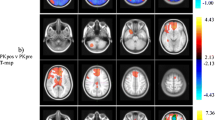
Abstract
Rationale
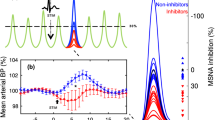
Peripheral physiologic changes accompany many central pharmacologic manipulations and can interact with brain activity and cerebral perfusion in complex ways. This considerably complicates the interpretation of drug-induced brain activity changes.
Objectives
To evaluate a method whereby drug-induced blood pressure (BP) changes are prevented.
Methods
A continuously adjusted infusion of the peripheral vasoconstrictor phenylephrine (PEP) was used to counter-regulate BP changes elicited by application of the dopamine receptor agonist apomorphine (APO) in the rat. Central effects of APO were measured using pharmacologic magnetic resonance imaging (phMRI) with blood oxygenation level dependent (BOLD) contrast at a field strength of 7 T.
Results
Compared to a NOPEP control group, the PEP blood pressure clamp successfully prevented BP changes and improved the detectability of central APO effects. Moreover, APO-induced central changes no longer correlated with BP time courses.
Conclusions
The method is suitable for isolating central drug effects from peripherally originating (BP) confounds in high-field functional magnetic resonance imaging (fMRI) studies. It may also be useful in fMRI studies of autonomic regulation, cognition, and emotion if the experimental manipulation entails BP changes.



Similar content being viewed by others
References
Auer DP, Elbel GK, Kalisch R, Delfino M, Czisch M, Winkelmann J, Murer MG, Gershanik O, Trenkwalder G (2001) Mapping of dopamine receptor hypersensitivity in an unilateral 6-OHDA model using BOLD imaging at 7T. Proc Int Soc Magn Reson Med 654
Bouthenet ML, Martres MP, Sales N, Schwartz JC (1987) A detailed mapping of dopamine D-2 receptors in rat central nervous system by autoradiography with [125I]iodosulpride. Neuroscience 20(1):117–155
Cenci MA, Kalen P, Mandel RJ, Wictorin K, Bjorklund A (1992) Dopaminergic transplants normalize amphetamine- and apomorphine-induced Fos expression in the 6-hydroxydopamine-lesioned striatum. Neuroscience 46(4):943–957
Delfino MA, Stefano AV, Ferrario JE, Taravini IR, Murer MG, Gershanik OS (2004) Behavioral sensitization to different dopamine agonists in a parkinsonian rodent model of drug-induced dyskinesias. Behav Brain Res 152(2):297–306
Green HD, Depaulis A (1956) Absence of vasomotor responses to epinephrine and arterenol in an isolated intracranial circulation. Circ Res 4:565–573
Greenfield JC Jr, Tindall GT (1968) Effect of norepinephrine, epinephrine, and angiotensin on blood flow in the internal carotid artery of man. J Clin Invest 47(7):1672–1684
Hebb MO, Robertson HA (1999) Motor effects and mapping of cerebral alterations in animal models of Parkinson’s and Huntington’s diseases. J Comp Neurol 410(1):99–114
James W (1884) What is an emotion? Mind 9:188–205
Kalisch R, Elbel GK, Gossl C, Czisch M, Auer DP (2001) Blood pressure changes induced by arterial blood withdrawal influence BOLD signal in anesthesized rats at 7 Tesla: implications for pharmacologic MRI. NeuroImage 14(4):891–898
Kannurpatti SS, Biswal BB (2004) Effect of anesthesia on CBF, MAP and fMRI-BOLD signal in response to apnea. Brain Res 1011(2):141–147
Labandeira-Garcia JL, Rozas G, Lopez-Martin E, Liste I, Guerra M (1996) Time course of striatal changes induced by 6-hydroxydopamine lesion of the nigrostriatal pathway, as studied by combined evaluation of rotational behavior and striatal Fos expression. Exp Brain Res 108(1):69–84
Luo F, Wu G, Li Z, Li SJ (2003) Characterization of effects of mean arterial blood pressure induced by cocaine and cocaine methiodide on BOLD signals in rat brain. Magn Reson Med 49(2):264–270
Mäkiranta MJ, Jauhiainen JPT, Oikarinen JT, Suominen K, Tervonen O, Alahuhta S, Jäntti V (2002) Functional magnetic resonance imaging of swine brain during change in thiopental anesthesia into EEG burst-suppression level—a preliminary study. MAGMA 15(1–3):27–35
Montorsi F, Perani D, Anchisi D, Salonia A, Scifo P, Rigiroli P, Deho F, De Vito ML, Heaton J, Rigatti P, Fazio F (2003a) Brain activation patterns during video sexual stimulation following the administration of apomorphine: results of a placebo-controlled study. Eur Urol 43(4):405–411
Montorsi F, Perani D, Anchisi D, Salonia A, Scifo P, Rigiroli P, Zanoni M, Heaton JP, Rigatti P, Fazio F (2003b) Apomorphine-induced brain modulation during sexual stimulation: a new look at central phenomena related to erectile dysfunction. Int J Impot Res 15(3):203–209
Murer MG, Dziewczapolski G, Menalled L, Garcia MC, Agid Y, Gershanik O, Raisman-Vozari R (1998) Chronic levodopa is not toxic for remaining dopaminergic neurons, but instead promotes their recovery, in rats with moderate nigrostriatal lesions. Ann Neurol 43:561–575
Nagaoka T, Harel N, Zhao F, Wang P, Kim SG (2002) Critical threshold of arterial blood pressure for BOLD response to visual stimulation in anesthesized cats. Annual Meeting of the International Society of Magnetic Resonance in Medicine (ISMRM) 2002, abstr. 1367
Nguyen TV, Brownell AL, Iris Chen YC, Livni E, Coyle JT, Rosen BR, Cavagna F, Jenkins BG (2000) Detection of the effects of dopamine receptor supersensitivity using pharmacological MRI and correlations with PET. Synapse 36(1):57–65
Olesen J (1972) The effect of intracarotid epinephrine, norepinephrine, and angiotensin on the regional cerebral blood flow in man. Neurology 22(9):978–987
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates. 4th edn. Academic, San Diego
Peters S, Suchan B, Rusin J, Daum I, Koster O, Przuntek H, Muller T, Schmid G (2003) Apomorphine reduces BOLD signal in fMRI during voluntary movement in Parkinsonian patients. NeuroReport 14(6):809–812
Savasta M, Dubois A, Benavides J, Scatton B (1986) Different neuronal location of [3H]SCH 23390 binding sites in pars reticulata and pars compacta of the substantia nigra in the rat. Neurosci Lett 72(3):265–271
Schallert T, Fleming SM, Leasure JL, Tillerson JL, Bland ST (2000) CNS plasticity and assessment of forelimb sensorimotor outcome in unilateral rat models of stroke, cortical ablation, parkinsonism and spinal cord injury. Neuropharmacology 39(5):777–787
Schmid G, Suchan B, Rusin J, Daum I, Koster O, Przuntek H, Muller T, Peters S (2004) Impact of apomorphine on BOLD signal during movement in normals. J Neural Transm Suppl 68:69–78
Schwarting RK, Huston JP (1996) Unilateral 6-hydroxydopamine lesions of meso-striatal dopamine neurons and their physiological sequelae. Prog Neurobiol 49(3):215–266
Tuor UI, McKenzie E, Tomanek B (2002) Functional magnetic resonance imaging of tonic pain and vasopressor effects in rats. Magn Reson Imaging 20:707–712
Ungerstedt U (1971) Postsynaptic supersensitivity after 6-hydroxy-dopamine induced degeneration of the nigrostriatal system. Acta Physiol Scand 82(Suppl 367):69–93
Wooten GF, Collins RC (1983) Effects of dopaminergic stimulation on functional brain metabolism in rats with unilateral substantia nigra lesions. Brain Res 263(2):267–275
Acknowledgements
We thank A. Yassouridis for help with statistics. All procedures described in “Materials and methods” followed the “Principles of laboratory animal care” http://www.nap.edu/readingroom/books/labrats and were approved by local authorities according to the current version of German Law on the Protection of Animals. M.D. was supported by a grant from Deutscher Akademischer Austauschdienst (DAAD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalisch, R., Delfino, M., Murer, M.G. et al. The phenylephrine blood pressure clamp in pharmacologic magnetic resonance imaging: reduction of systemic confounds and improved detectability of drug-induced BOLD signal changes. Psychopharmacology 180, 774–780 (2005). https://doi.org/10.1007/s00213-005-2252-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-005-2252-0