Abstract
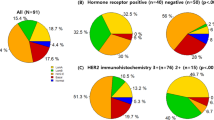
Recent clinical evidence shows that the antibody-drug conjugate (ADC) trastuzumab deruxtecan (T-DXd) can successfully treat patients with advanced HER2-mutant non-small cell lung cancer (NSCLC). We aimed to characterize HER2 mutations in cervical neuroendocrine carcinoma (NEC) among Taiwanese women to provide the rationale for exploring T-DXd as a tumor-agnostic targeted therapy option. We analyzed 12 archived primary cervical NEC samples from Taiwanese patients. Tumor-rich areas were marked for microdissection on 10 μm unstained sections. DNA was extracted, and HER2 hotspots were sequenced using a targeted panel on the Illumina MiSeq. HER2 missense mutations were identified in 5 of 12 cases (41.7%). Of the 5 cases with mutations, 2 patients (40%) had a single mutation, while 3 patients (60%) had double mutations. We detected 4 substitutions outside the tyrosine kinase domain (non-TKD), which were p.P1170A, p.S305C, p.I655V, and a novel T328K alteration. No mutations were found within the tyrosine kinase domain (TKD). The 41.7% HER2 mutation rate warrants expanded screening and future clinical investigation of the T-DXd targeting HER2 mutations in cervical NEC patients. Overall, this study contributes to the molecular understanding of cervical NEC and lays the groundwork for developing more effective treatment strategies.



Similar content being viewed by others
Data availability
All raw data generated and analyzed during this study are available in Table 1.
References
Adzhubei I, Jordan DM, Sunyaev SR (2013) Predicting functional effect of human missense mutations using PolyPhen-2. Curr Protoc Hum Genet. Chapter 7(Unit7):20. https://doi.org/10.1002/0471142905.hg0720s76
Chao A, Wu RC, Lin CY, Chang TC, Lai CH (2023) Small cell neuroendocrine carcinoma of the cervix: from molecular basis to therapeutic advances. Biomed J 46(5):100633. https://doi.org/10.1016/j.bj.2023.100633
Cho SY, Choi M, Ban HJ, Lee CH, Park S, Kim H, et al. (2017) Cervical small cell neuroendocrine tumor mutation profiles via whole exome sequencing. Oncotarget. 31;8(5):8095-8104. https://doi.org/10.18632/oncotarget.14098
Connell CM, Doherty GJ (2017) Activating HER2 mutations as emerging targets in multiple solid cancers. ESMO Open. 24;2(5):e000279. https://doi.org/10.1136/esmoopen-2017-000279
FDA-01 (2022), FDA gives nod to T-DXd for HER2-mutant NSCLC. Cancer Discov. 2022;12(10):2224. https://doi.org/10.1158/2159-8290.CD-NB2022-0053
FDA-02 (2022), FDA grants regular approval to fam-trastuzumab deruxtecan-nxki for breast cancer, https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-regular-approval-fam-trastuzumab-deruxtecan-nxki-breast-cancer. Accessed on Mar 18, 2024.
FDA-03 (2022), FDA approves fam-trastuzumab deruxtecan-nxki for HER2-positive gastric adenocarcinomas https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-fam-trastuzumab-deruxtecan-nxki-her2-positive-gastric-adenocarcinomas. Accessed on Mar 18, 2024.
FDA-04 (2022), FDA approves first targeted therapy for HER2-low breast cancer; https://www.fda.gov/news-events/press-announcements/fda-approves-first-targeted-therapy-her2-low-breast-cancer. Accessed on Mar 18, 2024.
Frumovitz M, Burzawa JK, Byers LA, Lyons YA, Ramalingam P, Coleman RL, et al. (2016) Brown J. Sequencing of mutational hotspots in cancer-related genes in small cell neuroendocrine cervical cancer. Gynecol Oncol. 141(3):588-591. https://doi.org/10.1016/j.ygyno.2016.04.001. Epub 2016 Apr 15. Erratum in: Gynecol Oncol. 2016 Oct;143(1):224
Goto K, Goto Y, Kubo T, Ninomiya K, Kim SW, Planchard D, Ahn MJ, Smit EF, et al. (2023) Trastuzumab deruxtecan in patients with HER2-mutant metastatic non-small-cell lung cancer: primary results from the randomized, phase II DESTINY-Lung02 trial. J Clin Oncol. 1;41(31):4852-4863. https://doi.org/10.1200/JCO.23.01361. Epub 2023 Sep 11, and Table A1 in the supplementary appendix for trastuzumab deruxtecan in patients with HER2-mutant metastatic non–small cell lung cancer: primary results from the randomized, phase 2 DESTINY-Lung02 trial—ONLINE ONLY. https://ascopubs.org/doi/suppl/10.1200/JCO.23.01361/suppl_file/DS_JCO.23.01361.pdf
Han Y, Xiong Y, Lu T, Chen R, Liu Y, Tang, et al (2023) Genomic landscape and efficacy of HER2-targeted therapy in patients with HER2-mutant non-small cell lung cancer. Front Oncol 3(13):1121708. https://doi.org/10.3389/fonc.2023.1121708
Li BT, Michelini F, Misale S, Cocco E, Baldino L, Cai Y, et al. (2020) HER2-mediated internalization of cytotoxic agents in ERBB2 amplified or mutant lung cancers. Cancer Discov. 10(5):674-687. https://doi.org/10.1158/2159-8290.CD-20-0215
Li BT, Smit EF, Goto Y, Nakagawa K, Udagawa H, Mazières J, Nagasaka M, et al. (2022) DESTINY-Lung01 trial investigators. Trastuzumab deruxtecan in HER2-mutant non-small-cell lung cancer. N Engl J Med. 20;386(3):241-251. https://doi.org/10.1056/NEJMoa2112431. Epub 2021 Sep 18. PMID: 34534430; PMCID: PMC9066448, and Table S1 in the Supplementary Appendix, https://www.nejm.org/doi/suppl/10.1056/NEJMoa2112431/suppl_file/nejmoa2112431_appendix.pdf
Loeffler E, Ancel J, Dalstein V, Deslée G, Polette M, Nawrocki-Raby B (2023) HER2 alterations in non-small cell lung cancer: biologico-clinical consequences and interest in therapeutic strategies. Life (Basel) 29;14(1):64. https://doi.org/10.3390/life14010064
Lu J, Li Y, Wang J (2022) Small cell (neuroendocrine) carcinoma of the cervix: an analysis for 19 cases and literature review. Front Cell Infect Microbiol. 13(12):916506. https://doi.org/10.3389/fcimb.2022.916506
Mishra R, Hanker AB, Garrett JT (2017) Genomic alterations of ERBB receptors in cancer: clinical implications. Oncotarget. 8(69):114371-114392. https://doi.org/10.18632/oncotarget.22825
Nakagawa K, Nagasaka M, Feli E, Pacheco J, Baik C, Goto Y et al (2021) OA04.05 trastuzumab deruxtecan in HER2-overexpressing metastatic non-small cell lung cancer: interim results of DESTINY-Lung01. J Thorac Oncol 16:S109–S110. https://doi.org/10.1016/j.jtho.2021.01.285
Ren X, Wu W, Li Q, Li W, Wang G (2023) Advances in research, diagnosis, and treatment of neuroendocrine cervical carcinoma: a review. Oncol Rev 17:11764. https://doi.org/10.3389/or.2023.11764
Rubin E, Shan KS, Dalal S, Vu DUD, Milillo-Naraine AM, Guaqueta D, et al. (2024) Molecular targeting of the human epidermal growth factor receptor-2 (HER2) genes across various cancers. Int J Mol Sci 15;25(2):1064. https://doi.org/10.3390/ijms25021064
Salvo G, Flores Legarreta A, Ramalingam P, Jhingran A, Bhosale P, Saab R, et al. (2023) Clinicopathologic characteristics, oncologic outcomes, and prognostic factors in neuroendocrine cervical carcinoma: a Neuroendocrine Cervical Tumor Registry study. Int J Gynecol Cancer 4;33(9):1359-1369. https://doi.org/10.1136/ijgc-2023-004708
Sankyo D (2023) Enhertu approved in the EU as the first HER2 directed therapy for patients with HER2 mutant advanced non-small cell lung cancer. News release. Oct 23, 2023. https://www.daiichisankyo.com/files/news/pressrelease/pdf/202310/20231023_E.pdf. Accessed on Mar 11, 2024.
Schultheis AM, de Bruijn I, Selenica P, Macedo GS, da Silva EM, Piscuoglio S, et al. (2022). Genomic characterization of small cell carcinomas of the uterine cervix. Mol Oncol. 16(4):833-845. https://doi.org/10.1002/1878-0261.12962
Seymour C. Zongertinib (2023) Demonstrates clinical activity in HER2-mutant solid tumors, 2023, Sept. 12, https://www.targetedonc.com/view/zongertinib-demonstrates-clinical-activity-in-her2-mutant-solid-tumors. Accessed on Mar 18, 2024.
Tangella AV, Yadlapalli DC (2023) Neuroendocrine carcinoma of cervix: a case series. Cureus 17;15(5):e39165. https://doi.org/10.7759/cureus.39165
Tempfer CB, Tischoff I, Dogan A, Hilal Z, Schultheis B, Kern P et al (2018) Neuroendocrine carcinoma of the cervix: a systematic review of the literature. BMC Cancer 18(1):530. https://doi.org/10.1186/s12885-018-4447-x
Tsai WC, Chen CK, Han CP, Chao WR (2019) Molecular profiling of c-KIT oncogene in high-grade neuroendocrine carcinoma of the uterine cervix: analysis of twelve cases. Taiwan J Obstet Gynecol 58(4):581–582. https://doi.org/10.1016/j.tjog.2019.05.029
Tsurutani J, Iwata H, Krop I, Jänne PA, Doi T, Takahashi S, et al. (2020) Targeting HER2 with trastuzumab deruxtecan: a dose-expansion, phase I study in multiple advanced solid tumors. Cancer Discov. 10(5):688-701. https://doi.org/10.1158/2159-8290.CD-19-1014. Epub 2020 Mar 25. Erratum in: Cancer Discov. 2020 Jul;10(7):1078.
Vathiotis IA, Bafaloukos D, Syrigos KN, Samonis G (2023) Evolving treatment landscape of HER2-mutant non-small cell lung cancer: trastuzumab deruxtecan and beyond. Cancers (Basel). 17;15(4):1286. https://doi.org/10.3390/cancers15041286
Verma S, Dubey H, Gupta S, Ranjan A, Goel H, Sharma A (2023) Neuroendocrine carcinoma of cervix and review literature. Int J Surg Case Rep. 105:107982. https://doi.org/10.1016/j.ijscr.2023.107982
Wang H, Miao J, Wen Y, Xia X, Chen Y, Huang M, Chen S et al (2022) Molecular landscape of ERBB2 alterations in 14,956 solid tumors. Pathol Oncol Res 13(28):1610360. https://doi.org/10.3389/pore.2022.1610360
Wei XW, Gao X, Zhang XC, Yang JJ, Chen ZH, Wu YL, Zhou Q (2020) Mutational landscape and characteristics of ERBB2 in non-small cell lung cancer. Thorac Cancer 11(6):1512–1521. https://doi.org/10.1111/1759-7714.13419
Wen W, Chen WS, Xiao N, Bender R, Ghazalpour A, Tan Z et al (2015) Mutations in the kinase domain of the HER2/ERBB2 gene identified in a wide variety of human cancers. J Mol Diagn 17(5):487–95. https://doi.org/10.1016/j.jmoldx.2015.04.003
Winer I, Kim C, Gehrig P (2021) Neuroendocrine tumors of the gynecologic tract update. Gynecol Oncol 162(1):210–219. https://doi.org/10.1016/j.ygyno.2021.04.039
Xiang L, Jiang W, Ye S, He T, Pei X, Li J et al (2018) ERBB2 mutation: a promising target in non-squamous cervical cancer. Gynecol Oncol 148(2):311–316. https://doi.org/10.1016/j.ygyno.2017.12.023
Xiang L, Li J, Jiang W, Shen X, Yang W, Wu X, Yang H (2015) Comprehensive analysis of targetable oncogenic mutations in Chinese cervical cancers. Oncotarget. 10;6(7):4968-75. https://doi.org/10.18632/oncotarget.3212
Xing D, Zheng G, Schoolmeester JK, Li Z, Pallavajjala A, Haley L et al (2018) Next-generation sequencing reveals recurrent somatic mutations in small cell neuroendocrine carcinoma of the uterine cervix. Am J Surg Pathol. 42(6):750–760. https://doi.org/10.1097/PAS.0000000000001042
Funding
This work was supported by grants CSH-2017-C-034 and CSH-2018-C-028 from Chung Shan Medical University Hospital in Taichung, Taiwan.
Author information
Authors and Affiliations
Contributions
Conception and design: GTS, HPS, CPH; administrative support: YJL; provision of study materials or patients: YJL, WRC; collection and assembly of data: WRC, MYL; manuscript writing: WRC, HPS, CPH; final approval of manuscript: all authors. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Declarations
Ethical standards
This research was conducted according to the International Conference on Harmonization (ICH) guidelines and complied with all applicable regulations for the protection of human research subjects, including review and approval by the Institutional Review Board at the Chung-Shan Medical University Hospital in Taichung, Taiwan.
Consent to participate
The tissue samples used were de-identified, so consent to participate declarations were not applicable.
Competing interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chao, WR., Lee, MY., Sheu, GT. et al. HER2 mutations in advanced cervical neuroendocrine carcinoma: implications for trastuzumab deruxtecan therapy. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-03066-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-03066-y