Abstract
Rationale
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by memory impairment, neuronal death, and synaptic loss in the hippocampus. Long-term potentiation (LTP), a type of synaptic plasticity, occurs during learning and memory. Serotonin receptor type 7 (5-HTR7) activation is suggested as a possible therapeutic target for AD.
Objective
The aim of the present study was to examine the effects of chronic treatment with the 5-HTR7 agonist, AS19, on cognitive function, memory, hippocampal plasticity, amyloid beta (Aβ) plaque accumulation, and apoptosis in an adult rat model of AD.
Methods
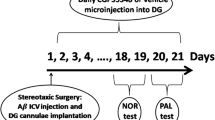
AD was induced in rats using Aβ (single 1 μg/μL intracerebroventricular (icv) injection during surgery). The following experimental groups were included: control, sham-operated, Aβ + saline (1 μL icv for 30 days), and Aβ + AS19 (1 μg/μL icv for 30 days) groups. The animals were tested for cognition and memory performance using the novel object recognition and passive avoidance tests, respectively. Next, anesthetized rats were placed in a stereotaxic apparatus for electrode implantation, and field potentials were recorded in the hippocampal dentate gyrus. Lastly, brains were removed and Aβ plaques and neuronal apoptosis were evaluated using Congo red staining and TUNEL assay, respectively.
Results
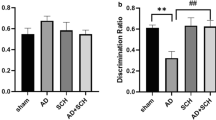
Administration of AS19 in the Aβ rats increased the discrimination index of the novel object recognition test. Furthermore, AS19 treatment decreased time spent in the dark compartment during the passive avoidance test. AS19 also enhanced both the population spike (PS) amplitude and the field excitatory postsynaptic potential (fEPSP) slope evoked potentials of the LTP components. Aβ plaques and neuronal apoptosis were decreased in the AS19-treated Aβ rats.
Conclusions
These results indicate that chronic treatment with a 5-HTR7 agonist can prevent Aβ-related impairments in cognition and memory performance by alleviating Aβ plaque accumulation and neuronal apoptosis, hence improving neuronal plasticity. AS19 may be useful as a therapeutic agent for AD.






Similar content being viewed by others
References
Alberghina L, Colangelo AM (2006) The modular systems biology approach to investigate the control of apoptosis in Alzheimer’s disease neurodegeneration. BMC Neurosci 7(Suppl 1):S2. https://doi.org/10.1186/1471-2202-7-s1-s2
Andersen P, Bliss TV, Skrede KK (1971) Lamellar organization of hippocampal pathways. Exp Brain Res 13:222–238
Anderson AJ, Stoltzner S, Lai F, Su J, Nixon RA (2000) Morphological and biochemical assessment of DNA damage and apoptosis in Down syndrome and Alzheimer disease, and effect of postmortem tissue archival on TUNEL. Neurobiol Aging 21:511–524
Babri S, Mohaddes G, Feizi I, Mohammadnia A, Niapour A, Alihemmati A, Amani M (2014) Effect of troxerutin on synaptic plasticity of hippocampal dentate gyrus neurons in a beta-amyloid model of Alzheimers disease: an electrophysiological study. Eur J Pharmacol 732:19–25. https://doi.org/10.1016/j.ejphar.2014.03.018
Bacon WL, Beck SG (2000) 5-Hydroxytryptamine7 receptor activation decreases slow after hyperpolarization amplitude in CA3 hippocampal pyramidal cells. J Pharmacol Exp Ther 294:672–679
Barzegar S, Komaki A, Shahidi S, Sarihi A, Mirazi N, Salehi I (2015) Effects of cannabinoid and glutamate receptor antagonists and their interactions on learning and memory in male rats. Pharmacol Biochem Behav 131:87–90. https://doi.org/10.1016/j.pbb.2015.02.005
Berumen LC, Rodriguez A, Miledi R, Garcia-Alcocer G (2012) Serotonin receptors in hippocampus TheScientificWorldJOURNAL 2012:823493 doi:https://doi.org/10.1100/2012/823493, 1, 15
Bickmeyer U, Heine M, Manzke T, Richter DW (2002) Differential modulation of I(h) by 5-HT receptors in mouse CA1 hippocampal neurons. Eur J Neurosci 16:209–218
Bliss TV, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31–39. https://doi.org/10.1038/361031a0
Bliss TV, Gardner-Medwin AR (1973) Long-lasting potentiation of synaptic transmission in the dentate area of the unanaestetized rabbit following stimulation of the perforant path. The J Physiol 232:357–374
Butzlaff M, Ponimaskin E (2016) The role of serotonin receptors in Alzheimer’s disease. Opera Medica et Physiologica 2:77–86
Chapman PF, White GL, Jones MW, Cooper-Blacketer D, Marshall VJ, Irizarry M, Younkin L, Good MA, Bliss TVP, Hyman BT, Younkin SG, Hsiao KK (1999) Impaired synaptic plasticity and learning in aged amyloid precursor protein transgenic mice. Nat Neurosci 2:271–276. https://doi.org/10.1038/6374
Chen G, Chen KS, Knox J, Inglis J, Bernard A, Martin SJ, Justice A, McConlogue L, Games D, Freedman SB, Morris RGM (2000) A learning deficit related to age and beta-amyloid plaques in a mouse model of Alzheimer’s disease. Nature 408:975–979. https://doi.org/10.1038/35050103
Chen KH, Reese EA, Kim HW, Rapoport SI, Rao JS (2011) Disturbed neurotransmitter transporter expression in Alzheimer’s disease brain. Journal of Alzheimer’s disease : JAD 26:755–766. https://doi.org/10.3233/jad-2011-110002
Citri A, Malenka RC (2008) Synaptic plasticity: multiple forms, functions, and mechanisms. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology 33:18–41. https://doi.org/10.1038/sj.npp.1301559
Costa L, Sardone LM, Lacivita E, Leopoldo M, Ciranna L (2015) Novel agonists for serotonin 5-HT7 receptors reverse metabotropic glutamate receptor-mediated long-term depression in the hippocampus of wild-type and Fmr1 KO mice, a model of Fragile X Syndrome. Front Behav Neurosci 9:65. https://doi.org/10.3389/fnbeh.2015.00065
Costa L, Spatuzza M, D’Antoni S, Bonaccorso CM, Trovato C, Musumeci SA, Leopoldo M, Lacivita E, Catania MV, Ciranna L (2012) Activation of 5-HT7 serotonin receptors reverses metabotropic glutamate receptor-mediated synaptic plasticity in wild-type and Fmr1 knockout mice, a model of Fragile X syndrome. Biol Psychiatry 72:924–933. https://doi.org/10.1016/j.biopsych.2012.06.008
Costa L, Trovato C, Musumeci SA, Catania MV, Ciranna L (2012) 5-HT(1A) and 5-HT(7) receptors differently modulate AMPA receptor-mediated hippocampal synaptic transmission. Hippocampus 22:790–801. https://doi.org/10.1002/hipo.20940
De Filippis B et al (2015) Long-lasting beneficial effects of central serotonin receptor 7 stimulation in female mice modeling Rett syndrome. Front Behav Neurosci 9:86. https://doi.org/10.3389/fnbeh.2015.00086
Dengler-Crish CM, Smith MA, Wilson GN (2017) Early evidence of low bone density and decreased serotonergic synthesis in the dorsal raphe of a tauopathy model of Alzheimer’s disease. Journal of Alzheimer’s disease : JAD 55:1605–1619. https://doi.org/10.3233/jad-160658
Fidalgo S, Ivanov DK, Wood SH (2013) Serotonin: from top to bottom. Biogerontology 14:21–45. https://doi.org/10.1007/s10522-012-9406-3
Filip M, Bader M (2009) Overview on 5-HT receptors and their role in physiology and pathology of the central nervous system. Pharmacological reports : PR 61:761–777
Fisher JR, Wallace CE, Tripoli DL, Sheline YI, Cirrito JR (2016) Redundant Gs-coupled serotonin receptors regulate amyloid-beta metabolism in vivo. Mol Neurodegener 11:45. https://doi.org/10.1186/s13024-016-0112-5
Garcia-Alloza M, Hirst WD, Chen CP, Lasheras B, Francis PT, Ramirez MJ (2004) Differential involvement of 5-HT(1B/1D) and 5-HT6 receptors in cognitive and non-cognitive symptoms in Alzheimer’s disease. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology 29:410–416. https://doi.org/10.1038/sj.npp.1300330
Gasbarri A, Cifariello A, Pompili A, Meneses A (2008) Effect of 5-HT 7 antagonist SB-269970 in the modulation of working and reference memory in the rat. Behav Brain Res 195:164–170
Ghahremanitamadon F, Shahidi S, Zargooshnia S, Nikkhah A, Ranjbar A, Soleimani Asl S (2014) Protective effects of Borago officinalis extract on amyloid beta-peptide(25-35)-induced memory impairment in male rats: a behavioral study Biomed Res Int 2014:798535 doi:https://doi.org/10.1155/2014/798535, 1, 8
Gorevic PD, Schur PH, Romain PL (2011) Overview of amyloidosis. PATHOGENESIS 5:6
Han LN, Zhang L, Sun YN, du CX, Zhang YM, Wang T, Zhang J, Liu J (2016) Serotonin 7 receptors in the lateral habenular nucleus regulate depressive-like behaviors in the hemiparkinsonian rats. Brain Res 1644:79–87
Hasanein P, Shahidi S (2012) Preventive effect of Teucrium polium on learning and memory deficits in diabetic rats Medical science monitor : international medical journal of experimental and clinical research 18:Br41-46
Hashemi-Firouzi N, Akhavan M, Komaki A, Shahidi S (2015) Effects of acute administration of Urtica dioica on the novel object-recognition task in mice. Avicenna Journal of Neuro Psych Physiology 2
Hauser SR, Hedlund PB, Roberts AJ, Sari Y, Bell RL, Engleman EA (2014) The 5-HT7 receptor as a potential target for treating drug and alcohol abuse. Front Neurosci 8:448. https://doi.org/10.3389/fnins.2014.00448
Hedlund PB (2009) The 5-HT7 receptor and disorders of the nervous system: an overview. Psychopharmacology 206:345–354. https://doi.org/10.1007/s00213-009-1626-0
Hoyer S, Lannert H (2007) Long-term abnormalities in brain glucose/energy metabolism after inhibition of the neuronal insulin receptor: implication of tau-protein. J Neural Transm Suppl:195–202
Jabbarpour Z, Shahidi S, Saidijam M, Sarihi A, Hassanzadeh T, Esmaeili R (2014) Effect of tempol on the passive avoidance and novel object recognition task in diabetic rats. Brain Res Bull 101:51–56. https://doi.org/10.1016/j.brainresbull.2013.12.013
Jacobsen JS et al. (2006) Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer’s disease Proceedings of the National Academy of Sciences of the United States of America 103:5161-5166 doi:https://doi.org/10.1073/pnas.0600948103
Karamian R, Komaki A, Salehi I, Tahmasebi L, Komaki H, Shahidi S, Sarihi A (2015) Vitamin C reverses lead-induced deficits in hippocampal synaptic plasticity in rats. Brain Res Bull 116:7–15. https://doi.org/10.1016/j.brainresbull.2015.05.004
Karimi SA, Komaki A, Salehi I, Sarihi A, Shahidi S (2015) Role of group II metabotropic glutamate receptors (mGluR2/3) blockade on long-term potentiation in the dentate gyrus region of hippocampus in rats fed with high-fat diet. Neurochem Res 40:811–817. https://doi.org/10.1007/s11064-015-1531-3
Kawasaki H, Springett GM, Mochizuki N, Toki S, Nakaya M, Matsuda M, Housman DE, Graybiel AM (1998) A family of cAMP-binding proteins that directly activate Rap1. Science 282:2275–2279
Kazim SF, Blanchard J, Dai CL, Tung YC, LaFerla FM, Iqbal IG, Iqbal K (2014) Disease modifying effect of chronic oral treatment with a neurotrophic peptidergic compound in a triple transgenic mouse model of Alzheimer’s disease. Neurobiol Dis 71:110–130. https://doi.org/10.1016/j.nbd.2014.07.001
Khodamoradi N, Komaki A, Salehi I, Shahidi S, Sarihi A (2015) Effect of vitamin E on lead exposure-induced learning and memory impairment in rats. Physiol Behav 144:90–94. https://doi.org/10.1016/j.physbeh.2015.03.015
Knobloch M, Farinelli M, Konietzko U, Nitsch RM, Mansuy IM (2007) Abeta oligomer-mediated long-term potentiation impairment involves protein phosphatase 1-dependent mechanisms. The Journal of neuroscience : the official journal of the Society for Neuroscience 27:7648–7653. https://doi.org/10.1523/jneurosci.0395-07.2007
Kobe F, Guseva D, Jensen TP, Wirth A, Renner U, Hess D, Muller M, Medrihan L, Zhang W, Zhang M, Braun K, Westerholz S, Herzog A, Radyushkin K, el-Kordi A, Ehrenreich H, Richter DW, Rusakov DA, Ponimaskin E (2012) 5-HT7R/G12 signaling regulates neuronal morphology and function in an age-dependent manner. The Journal of neuroscience : the official journal of the Society for Neuroscience 32:2915–2930. https://doi.org/10.1523/jneurosci.2765-11.2012
Komaki A, Karimi SA, Salehi I, Sarihi A, Shahidi S, Zarei M (2015) The treatment combination of vitamins E and C and astaxanthin prevents high-fat diet induced memory deficits in rats. Pharmacol Biochem Behav 131:98–103. https://doi.org/10.1016/j.pbb.2015.02.008
Komaki A, Khalili A, Salehi I, Shahidi S, Sarihi A (2014) Effects of exposure to an extremely low frequency electromagnetic field on hippocampal long-term potentiation in rat. Brain Res 1564:1–8. https://doi.org/10.1016/j.brainres.2014.03.041
Krafft GA, Klein WL (2010) ADDLs and the signaling web that leads to Alzheimer’s disease. Neuropharmacology 59:230–242. https://doi.org/10.1016/j.neuropharm.2010.07.012
Lacor PN, Buniel MC, Chang L, Fernandez SJ, Gong Y, Viola KL, Lambert MP, Velasco PT, Bigio EH, Finch CE, Krafft GA, Klein WL (2004) Synaptic targeting by Alzheimer’s-related amyloid beta oligomers. The Journal of neuroscience : the official journal of the Society for Neuroscience 24:10191–10200. https://doi.org/10.1523/jneurosci.3432-04.2004
Lashgari R, Khakpour-Taleghani B, Motamedi F, Shahidi S (2008) Effects of reversible inactivation of locus coeruleus on long-term potentiation in perforant path-DG synapses in rats. Neurobiol Learn Mem 90:309–316. https://doi.org/10.1016/j.nlm.2008.05.012
Leopoldo M, Lacivita E, Berardi F, Perrone R, Hedlund PB (2011) Serotonin 5-HT 7 receptor agents: structure-activity relationships and potential therapeutic applications in central nervous system disorders. Pharmacol Ther 129:120–148
Li YH, Han L, Wu KLK, Chan YS (2017) Activation of 5-HT7 receptors reverses NMDA-R-dependent LTD by activating PKA in medial vestibular neurons. Neuropharmacology 123:242–248. https://doi.org/10.1016/j.neuropharm.2017.05.005
Luscher C, Malenka RC (2012) NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb Perspect Biol 4. https://doi.org/10.1101/cshperspect.a005710
Matsumoto M, Kojima T, Togashi H, Mori K, Ohashi S, Ueno K, Yoshioka M (2002) Differential characteristics of endogenous serotonin-mediated synaptic transmission in the hippocampal CA1 and CA3 fields of anaesthetized rats. Naunyn Schmiedeberg's Arch Pharmacol 366:570–577. https://doi.org/10.1007/s00210-002-0634-y
Meneses A (2014) Memory formation and memory alterations: 5-HT6 and 5-HT7 receptors, novel alternative. Rev Neurosci 25:325–356. https://doi.org/10.1515/revneuro-2014-0001
Mucke L, Masliah E, Yu GQ, Mallory M, Rockenstein EM, Tatsuno G, Hu K, Kholodenko D, Johnson-Wood K, McConlogue L (2000) High-level neuronal expression of Aβ1–42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J Neurosci 20:4050–4058
Mufson EJ, Ikonomovic MD, Counts SE, Perez SE, Malek-Ahmadi M, Scheff SW, Ginsberg SD (2016) Molecular and cellular pathophysiology of preclinical Alzheimer’s disease. Behav Brain Res 311:54–69. https://doi.org/10.1016/j.bbr.2016.05.030
Nativio P et al. (2015) Stimulation of 5-HT7 receptor during adolescence determines its persistent upregulation in adult rat forebrain areas Synapse (New York, NY) 69:533-542 doi:https://doi.org/10.1002/syn.21846
Nikiforuk A, Kos T, Fijal K, Holuj M, Rafa D, Popik P (2013) Effects of the selective 5-HT7 receptor antagonist SB-269970 and amisulpride on ketamine-induced schizophrenia-like deficits in rats. PLoS One 8:e66695. https://doi.org/10.1371/journal.pone.0066695
Obulesu M, Lakshmi MJ (2014) Apoptosis in Alzheimer’s disease: an understanding of the physiology, pathology and therapeutic avenues. Neurochem Res 39:2301–2312. https://doi.org/10.1007/s11064-014-1454-4
Palmer AM, Francis PT, Benton JS, Sims NR, Mann DMA, Neary D, Snowden JS, Bowen DM (1987) Presynaptic serotonergic dysfunction in patients with Alzheimer’s disease. J Neurochem 48:8–15
Park SW, Jang HJ, Cho KH, Kim MJ, Yoon SH (2012) Rhie DJ, Developmental switch of the serotonergic role in the induction of synaptic long-term potentiation in the rat visual cortex. The Korean journal of physiology & pharmacology : official journal of the Korean Physiological Society and the Korean Society of Pharmacology 16:65–70. https://doi.org/10.4196/kjpp.2012.16.1.65
Paxinos GW C. (2007) The rat brain in stereotaxic coordinates Burlington MA Elsevier Inc
Perez-Garcia G, Gonzalez-Espinosa C, Meneses A (2006) An mRNA expression analysis of stimulation and blockade of 5-HT7 receptors during memory consolidation. Behav Brain Res 169:83–92. https://doi.org/10.1016/j.bbr.2005.12.013
Perez-Garcia G, Meneses A (2009) Memory time-course: mRNA 5-HT1A and 5-HT7 receptors. Behav Brain Res 202:102–113. https://doi.org/10.1016/j.bbr.2009.03.027
Perez-Garcia GS, Meneses A (2005) Effects of the potential 5-HT7 receptor agonist AS 19 in an autoshaping learning task. Behav Brain Res 163:136–140. https://doi.org/10.1016/j.bbr.2005.04.014
Pourheydar B, Soleimani Asl S, Azimzadeh M, Rezaei Moghadam A, Marzban A, Mehdizadeh M (2016) Neuroprotective Effects of Bone Marrow Mesenchymal Stem Cells on Bilateral Common Carotid Arteries Occlusion Model of Cerebral Ischemia in Rat. Behavioural Neurology 2016:2964712–2964710. https://doi.org/10.1155/2016/2964712
Querfurth HW, LaFerla FM (2010) Alzheimer’s disease. N Engl J Med 362:329–344. https://doi.org/10.1056/NEJMra0909142
Radi E, Formichi P, Battisti C, Federico A (2014) Apoptosis and oxidative stress in neurodegenerative diseases. Journal of Alzheimer’s disease : JAD 42(Suppl 3):S125–S152. https://doi.org/10.3233/jad-132738
Salehi I, Karamian R, Komaki A, Tahmasebi L, Taheri M, Nazari M, Shahidi S, Sarihi A (2015) Effects of vitamin E on lead-induced impairments in hippocampal synaptic plasticity. Brain Res 1629:270–281. https://doi.org/10.1016/j.brainres.2015.10.007
Sarkisyan G, Hedlund PB (2009) The 5-HT7 receptor is involved in allocentric spatial memory information processing. Behav Brain Res 202:26–31. https://doi.org/10.1016/j.bbr.2009.03.011
Shahidi S, Hashemi-Firouzi N (2014) The effects of a 5-HT7 receptor agonist and antagonist on morphine withdrawal syndrome in mice. Neurosci Lett 578:27–32. https://doi.org/10.1016/j.neulet.2014.06.027
Shahidi S, Zargooshnia S, Asl SS, Komaki A, Sarihi A (2017) Influence of N-acetyl cysteine on beta-amyloid-induced Alzheimer’s disease in a rat model: a behavioral and electrophysiological study. Brain Res Bull 131:142–149. https://doi.org/10.1016/j.brainresbull.2017.04.001
Sloviter RS, Lomo T (2012) Updating the lamellar hypothesis of hippocampal organization. Frontiers in neural circuits 6:102. https://doi.org/10.3389/fncir.2012.00102
Soga F, Katoh N, Inoue T, Kishimoto S (2007) Serotonin activates human monocytes and prevents apoptosis. The Journal of investigative dermatology 127:1947–1955. https://doi.org/10.1038/sj.jid.5700824
Speranza L, Giuliano T, Volpicelli F, de Stefano ME, Lombardi L, Chambery A, Lacivita E, Leopoldo M, Bellenchi GC, di Porzio U, Crispino M, Perrone-Capano C (2015) Activation of 5-HT7 receptor stimulates neurite elongation through mTOR, Cdc42 and actin filaments dynamics. Front Behav Neurosci 9:62. https://doi.org/10.3389/fnbeh.2015.00062
Speranza L, Labus J, Volpicelli F, Guseva D, Lacivita E, Leopoldo M, Bellenchi GC, di Porzio U, Bijata M, Perrone-Capano C, Ponimaskin E (2017) Serotonin 5-HT7 receptor increases the density of dendritic spines and facilitates synaptogenesis in forebrain neurons. J Neurochem 141:647–661. https://doi.org/10.1111/jnc.13962
Stiedl O, Pappa E, Konradsson-Geuken A, Ogren SO (2015) The role of the serotonin receptor subtypes 5-HT1A and 5-HT7 and its interaction in emotional learning and memory. Front Pharmacol 6:162. https://doi.org/10.3389/fphar.2015.00162
Tahmasebi L, Komaki A, Karamian R, Shahidi S, Sarihi A, Salehi I, Nikkhah A (2015) The interactive role of cannabinoid and vanilloid systems in hippocampal synaptic plasticity in rats. Eur J Pharmacol 757:68–73. https://doi.org/10.1016/j.ejphar.2015.03.063
Takuma K, Hara Y, Kataoka S, Kawanai T, Maeda Y, Watanabe R, Takano E, Hayata-Takano A, Hashimoto H, Ago Y, Matsuda T (2014) Chronic treatment with valproic acid or sodium butyrate attenuates novel object recognition deficits and hippocampal dendritic spine loss in a mouse model of autism. Pharmacol Biochem Behav 126:43–49. https://doi.org/10.1016/j.pbb.2014.08.013
Taube JS, Schwartzkroin PA (1988) Mechanisms of long-term potentiation: EPSP/spike dissociation, intradendritic recordings, and glutamate sensitivity. The Journal of neuroscience : the official journal of the Society for Neuroscience 8:1632–1644
Tokarski K, Zahorodna A, Bobula B, Grzegorzewska M, Pitra P, Hess G (2005) Repeated administration of citalopram and imipramine alters the responsiveness of rat hippocampal circuitry to the activation of 5-HT7 receptors. Eur J Pharmacol 524:60–66. https://doi.org/10.1016/j.ejphar.2005.09.014
Tokarski K, Zahorodna A, Bobula B, Hess G (2003) 5-HT7 receptors increase the excitability of rat hippocampal CA1 pyramidal neurons. Brain Res 993:230–234
Vasefi MS, Yang K, Li J, Kruk JS, Heikkila JJ, Jackson MF, MacDonald JF, Beazely MA (2013) Acute 5-HT7 receptor activation increases NMDA-evoked currents and differentially alters NMDA receptor subunit phosphorylation and trafficking in hippocampal neurons. Molecular brain 6:24. https://doi.org/10.1186/1756-6606-6-24
Wang W, Pan YW, Zou J, Li T, Abel GM, Palmiter RD, Storm DR, Xia Z (2014) Genetic activation of ERK5 MAP kinase enhances adult neurogenesis and extends hippocampus-dependent long-term memory. The Journal of neuroscience : the official journal of the Society for Neuroscience 34:2130–2147. https://doi.org/10.1523/jneurosci.3324-13.2014
Waters KA, Stean TO, Hammond B, Virley DJ, Upton N, Kew JN, Hussain I (2012) Effects of the selective 5-HT(7) receptor antagonist SB-269970 in animal models of psychosis and cognition. Behav Brain Res 228:211–218. https://doi.org/10.1016/j.bbr.2011.12.009
Wesolowska A, Nikiforuk A, Stachowicz K (2006) Potential anxiolytic and antidepressant effects of the selective 5-HT7 receptor antagonist SB 269970 after intrahippocampal administration to rats. Eur J Pharmacol 553:185–190. https://doi.org/10.1016/j.ejphar.2006.09.064
Wesołowska A, Nikiforuk A, Stachowicz K, Tatarczyńska E (2006) Effect of the selective 5-HT7 receptor antagonist SB 269970 in animal models of anxiety and depression. Neuropharmacology 51:578–586. https://doi.org/10.1016/j.neuropharm.2006.04.017
Xiang K, Zhao X, Li Y, Zheng L, Wang J, Li YH (2016) Selective 5-HT7 receptor activation may enhance synaptic plasticity through N-methyl-D-aspartate (NMDA) receptor activity in the visual cortex. Curr Neurovasc Res 13:321–328
Xu Y, Yan J, Zhou P, Li J, Gao H, Xia Y (2012) Wang Q, Neurotransmitter receptors and cognitive dysfunction in Alzheimer’s disease and Parkinson’s disease. Progress in neurobiology 97:1–13. https://doi.org/10.1016/j.pneurobio.2012.02.002
Zhang QJ, du CX, Tan HH, Zhang L, Li LB, Zhang J, Niu XL, Liu J (2015) Activation and blockade of serotonin7 receptors in the prelimbic cortex regulate depressive-like behaviors in a 6-hydroxydopamine-induced Parkinson’s disease rat model. Neuroscience 311:45–55. https://doi.org/10.1016/j.neuroscience.2015.10.016
Zhang Y, McLaughlin R, Goodyer C, LeBlanc A (2002) Selective cytotoxicity of intracellular amyloid beta peptide1-42 through p53 and Bax in cultured primary human neurons. J Cell Biol 156:519–529. https://doi.org/10.1083/jcb.200110119
Acknowledgments
This work was supported by a grant (Grant number: 9312186737) from the Neurophysiology Research Centre, Hamadan University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animal care, treatment, and surgical procedures were approved by the ethics committees of the Hamadan University of Medical Sciences (Ref. No.: 6383), and performed according to the Guide for Care and Use of laboratory animals published by the National Institute of Health, USA (NIH Publication No. 85-23, revised 1985).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Shahidi, S., Asl, S.S., Komaki, A. et al. The effect of chronic stimulation of serotonin receptor type 7 on recognition, passive avoidance memory, hippocampal long-term potentiation, and neuronal apoptosis in the amyloid β protein treated rat. Psychopharmacology 235, 1513–1525 (2018). https://doi.org/10.1007/s00213-018-4862-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4862-3