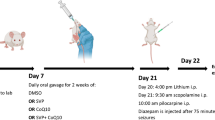
Abstract
Epilepsy is a condition marked by sudden, self-sustained, and recurring brain events, showcasing unique electro-clinical and neuropathological phenomena that can alter the structure and functioning of the brain, resulting in diverse manifestations. Antiepileptic drugs (AEDs) can be very effective in 30% of patients in controlling seizures. Several factors contribute to this: drug resistance, individual variability, side effects, complexity of epilepsy, incomplete understanding, comorbidities, drug interactions, and no adherence to treatment. Therefore, research into new AEDs is important for several reasons such as improved efficacy, reduced side effects, expanded treatment options, treatment for drug-resistant epilepsy, improved safety profiles, targeted therapies, and innovation and progress. Animal models serve as crucial biological tools for comprehending neuronal damage and aiding in the discovery of more effective new AEDs. The utilization of antioxidant agents that act on the central nervous system may serve as a supplementary approach in the secondary prevention of epilepsy, both in laboratory animals and potentially in humans. Chlorogenic acid (CGA) is a significant compound, widely prevalent in numerous medicinal and food plants, exhibiting an extensive spectrum of biological activities such as neuroprotection, antioxidant, anti-inflammatory, and analgesic effects, among others. In this research, we assessed the neuroprotective effects of commercially available CGA in Wistar rats submitted to lithium-pilocarpine-induced status epilepticus (SE) model. After 72-h induction of SE, rats received thiopental and were treated for three consecutive days (1st, 2nd, and 3rd doses). Next, brains were collected and studied histologically for viable cells in the hippocampus with staining for cresyl-violet (Nissl staining) and for degenerating cells with Fluoro-Jade C (FJC) staining. Moreover, to evaluate oxidative stress, the presence of malondialdehyde (MDA) and superoxide dismutase (SOD) was quantified. Rats administered with CGA (30 mg/kg) demonstrated a significant decrease of 59% in the number of hippocampal cell loss in the CA3, and of 48% in the hilus layers after SE. A significant reduction of 75% in the cell loss in the CA3, shown by FJC+ staining, was also observed with the administration of CGA (30 mg/kg). Furthermore, significant decreases of 49% in MDA production and 72% in the activity of SOD were seen, when compared to animals subjected to SE that received vehicle. This study introduces a novel finding: the administration of CGA at a dosage of 30 mg/kg effectively reduced oxidative stress induced by lithium-pilocarpine, with its effects lasting until the peak of neural damage 72 h following the onset of SE. Overall, the research and development of new AEDs are essential for advancing epilepsy treatment, improving patient outcomes, and ultimately enhancing the quality of life for individuals living with epilepsy.




Similar content being viewed by others
Data availability
The data that supports the findings of this study are available upon request.
Abbreviations
- 6-OHDA:
-
6-Hydroxydopamine
- ANOVA:
-
Analysis of variance
- CGA:
-
Chlorogenic acid
- CQA:
-
4,5-di-O-[E]-Caffeoylquinic acid
- FJC:
-
Fluoro-Jade C
- FJC+:
-
Fluoro-Jade C positive
- ER:
-
Endoplasmic reticulum
- Li-Pilo:
-
Lithium-pilocarpine
- MDA:
-
Malondialdehyde
- NO:
-
Nitric oxide
- ROS:
-
Reactive oxygen species
- SE:
-
Status epilepticus
- SOD:
-
Superoxide dismutase
- SRS:
-
Spontaneous recurrent seizures
References
Akkaya R, Akkaya B, Bello RO (2023) Molecular docking to investigate the inhibitory activity and the role of nitric oxide in anticonvulsant effects of vitamin D on pentylenetetrazole-induced epileptic seizures in rats. J Mol Struct. 1273:134246. https://doi.org/10.1016/j.molstruc.2022.134246
Alkis HE, Kuzhan A, Dirier A, Tarakcioglu M, Demir E, Saricicek E, Demir T, Ahlatci A, Demirci A, Cinar K, Taysi S (2015) Neuroprotective effects of propolis and caffeic acidphenethyl ester (CAPE) on the radiation-injured brain tissue (Neuroprotective effects of propolis and CAPE). Int J Rad Res, 13,(4): https://doi.org/10.7508/ijrr.2015.04.002
Andreyev AY, Kushnareva YE, Starkov AA (2005) Mitochondrial metabolism of reactive oxygen species. Biochemistry (Mosc) 70:200–214. https://doi.org/10.1007/s10541-005-0102-7
Barros DO, Xavier SML, Barbosa CO, Silva RF, Freitas RLM, Maia FD, Oliveira AA, Freitas RM, Takahashi RN (2007) Effects of the vitamin E in catalase activities in hippocampus after status epilepticus induced by pilocarpine in Wistar rats. Neurosci Lett 416:227–230. https://doi.org/10.1016/j.neulet.2007.01.057
Bymaster FP, McKinzie DL, Felder CC, Felder CC (2003) Use of M1–M5 muscarinic receptor knockout mice as novel tools to delineate the physiological roles of the muscarinic cholinergic system. Neurochem Res 28:437–442. https://doi.org/10.1023/a:1022844517200
Camkurt MA, Fındıklı E, Bakacak M, Tolun Fİ, Karaaslan MF (2017) Evaluation of malondialdehyde, superoxide dismutase and catalase activity in fetal cord blood of depressed mothers. Clin Psychopharmacol Neurosci 15:35–39. https://doi.org/10.9758/cpn.2017.15.1.35
Cao H, Zhang L, Qu Z, Tian S, Wang Z, Jiang Y, Hou Q, Jia L, Weiping Wang W (2021) The protective effect of hydroxylated fullerene pretreatment on pilocarpine-induced status epilepticus. Brain Res. 1764:147468. https://doi.org/10.1016/j.brainres.2021.147468
Chan L, Hong C-T, Bai C-H (2021) Coffee consumption and the risk of cerebrovascular disease: a meta-analysis of prospective cohort studies. BMC Neurol 21:380. https://doi.org/10.1186/s12883-021-02411-5
Chen Y, Sun H, Huang L, Li J, Zhou W, Chang J (2015) Neuroprotective effect of radix trichosanthis saponins on subarachnoid hemorrhage. Evid Based Complement Alternat Med 2015:313657. https://doi.org/10.1155/2015/313657
Colombo R, Papetti A (2020) An outlook on the role of decaffeinated coffee in neurodegenerative diseases. Crit Rev Food Sci Nutr 60:760–779. https://doi.org/10.1080/10408398.2018.1550384
Curia G, Longo D, Biagini G, Jones RSG, Avoli M (2008) The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods 172:143–157. https://doi.org/10.1016/j.jneumeth.2008.04.019
Deepa D, Jayakumari B, Thomas SV (2008) Lipid peroxidation in women with epilepsy. Ann Indian Acad Neurol 11:44–46. https://doi.org/10.4103/0972-2327.40225
dos Santos MD, Chen G, Almeida MC, Soares DM, de Souza GEP, Lopes NP, Lan R C (2010) Effects of caffeoylquinic acid derivatives and C-flavonoid from Lychnophora ericoides on in vitro inflammatory mediator production. Nat Prod Commun; 5: 733–740. In Internet: http://www.ncbi.nlm.nih.gov/pubmed/20521538
Foettinger A, Melmer M, Leitner A, Lindner W (2007) Reaction of the indole group with malondialdehyde: application for the derivatization of tryptophan residues in peptides. Bioconjug Chem 18:1678–1683. https://doi.org/10.1021/bc070001h
Fountain NB (2000) Status epilepticus: risk factors and complications. Epilepsia 41(Suppl 2):S23-30. https://doi.org/10.1111/j.1528-1157.2000.tb01521.x
Fujikawa DG (1996) The temporal evolution of neuronal damage from pilocarpine-induced status epilepticus. Brain Res 725:11–22. https://doi.org/10.1016/0006-8993(96)00203-x
Gao X, You Z, Huang C, Liu Z, Tan Z, Jiran Li J, Liu Y, Liu X, Wei F, Fan Z, Qi S, Sun J (2023) NCBP1 improves cognitive function in mice by reducing oxidative stress, neuronal loss, and glial activation after status epilepticus. Mol Neurobiol. 60(11):6676–6688. https://doi.org/10.1007/s12035-023-03497-3
Green JL, Dos Santos WF, Fontana ACK (2021) Role of glutamate excitotoxicity and glutamate transporter EAAT2 in epilepsy: opportunities for novel therapeutics development. Biochem Pharmacol 193:114786. https://doi.org/10.1016/j.bcp.2021.114786
Hamilton SE, Loose MD, Qi M, Levey AI, Hille B, McKnight GS, Idzerda RL, Nathanson NM (1997) Disruption of the M1 receptor gene a blates muscarinic receptor-dependent M current regulation and seizure activity in mice. Proc Natl Acad Sci U S A 94:13311–13316. https://doi.org/10.1073/pnas.94.24.13311
Keles LC, de Melo NI, de Aguiar GP, Wakabayashi KAL, de Carvalho CE, Cunha WR, Crotti AEM (2010) Lychnophorinae (asteraceae): a survey of its chemical constituents and biological activities. Quim Nova 33:2245–2260. https://doi.org/10.1590/S0100-40422010001000038
Kwak SE, Kim JE, Kim DS, Jung JY, Won MH, Kwon OS, Choi SY, Kang TC (2005) Effects of GABAergic transmissions on the immunoreactivities of calcium binding proteins in the gerbil hippocampus. J Comp Neurol 485:153–164. https://doi.org/10.1002/cne.20482
Liang N, Kitts DD (2015) Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients; 8. https://doi.org/10.3390/nu8010016
Liao K-H, Mei Q-Y, Zhou Y-C (2004) Determination of antioxidants in plasma and erythrocyte in patients with epilepsy. Zhong Nan Da Xue Xue Bao Yi Xue Ban. Journal of Central South University (Medical Sciences) 29:72–74. In Internet: http://www.ncbi.nlm.nih.gov/pubmed/16137011
Liberato JL, Rosa MN, Miranda MCR, Lopes JLC, Lopes NP, Gobbo-Neto L, Fontana ACK, dos Santos WF (2023) Neuroprotective properties of chlorogenic acid and 4,5-caffeoylquinic acid from Brazilian arnica (Lychnophora ericoides) after acute retinal ischemia. Planta Med 89:183–193. https://doi.org/10.1055/a-1903-2387
Liu J, Wang A, Li L, Huang Y, Xue P, Hao A (2010) Oxidative stress mediates hippocampal neuron death in rats after lithium-pilocarpine-induced status epilepticus. Seizure 19:165–172. https://doi.org/10.1016/j.seizure.2010.01.010
Liu D, Wang H, Zhang Y, Zhang Z (2020) Protective effects of chlorogenic acid on cerebral ischemia/reperfusion injury rats by regulating oxidative stress-related Nrf2 pathway. Drug Des Devel Ther. 14:51–60. https://doi.org/10.2147/DDDT.S228751
Lu H, Tian Z, Cui Y, Liu Z, Ma X (2020) Chlorogenic acid: a comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanisms of action, and future directions. Compr Rev Food Sci Food Saf. 19(6):3130–3158. https://doi.org/10.1111/1541-4337.12620
Manouze H, Ghestem A, Bennis M, Sokar Z, Benoliel J-J, Becker C, Ba-M’hamed S, Bernard C (2021) Antiseizure effects of Anacyclus pyrethrum in socially isolated rats with and without a positive handling strategy. Epilepsia. 62(10):2551–2564. https://doi.org/10.1111/epi.17028
Matzen J, Buchheim K, van Landeghem FKH, Meierkord H, Holtkamp M (2008) Functional and morphological changes in the dentate gyrus after experimental status epilepticus. Seizure 17:76–83. https://doi.org/10.1016/j.seizure.2007.07.008
Miller RJ (1991) The control of neuronal Ca2+ homeostasis. Prog Neurobiol 37:255–285. https://doi.org/10.1016/0301-0082(91)90028-y
Moscone D, Mascini M (1988) Determination of superoxide dismutase activity with an electrochemical oxygen probe. Anal Chim Acta 211:195–204. https://doi.org/10.1016/S0003-2670(00)83679-X
Müller CJ, Bankstahl M, Gröticke I, Löscher W (2009) Pilocarpine vs. lithium-pilocarpine for induction of status epilepticus in mice: development of spontaneous seizures, behavioral alterations and neuronal damage. Eur J Pharmacol 619:15–24. https://doi.org/10.1016/j.ejphar.2009.07.020
Naveed M, Hejazi V, Abbas M, Kamboh AA, Khan GJ, Shumzaid M, Ahmad F, Babazadeh D, Fang Fang X, Modarresi-Ghazani F, Wen Hua L, Xiao Hui Z (2018) Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed Pharmacother 97:67–74. https://doi.org/10.1016/j.biopha.2017.10.064
Pai M-S, Wang K-C, Yeh K-C, Wang S-J (2023) Stabilization of mitochondrial function by chlorogenic acid protects against kainic acid-induced seizures and neuronal cell death in rats. Europ J of Pharmacol. 961:176197. https://doi.org/10.1016/j.ejphar.2023.176197
Paxinos G, Watson C (2006) The rat brain in stereotaxic coordinates. 6th Editio. Academic Press; eBook ISBN: 9780080475158
Pazdernik TL, Emerson MR, Cross R, Nelson SR, Samson FE (2001) Soman-induced seizures: limbic activity, oxidative stress and neuroprotective proteins. J Appl Toxicol 21(Suppl 1):S87-94. https://doi.org/10.1002/jat.818
Schmued LC, Hopkins KJ (2000) Fluoro-Jade: novel fluorochromes for detecting toxicant-induced neuronal degeneration. Toxicol Pathol 28:91–99. https://doi.org/10.1177/019262330002800111
Scorza FA, Arida RM, Naffah-Mazzacoratti MG, Scerni DA, Calderazzo L, Cavalheiro EA (2009) The pilocarpine model of epilepsy: what have we learned? An Acad Bras Cienc. 81(3):345–65. https://doi.org/10.1590/s0001-37652009000300003
Seclen S S, Baracco M R, Mohanna B S (2006) Antioxidantes en poblaciones adultas del nivel del mar y de grandes alturas: Actividad dela superóxido dismutasa. In: Rev Med Hered [online]. Lima, Perú. In Internet: Rev Med Hered v.17 n.1 Lima ene./mar. http://www.scielo.org.pe/scielo.php?script=sci_arttext&pid=S1018130X2006000100002&lng=es&nrm=iso
Shan S, Tian L, Fang R (2019) Chlorogenic acid exerts beneficial effects in 6-hydroxydopamine-induced neurotoxicity by inhibition of endoplasmic reticulum stress. Med Sci Monit 25:453–459. https://doi.org/10.12659/MSM.911166
Shishmanova-Doseva M, Peychev L, Yoanidu L, Uzunova Y, Milena Atanasova M, Georgieva K, Tchekalarova J (2021) Anticonvulsant effects of topiramate and lacosamide on pilocarpine-induced status epilepticus in rats: a role of reactive oxygen species and inflammation. Int J Mol Sci 22(5):2264. https://doi.org/10.3390/ijms22052264
Socała K, Szopa A, Serefko A, Poleszak E, Wlaź P (2020) Neuroprotective effects of coffee bioactive compounds: a review. Int J Mol Sci 22:107. https://doi.org/10.3390/ijms22010107
Socodato R, Portugal CC, Canedo T, Domith I, Oliveira NA, Paes-de-Carvalho R, Relvas JB, Cossenza M (2015) c-Src deactivation by the polyphenol 3-O-caffeoylquinic acid abrogates reactive oxygen species-mediated glutamate release from microglia and neuronal excitotoxicity. Free Radic Biol Med 79:45–55. https://doi.org/10.1016/j.freeradbiomed.2014.11.019
Sudha K, Rao AV, Rao A (2001) Oxidative stress and antioxidants in epilepsy. Clin Chim Acta 303:19–24. https://doi.org/10.1016/s0009-8981(00)00337-5
Tajik N, Tajik M, Mack I, Enck P (2017) The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: a comprehensive review of the literature. Eur J Nutr 56:2215–2244. https://doi.org/10.1007/s00394-017-1379-1
Tommerdahl KL, Hu EA, Selvin E, Steffen LM, Coresh J, Grams ME, Bjornstad P, Rebholz CM, Parikh CR (2022) Coffee consumption may mitigate the risk for acute kidney injury: results from the atherosclerosis risk in communities study. Kidney Int Rep 7:1665–1672. https://doi.org/10.1016/j.ekir.2022.04.091
Turski WA, Cavalheiro EA, Schwarz M, Czuczwar SJ, Kleinrok Z, Turski L (1983) Limbic seizures produced by pilocarpine in rats: behavioural, electroencephalographic and neuropathological study. Behav Brain Res 9:315–335. https://doi.org/10.1016/0166-4328(83)90136-5
Wang L, Liu Y-HH, Huang Y-GG, Chen L-WW (2008) Time-course of neuronal death in the mouse pilocarpine model of chronic epilepsy using fluoro-jade C staining. Brain Res 1241(November):157–67. https://doi.org/10.1016/j.brainres.2008.07.097
Wasim S, Kukkar V, Awad VM, Sakhamuru S, Malik BH (2020) Neuroprotective and neurodegenerative aspects of coffee and its active ingredients in view of scientific literature. Cureus 12:e9578. https://doi.org/10.7759/cureus.9578
Xi Y, Li H, Yu M, Li X, Li Y, Hui B, Zeng X, Wang J, Li J (2022) Protective effects of chlorogenic acid on trimethyltin chloride-induced neurobehavioral dysfunctions in mice relying on the gut microbiota. Food Funct. 13(3):1535–1550. https://doi.org/10.1039/d1fo03334d
Xi Y, Li W, Wang J, Yu M, Zeng X, Li H, Li J (2024) Cyanidin-3-O-glucoside alleviates trimethyltin chloride-induced neurodegeneration by maintaining glutamate homeostasis through modulation of the gut microbiota. Food Sci Human Wellness. 3(2):1093–1107. https://doi.org/10.26599/FSHW.2022.9250141
Zhang C, Xu Y, Tan HY, Li S, Wang N, Zhang Y, Feng Y (2018) Neuroprotective effect of He-Ying-Qing-Re formula on retinal ganglion cell in diabetic retinopathy. J Ethnopharmacol 214:179–189. https://doi.org/10.1016/j.jep.2017.12.018
Zhang H, Lian Y, Xie N, Cheng X, Chen C, Xu H, Zheng Y (2020) Antagomirs targeting miR-142-5p attenuate pilocarpine-induced status epilepticus in mice. Exp Cell Res. 393(2):112089. https://doi.org/10.1016/j.yexcr.2020.112089
Acknowledgements
We would like to acknowledge Dr. Javier Marín-Prida (Center for Research and Biological Evaluations (CEIEB), Institute of Pharmacy and Food, University of Havana Havana, Cuba) for his help with the oxidative stress protocols.
Funding
This work was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES–PROEX) to AJCG and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), both from Brazil.
Author information
Authors and Affiliations
Contributions
AJC-G wrote, and edited the first step of the manuscript, also contributed to data analysis tools; these data were part of his master’s thesis. JLL wrote, reviewed, and edited the manuscript, also contributed to data analysis tools. MVC contributed by doing the experiments and to data analysis tools. NPL conceived and designed the analysis, wrote the paper, and acquired funding. JLCL conceived and designed the analysis, and wrote the manuscript. LGN collected the data, and performed the analysis. ACKF conceived and designed the analysis, and wrote, reviewed, and edited the manuscript. WFS conceived and designed the analysis; wrote, reviewed, and edited the manuscript; and acquired funding. All data were generated in-house, and no paper mill was used. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Ethics approval
The experimental protocol was in accordance with and approved by the local Animal Care Committee (protocol number 08.1.834.53.9 CEUA–Campus University of São Paulo, Ribeirão Preto, Brazil).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carreño-González, A.J., Liberato, J.L., Celani, M.V.B. et al. Neuroprotective effects of chlorogenic acid against oxidative stress in rats subjected to lithium-pilocarpine-induced status epilepticus. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-03080-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-03080-0