used a mouse model of chronic EtOH binge as well as a single acute Poly(I:C) treatment. C57BL/6 mice underwent a 10-day EtOH binge (5 g/kg, daily i.g.) or a single dose of TLR3 agonist Poly(I:C) (13 mg/kg, i.p.) and sacrificed 24 h later to assess TRAIL expression in brain We found both EtOH and Poly IC increased the number of TRAIL immunopositive reactive cells (TRAIL +IR) by approximately 20% in orbitofrontal cortex and entorhinal cortex relative to controls; EtOH (p < 0.05) or Poly(I:C) (p < 0.05) (Fig. 8a, b). We also observed that the TLR4 agonist LPS (0.5 mg/kg, i.p.) caused a similar induction of TRAIL (data not shown), suggesting TRAIL upregulation is sensitive to several immune stimuli. Thus, TRAIL is induced by EtOH and Poly(I:C) in mouse brain.
Ethanol and Poly(I:C) increase TRAIL +IR in mouse orbitofrontal and entorhinal cortex. C57BL6/J male mice (8 weeks old) were treated with either EtOH (once daily, 10 days, 5 g/kg, i.g.), Poly(I:C) (single dose, 13 mg/kg, i.p.), water (once daily, 10 days, i.g.), or saline (i.p.). Since no significant differences were seen between water (i.g.) and saline (i.p.) controls, these were grouped together into a single control [CON] group. Brains were processed for immunohistochemical staining by Neurosciences Associates and stained for TRAIL positive immunoreactive (+IR) cells. a The number of TRAIL +IR cells in both orbitofrontal and entorhinal cortex was increased by EtOH (p < 0.05) and Poly(I:C) (p < 0.05). One-way ANOVA [F(2,10) = 6.19, p = 0.0178] followed by Dunnet’s post hoc in orbitofrontal cortex, and one-way ANOVA [F(2,10)] = 4.22, p = 0.046] followed by Dunnet’s post hoc in entorhinal cortex. b Representative image of TRAIL +IR cells in entorhinal cortex, imaged at ×100. Positive cells are indicated by darker staining. *p < 0.05 vs. CON
Discussion
In this manuscript, we found that EtOH induces TLR3, IFNs, and TRAIL in both U373 astrocytes and SH-SY5Y neurons, but not in BV2 microglia. Our studies find that TRAIL contributes to both autocrine and paracrine forms of neuroimmune signaling in brain, as diagramed in Fig. 9. In SH-SY5Y neurons, EtOH directly induced IFN gene expression that was prevented by TRAIL neutralizing antibody. Also, astrocytes signal in a paracrine fashion to neurons via TRAIL to induce neuronal IFNs. Several other studies have identified paracrine glial-neuronal signaling pathways in the brain, such as fractalkine which may modulate synaptic plasticity (Sheridan et al. 2014) as well as dysregulated amyloid β-complement signaling in Alzheimer’s disease models (Lian et al. 2015). Our study finds a novel paracrine signaling mechanism from astrocytes to neurons involving TRAIL-mediated IFN induction in neurons due to alcohol. The relationship between TRAIL and IFNs has been identified previously in other cell types (Huang et al. 2009; Peteranderl and Herold 2017). In peripheral immune cells, TRAIL is often considered an IRG, one of dozens of genes induced by IFN receptor signaling (Huang et al. 2009; Levy and Darnell Jr 2002; Peteranderl and Herold 2017; Sato et al. 2001). We found that TRAIL induces IFNs in SH-SY5Y neurons. This has also been observed previously in breast carcinoma cells, showing a reciprocal interaction between these signaling systems (Kumar-Sinha et al. 2002). TRAIL was originally discovered to be involved in the initiation of cell death pathways (Pan et al. 1997). However, TRAIL has also been found to result in cytokine induction through NFkB and IRF mediated pathways (Henry and Martin 2017; Kumar-Sinha et al. 2002).
Summary of TRAIL-IFN interaction between U373 astrocytes and SH-SY5Y neurons. EtOH increases TLR3 and IFN mRNA expression in both U373 astrocytes and SH-SY5Y neurons. EtOH also increases release of TRAIL from U373 astrocytes, which further causes an increase of IFN expression in SH-SY5Y neurons. Blocking TRAIL signaling via a neutralizing antibody (αTRAIL) negates both EtOH-induced and TRAIL-induced IFN expression in SH-SY5Y neurons
In our studies, TRAIL did not cause detectable cell death in SH-SY5Y at concentrations of 200 ng/mL (24-h treatment). These findings are consistent with previous studies in SH-SY5Y neurons finding a 16-h treatment with TRAIL (500 ng/mL) did not result in cell death (Ruggeri et al. 2016). Some studies find that a longer 48-h treatment of TRAIL (100 ng/mL) reduces SH-SY5Y neuronal viability by approximately 30% (Cantarella et al. 2003) and induction of cell death by TRAIL in resected human brain tissue (Nitsch et al. 2000). Others have found that TRAIL induces cell death in SH-SY5Y neurons after NGF treatment (Ruggeri et al. 2016). This suggests that cell death pathways activated by TRAIL in neurons can vary and may be slower to initiate than IFN signaling in SH-SY5Y. Nevertheless, in our setting, we observe neuronal induction of IFNs by TRAIL in the absence of clear cell death. While implications of this in vivo are unclear, it is possible that persistent or ongoing upregulation of TRAIL signaling is needed to initiate neuronal cell death, as is observed in multiple sclerosis models (Aktas et al. 2005).
We found important differences between TRAIL-IFN signaling in response to EtOH between neurons and astrocytes. TRAIL antagonism prevented EtOH induction of IFNs in neurons but not astrocytes. Previously, we also found that TLR3 agonist Poly(I:C) induces a strong cytokine response in SH-SY5Y neurons (Lawrimore and Crews 2017); however, our current data suggests TRAIL, rather than TLR3 may be required for EtOH-induction of IFN in neurons. In astrocytes, however, we suspect that TLR3 signaling may be required for EtOH-induced IFN-TRAIL signaling. TLR3 stimulation via Poly(I:C) in U373 astrocytes strongly upregulated expression of IFNs as well TRAIL. TLR3 is upstream of IFN signaling and is prominently expressed in astrocytes (Farina et al. 2005; McCarthy et al. 2017a). We demonstrated here that Poly(I:C) strongly upregulates cytokines as well as IFNs in U373 astrocytes (see Table 4), consistent with studies conducted in primary human astrocytes (Farina et al. 2005; Serramia et al. 2015). The subdued effect of TLR4 and TLR7 stimulation in U373 astrocytes we observed is similar to previous studies that found little proinflammatory stimulation by TLR agonists other than Poly(I:C) in primary astrocytes (Farina et al. 2005). This is consistent with strong TLR3 responsivity in astrocytes. Importantly, we uniquely demonstrated that Poly(I:C) induces a strong upregulation of TRAIL as well in U373 astrocytes, with TRAIL antagonism having no effect on IFN induction, supporting the hypothesis that TLR3 is upstream of IFNs and TRAIL in astrocytes. Further, our finding of increased media TRAIL at 1-h post-treatment suggests that cleavage of membrane bound TRAIL might also be occurring, as TRAIL has been shown to be cleaved and released by cysteine proteases (Mariani and Krammer 1998). Future studies are required to definitively dissect the mechanism of TRAIL secretion in astrocytes. While we found that Poly(I:C) induces TRAIL in U373 astrocytes, previous studies indicate that Poly(I:C) also upregulates other IFN stimulated genes in astrocytes, such as ISG54 and ISG56 (Imaizumi et al. 2014), suggesting that further research into TLR3-TRAIL-IFN-mediated responses in astrocytes is warranted, especially given the ability of astrocytes to secrete factors that alter neurons (Liddelow et al. 2017).
While in the periphery IFNs are known for their anti-viral activity, in the brain, they may play different roles. In particular, exogenous IFNs have been well-documented in their ability to cause depression in clinical settings and in rodents (Borsini et al. 2017; Callaghan et al. 2018; Fritz et al. 2018; Lian et al. 2015; Mina et al. 2015; Pinto and Andrade 2016). Negative affect and depressive phenotypes are critical components in the cycle of addiction (Coleman Jr. and Crews 2018; Koob 2015; Koob and Volkow 2010; Volkow et al. 2016). Both the negative affect/depressive phenotypes found with exogenous IFNs and the development of negative affect in rodent models of alcohol addiction follow similar patterns of progressive onset (Breese et al. 2008; Breese et al. 2005; Raison et al. 2009; Wills et al. 2009; Zheng et al. 2014). Further, IFNγ was found to be upregulated in postmortem human alcoholic cortex (Johnson et al. 2015), and chronic EtOH in vivo can induce IFNγ (Duncan et al. 2016; Pascual et al. 2015). IFNγ has also been suggested to play a role in modulating social behavior in mice (Filiano et al. 2016), and IFNγ knockout mice lack LPS-induced conditioned place aversion, indicating a role for IFNγ in negative affect behaviors (Fritz et al. 2018). Therefore, blocking IFN induction by TRAIL in neurons could be a potential cell-specific target for AUDs and other neurologic disorders (Cantarella et al. 2015). Further research is needed to investigate the possible therapeutic potential of TRAIL inhibition as well as the mechanisms underlying cell-to-cell TRAIL signaling in the brain.
Although microglia are generally associated with neuroimmune signaling and primary microglia have been reported to release IFNβ (McDonough et al. 2017), we find IFN and TLR3 receptor induction by EtOH is primarily in astrocytes and neurons. Astrocytes play a vital role in modulating neuronal plasticity in the brain (Haydon and Nedergaard 2014) and are becoming a prominent focus of alcohol research (Adermark and Bowers 2016). Previous studies have indicated that the astrocyte secretome, such as multiple extracellular matrix proteins, is changed following chronic EtOH exposure during early postnatal life (Trindade et al. 2016), and that EtOH upregulates immune associated factors (e.g., COX-2, iNOS) in primary cultured rodent astrocytes (Valles et al. 2004). Thus, though microglia undoubtedly play important roles in neuroimmune responses to EtOH, the contribution of astrocytes should be further investigated.
It is important to note that our in vitro studies use immortalized cell lines, which may have different responses compared to primary cells or in vivo. However, these selected cell lines are widely utilized cell culture models for providing insight into neuronal, microglial, and astrocyte biology, including immune function. For instance, retinoic acid-differentiated SH-SY5Y neurons have been used extensively in Parkinson’s disease models (for reviews, see Xicoy et al. 2017; Xie et al. 2010) and much like dopaminergic neurons in vivo can actively uptake MPP(+) (Korecka et al. 2013). SH-SY5Y also have increased expression of immediate early genes (FOS and ARC) following stimulation with KCl, a response indicative of neuronal activation (Barry et al. 2017). SH-SY5Y also model innate immune signaling similar to primary neurons, as in our previous study, we found that TLR2, TLR3, TLR4, TLR7, and TLR8 are expressed in SH-SY5Y (Lawrimore and Crews 2017), similar to studies using primary neurons in mice and in vivo (Lehmann et al. 2012; Lok et al. 2015; Tang et al. 2007). BV2 microglia demonstrate a similar response to primary microglia following LPS (TLR4) stimulation (Henn et al. 2009). U373 express astrocyte markers and have similar cytokine responses as primary astrocytes (Imaizumi et al. 2013; Rosenberger et al. 2016). Further, similar to our findings in U373 astrocytes, primary human astrocytes have also been found to express IFN response gene TRAIL following treatment with proinflammatory cytokines (Choi et al. 1999). Therefore, though cell line experiments must be evaluated in the proper context, the literature indicates that the cell lines we have selected are appropriate in investigating innate immune function.
We also extended our in vitro findings with our in vivo experiments. To investigate the impact of EtOH on TRAIL in vivo in brain, we used our 10-day binge model of EtOH intoxication in mice (Qin and Crews 2012; Qin et al. 2008) that induces brain neuroimmune gene responses and determined TRAIL +IHC. TRAIL +IR was increased in both orbitofrontal and entorhinal cortex by EtOH treatment, regions that are affected by addiction pathology (Crews and Boettiger 2009; Schoenbaum and Shaham 2008). Thus, EtOH induces TRAIL in our selected in vitro models as well as in our in vivo binge EtOH model of AUD induced cortical pathology. In conclusion, we identify a novel signaling system whereby EtOH induces IFNs in neurons via neuronal and astrocytic secretion of TRAIL.
References
Adermark L, Bowers MS (2016) Disentangling the role of astrocytes in alcohol use disorder. Alcohol Clin Exp Res 40:1802–1816. https://doi.org/10.1111/acer.13168
Aktas O, Smorodchenko A, Brocke S, Infante-Duarte C, Topphoff US, Vogt J, Prozorovski T, Meier S, Osmanova V, Pohl E, Bechmann I, Nitsch R, Zipp F (2005) Neuronal damage in autoimmune neuroinflammation mediated by the death ligand TRAIL. Neuron 46:421–432. https://doi.org/10.1016/j.neuron.2005.03.018
Barry G, Briggs JA, Hwang DW, Nayler SP, Fortuna PRJ, Jonkhout N, Dachet F, Maag JLV, Mestdagh P, Singh EM, Avesson L, Kaczorowski DC, Ozturk E, Jones NC, Vetter I, Arriola-Martinez L, Hu J, Franco GR, Warn VM, Gong A, Dinger ME, Rigo F, Lipovich L, Morris MJ, O’Brien TJ, Lee DS, Loeb JA, Blackshaw S, Mattick JS, Wolvetang EJ (2017) The long non-coding RNA NEAT1 is responsive to neuronal activity and is associated with hyperexcitability states. Sci Rep 7:40127. https://doi.org/10.1038/srep40127
Boden JM, Fergusson DM (2011) Alcohol and depression. Addiction 106:906–914. https://doi.org/10.1111/j.1360-0443.2010.03351.x
Borsini A et al (2017) Interferon-alpha reduces human hippocampal neurogenesis and increases apoptosis via activation of distinct STAT1-dependent mechanisms. Int J Neuropsychopharmacol. https://doi.org/10.1093/ijnp/pyx083
Breese GR, Overstreet DH, Knapp DJ (2005) Conceptual framework for the etiology of alcoholism: a “kindling”/stress hypothesis. Psychopharmacology 178:367–380. https://doi.org/10.1007/s00213-004-2016-2
Breese GR, Knapp DJ, Overstreet DH, Navarro M, Wills TA, Angel RA (2008) Repeated lipopolysaccharide (LPS) or cytokine treatments sensitize ethanol withdrawal-induced anxiety-like behavior. Neuropsychopharmacology 33:867–876. https://doi.org/10.1038/sj.npp.1301468
Callaghan CK, Rouine J, Dean RL, Knapp BI, Bidlack JM, Deaver DR, O'Mara SM (2018) Antidepressant-like effects of 3-carboxamido seco-nalmefene (3CS-nalmefene), a novel opioid receptor modulator, in a rat IFN-alpha-induced depression model. Brain Behav Immun 67:152–162. https://doi.org/10.1016/j.bbi.2017.08.016
Cantarella G, Uberti D, Carsana T, Lombardo G, Bernardini R, Memo M (2003) Neutralization of TRAIL death pathway protects human neuronal cell line from beta-amyloid toxicity. Cell Death Differ 10:134–141. https://doi.org/10.1038/sj.cdd.4401143
Cantarella G, di Benedetto G, Puzzo D, Privitera L, Loreto C, Saccone S, Giunta S, Palmeri A, Bernardini R (2015) Neutralization of TNFSF10 ameliorates functional outcome in a murine model of Alzheimer’s disease. Brain 138:203–216. https://doi.org/10.1093/brain/awu318
Cheung YT, Lau WK, Yu MS, Lai CS, Yeung SC, So KF, Chang RC (2009) Effects of all-trans-retinoic acid on human SH-SY5Y neuroblastoma as in vitro model in neurotoxicity research. Neurotoxicology 30:127–135. https://doi.org/10.1016/j.neuro.2008.11.001
Choi C, Park JY, Lee J, Lim JH, Shin EC, Ahn YS, Kim CH, Kim SJ, Kim JD, Choi IS, Choi IH (1999) Fas ligand and Fas are expressed constitutively in human astrocytes and the expression increases with IL-1, IL-6, TNF-alpha, or IFN-gamma. J Immunol 162:1889–1895
Coleman LG Jr, Crews FT (2018) Innate Immune Signaling and Alcohol Use Disorders. Handb Exp Pharmacol. https://doi.org/10.1007/164_2018_92
Crews FT, Boettiger CA (2009) Impulsivity, frontal lobes and risk for addiction. Pharmacol Biochem Behav 93:237–247. https://doi.org/10.1016/j.pbb.2009.04.018
Crews FT, Qin L, Sheedy D, Vetreno RP, Zou J (2013) High mobility group box 1/Toll-like receptor danger signaling increases brain neuroimmune activation in alcohol dependence. Biol Psychiatry 73:602–612. https://doi.org/10.1016/j.biopsych.2012.09.030
Crews FT, Lawrimore CJ, Walter TJ, Coleman LG Jr (2017) The role of neuroimmune signaling in alcoholism. Neuropharmacology 122:56–73. https://doi.org/10.1016/j.neuropharm.2017.01.031
Croft M, Siegel RM (2017) Beyond TNF: TNF superfamily cytokines as targets for the treatment of rheumatic diseases. Nat Rev Rheumatol 13:217–233. https://doi.org/10.1038/nrrheum.2017.22
Duncan JW, Johnson S, Zhang X, Zheng B, Luo J, Ou XM, Stockmeier CA, Wang JM (2016) Up-regulation of PKR signaling pathway by ethanol displays an age of onset-dependent relationship. Alcohol Clin Exp Res 40:2320–2328. https://doi.org/10.1111/acer.13209
Farina C, Krumbholz M, Giese T, Hartmann G, Aloisi F, Meinl E (2005) Preferential expression and function of Toll-like receptor 3 in human astrocytes. J Neuroimmunol 159:12–19. https://doi.org/10.1016/j.jneuroim.2004.09.009
Filiano AJ, Xu Y, Tustison NJ, Marsh RL, Baker W, Smirnov I, Overall CC, Gadani SP, Turner SD, Weng Z, Peerzade SN, Chen H, Lee KS, Scott MM, Beenhakker MP, Litvak V, Kipnis J (2016) Unexpected role of interferon-gamma in regulating neuronal connectivity and social behaviour. Nature 535:425–429. https://doi.org/10.1038/nature18626
Fritz M, Klawonn AM, Jaarola M, Engblom D (2018) Interferon- mediated signaling in the brain endothelium is critical for inflammation-induced aversion. Brain Behav Immun 67:54–58. https://doi.org/10.1016/j.bbi.2017.08.020
Gay N et al (2013) Preconditioning of microglia by α-synuclein strongly affects the response induced by Toll-like receptor (TLR) stimulation. PLoS One 8:e79160. https://doi.org/10.1371/journal.pone.0079160
Haydon PG, Nedergaard M (2014) How do astrocytes participate in neural plasticity? Cold Spring Harb Perspect Biol 7:a020438. https://doi.org/10.1101/cshperspect.a020438
Henn A, Lund S, Hedtjarn M, Schrattenholz A, Porzgen P, Leist M (2009) The suitability of BV2 cells as alternative model system for primary microglia cultures or for animal experiments examining brain inflammation. Altex 26:83–94
Henry CM, Martin SJ (2017) Caspase-8 acts in a non-enzymatic role as a scaffold for assembly of a pro-inflammatory “FADDosome” complex upon TRAIL stimulation. Mol Cell 65:715–729 e715. https://doi.org/10.1016/j.molcel.2017.01.022
Huang Y, Walstrom A, Zhang L, Zhao Y, Cui M, Ye L, Zheng JC (2009) Type I interferons and interferon regulatory factors regulate TNF-related apoptosis-inducing ligand (TRAIL) in HIV-1-infected macrophages. PLoS One 4:e5397. https://doi.org/10.1371/journal.pone.0005397
Imaizumi T, Murakami K, Ohta K, Seki H, Matsumiya T, Meng P, Hayakari R, Xing F, Aizawa-Yashiro T, Tatsuta T, Yoshida H, Kijima H (2013) MDA5 and ISG56 mediate CXCL10 expression induced by toll-like receptor 4 activation in U373MG human astrocytoma cells. Neurosci Res 76:195–206. https://doi.org/10.1016/j.neures.2013.05.002
Imaizumi T, Numata A, Yano C, Yoshida H, Meng P, Hayakari R, Xing F, Wang L, Matsumiya T, Tanji K, Tatsuta T, Murakami M, Tanaka H (2014) ISG54 and ISG56 are induced by TLR3 signaling in U373MG human astrocytoma cells: possible involvement in CXCL10 expression. Neurosci Res 84:34–42. https://doi.org/10.1016/j.neures.2014.03.001
Jang Y, Lee MH, Park JH, Han SY, Kim DK (2016) TLR3 deficiency increases voluntary alcohol consumption. Neuroreport 27:356–360. https://doi.org/10.1097/WNR.0000000000000549
Johnson S, Duncan J, Hussain SA, Chen G, Luo J, Mclaurin C, May W, Rajkowska G, Ou XM, Stockmeier CA, Wang JM (2015) The IFNgamma-PKR pathway in the prefrontal cortex reactions to chronic excessive alcohol use. Alcohol Clin Exp Res 39:476–484. https://doi.org/10.1111/acer.12650
Koob GF (2015) The dark side of emotion: the addiction perspective. Eur J Pharmacol 753:73–87. https://doi.org/10.1016/j.ejphar.2014.11.044
Koob GF, Volkow ND (2010) Neurocircuitry of addiction. Neuropsychopharmacology 35:217–238. https://doi.org/10.1038/npp.2009.110
Korecka JA, van Kesteren RE, Blaas E, Spitzer SO, Kamstra JH, Smit AB, Swaab DF, Verhaagen J, Bossers K (2013) Phenotypic characterization of retinoic acid differentiated SH-SY5Y cells by transcriptional profiling. PLoS One 8:e63862. https://doi.org/10.1371/journal.pone.0063862
Kovalevich J, Langford D (2013) Considerations for the use of SH-SY5Y neuroblastoma cells in neurobiology. Methods Mol Biol 1078:9–21. https://doi.org/10.1007/978-1-62703-640-5_2
Kumar-Sinha C, Varambally S, Sreekumar A, Chinnaiyan AM (2002) Molecular cross-talk between the TRAIL and interferon signaling pathways. J Biol Chem 277:575–585. https://doi.org/10.1074/jbc.M107795200
Lawrimore CJ, Crews FT (2017) Ethanol, TLR3, and TLR4 agonists have unique innate immune responses in neuron-like SH-SY5Y and microglia-like BV2. Alcohol Clin Exp Res 41:939–954. https://doi.org/10.1111/acer.13368
LeBlanc HN, Ashkenazi A (2003) Apo2L/TRAIL and its death and decoy receptors. Cell Death Differ 10:66–75. https://doi.org/10.1038/sj.cdd.4401187
Lehmann SM, Rosenberger K, Kruger C, Habbel P, Derkow K, Kaul D, Rybak A, Brandt C, Schott E, Wulczyn FG, Lehnardt S (2012) Extracellularly delivered single-stranded viral RNA causes neurodegeneration dependent on TLR7. J Immunol 189:1448–1458. https://doi.org/10.4049/jimmunol.1201078
Levy DE, Darnell JE Jr (2002) Stats: transcriptional control and biological impact. Nat Rev Mol Cell Biol 3:651–662. https://doi.org/10.1038/nrm909
Lian H, Yang L, Cole A, Sun L, Chiang ACA, Fowler SW, Shim DJ, Rodriguez-Rivera J, Taglialatela G, Jankowsky JL, Lu HC, Zheng H (2015) NFkappaB-activated astroglial release of complement C3 compromises neuronal morphology and function associated with Alzheimer’s disease. Neuron 85:101–115. https://doi.org/10.1016/j.neuron.2014.11.018
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Münch AE, Chung WS, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM, Stevens B, Barres BA (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541:481–487. https://doi.org/10.1038/nature21029
Lippai D, Bala S, Petrasek J, Csak T, Levin I, Kurt-Jones EA, Szabo G (2013) Alcohol-induced IL-1beta in the brain is mediated by NLRP3/ASC inflammasome activation that amplifies neuroinflammation. J Leukoc Biol 94:171–182. https://doi.org/10.1189/jlb.1212659
Lok KZ, Basta M, Manzanero S, Arumugam TV (2015) Intravenous immunoglobulin (IVIg) dampens neuronal Toll-like receptor-mediated responses in ischemia. J Neuroinflammation 12:73. https://doi.org/10.1186/s12974-015-0294-8
Mariani SM, Krammer PH (1998) Differential regulation of TRAIL and CD95 ligand in transformed cells of the T and B lymphocyte lineage. Eur J Immunol 28:973–982. https://doi.org/10.1002/(sici)1521-4141(199803)28:03<973::Aid-immu973>3.0.Co;2-t
Mayfield J, Ferguson L, Harris RA (2013) Neuroimmune signaling: a key component of alcohol abuse. Curr Opin Neurobiol 23:513–520. https://doi.org/10.1016/j.conb.2013.01.024
McCarthy GM, Bridges CR, Blednov YA, Harris RA (2017a) CNS cell-type localization and LPS response of TLR signaling pathways. F1000Res 6:1144. https://doi.org/10.12688/f1000research.12036.1
McCarthy GM, Warden AS, Bridges CR, Blednov YA, Harris RA (2017b) Chronic ethanol consumption: role of TLR3/TRIF-dependent signaling. Addict Biol. https://doi.org/10.1111/adb.12539
McDonough A, Lee RV, Noor S, Lee C, le T, Iorga M, Phillips JLH, Murphy S, Möller T, Weinstein JR (2017) Ischemia/reperfusion induces interferon-stimulated gene expression in microglia. J Neurosci 37:8292–8308. https://doi.org/10.1523/JNEUROSCI.0725-17.2017
Mina VA et al (2015) The influence of inflammatory cytokines in physiopathology of suicidal behavior. J Affect Disord 172:219–230. https://doi.org/10.1016/j.jad.2014.09.057
Nitsch R, Bechmann I, Deisz RA, Haas D, Lehmann TN, Wendling U, Zipp F (2000) Human brain-cell death induced by tumour-necrosis-factor-related apoptosis-inducing ligand (TRAIL). Lancet 356:827–828. https://doi.org/10.1016/S0140-6736(00)02659-3
Pan G, O'Rourke K, Chinnaiyan AM, Gentz R, Ebner R, Ni J, Dixit VM (1997) The receptor for the cytotoxic ligand TRAIL. Science 276:111–113
Pascual M, Balino P, Aragon CM, Guerri C (2015) Cytokines and chemokines as biomarkers of ethanol-induced neuroinflammation and anxiety-related behavior: role of TLR4 and TLR2. Neuropharmacology 89:352–359. https://doi.org/10.1016/j.neuropharm.2014.10.014
Peteranderl C, Herold S (2017) The impact of the interferon/TNF-related apoptosis-inducing ligand signaling axis on disease progression in respiratory viral infection and beyond. Front Immunol 8:313. https://doi.org/10.3389/fimmu.2017.00313
Pinto EF, Andrade C (2016) Interferon-related depression: a primer on mechanisms, treatment, and prevention of a common clinical problem. Curr Neuropharmacol 14:743–748
Qin L, Crews FT (2012) Chronic ethanol increases systemic TLR3 agonist-induced neuroinflammation and neurodegeneration. J Neuroinflammation 9:130. https://doi.org/10.1186/1742-2094-9-130
Qin L, He J, Hanes RN, Pluzarev O, Hong JS, Crews FT (2008) Increased systemic and brain cytokine production and neuroinflammation by endotoxin following ethanol treatment. J Neuroinflammation 5:10. https://doi.org/10.1186/1742-2094-5-10
Raison CL, Borisov AS, Majer M, Drake DF, Pagnoni G, Woolwine BJ, Vogt GJ, Massung B, Miller AH (2009) Activation of central nervous system inflammatory pathways by interferon-alpha: relationship to monoamines and depression. Biol Psychiatry 65:296–303. https://doi.org/10.1016/j.biopsych.2008.08.010
Rosenberger AF et al (2016) Increased occurrence of protein kinase CK2 in astrocytes in Alzheimer’s disease pathology. J Neuroinflammation 13:4. https://doi.org/10.1186/s12974-015-0470-x
Ruggeri P, Cappabianca L, Farina AR, Gneo L, Mackay AR (2016) NGF sensitizes TrkA SH-SY5Y neuroblastoma cells to TRAIL-induced apoptosis. Cell Death Discov 2:16004. https://doi.org/10.1038/cddiscovery.2016.4
Sato K et al (2001) An tiviral response by natural killer cells through TRAIL gene induction by IFN-alpha/beta. Eur J Immunol 31:3138–3146. https://doi.org/10.1002/1521-4141(200111)31:11<3138::AID-IMMU3138>3.0.CO;2-B
Schoenbaum G, Shaham Y (2008) The role of orbitofrontal cortex in drug addiction: a review of preclinical studies. Biol Psychiatry 63:256–262. https://doi.org/10.1016/j.biopsych.2007.06.003
Sen GC, Sarkar SN (2005) Transcriptional signaling by double-stranded RNA: role of TLR3. Cytokine Growth Factor Rev 16:1–14. https://doi.org/10.1016/j.cytogfr.2005.01.006
Serramia MJ, Munoz-Fernandez MA, Alvarez S (2015) HIV-1 increases TLR responses in human primary astrocytes. Sci Rep 5:17887. https://doi.org/10.1038/srep17887
Sheridan GK, Wdowicz A, Pickering M, Watters O, Halley P, O’Sullivan NC, Mooney C, O’Connell DJ, O’Connor JJ, Murphy KJ (2014) CX3CL1 is up-regulated in the rat hippocampus during memory-associated synaptic plasticity. Front Cell Neurosci 8:233. https://doi.org/10.3389/fncel.2014.00233
Shin JH, Lee HK, Lee HB, Jin Y, Lee JK (2014) Ethyl pyruvate inhibits HMGB1 phosphorylation and secretion in activated microglia and in the postischemic brain. Neurosci Lett 558:159–163. https://doi.org/10.1016/j.neulet.2013.11.006
Tang SC, Arumugam TV, Xu X, Cheng A, Mughal MR, Jo DG, Lathia JD, Siler DA, Chigurupati S, Ouyang X, Magnus T, Camandola S, Mattson MP (2007) Pivotal role for neuronal Toll-like receptors in ischemic brain injury and functional deficits. Proc Natl Acad Sci U S A 104:13798–13803. https://doi.org/10.1073/pnas.0702553104
Trindade P, Hampton B, Manhaes AC, Medina AE (2016) Developmental alcohol exposure leads to a persistent change on astrocyte secretome. J Neurochem 137:730–743. https://doi.org/10.1111/jnc.13542
Valles SL, Blanco AM, Pascual M, Guerri C (2004) Chronic ethanol treatment enhances inflammatory mediators and cell death in the brain and in astrocytes. Brain Pathol 14:365–371
Vetreno RP, Crews FT (2012) Adolescent binge drinking increases expression of the danger signal receptor agonist HMGB1 and Toll-like receptors in the adult prefrontal cortex. Neuroscience 226:475–488. https://doi.org/10.1016/j.neuroscience.2012.08.046
Vetreno RP, Qin L, Crews FT (2013) Increased receptor for advanced glycation end product expression in the human alcoholic prefrontal cortex is linked to adolescent drinking. Neurobiol Dis 59:52–62. https://doi.org/10.1016/j.nbd.2013.07.002
Volkow ND, Koob GF, McLellan AT (2016) Neurobiologic advances from the brain disease model of addiction. N Engl J Med 374:363–371. https://doi.org/10.1056/NEJMra1511480
Walter TJ, Crews FT (2017) Microglial depletion alters the brain neuroimmune response to acute binge ethanol withdrawal. J Neuroinflammation 14:86. https://doi.org/10.1186/s12974-017-0856-z
Wills TA, Knapp DJ, Overstreet DH, Breese GR (2009) Sensitization, duration, and pharmacological blockade of anxiety-like behavior following repeated ethanol withdrawal in adolescent and adult rats. Alcohol Clin Exp Res 33:455–463. https://doi.org/10.1111/j.1530-0277.2008.00856.x
Wilson NS, Dixit V, Ashkenazi A (2009) Death receptor signal transducers: nodes of coordination in immune signaling networks. Nat Immunol 10:348–355. https://doi.org/10.1038/ni.1714
Xicoy H, Wieringa B, Martens GJ (2017) The SH-SY5Y cell line in Parkinson’s disease research: a systematic review. Mol Neurodegener 12:10. https://doi.org/10.1186/s13024-017-0149-0
Xie HR, Hu LS, Li GY (2010) SH-SY5Y human neuroblastoma cell line: in vitro cell model of dopaminergic neurons in Parkinson’s disease. Chin Med J 123:1086–1092
Zheng LS, Hitoshi S, Kaneko N, Takao K, Miyakawa T, Tanaka Y, Xia H, Kalinke U, Kudo K, Kanba S, Ikenaka K, Sawamoto K (2014) Mechanisms for interferon-alpha-induced depression and neural stem cell dysfunction. Stem Cell Rep 3:73–84. https://doi.org/10.1016/j.stemcr.2014.05.015
Funding
NIH-NIAAA P60 AA011605, U01 AA020023, U24 AA020024, U54 AA019767, T32AA007573, K08AA024829, 3U01AA020023-09S1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to a Special Issue on Neuroimmune Signaling in Psychiatric Disease
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lawrimore, C.J., Coleman, L.G. & Crews, F.T. Ethanol induces interferon expression in neurons via TRAIL: role of astrocyte-to-neuron signaling. Psychopharmacology 236, 2881–2897 (2019). https://doi.org/10.1007/s00213-018-5153-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-5153-8