Abstract
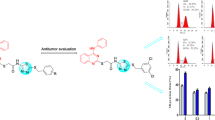
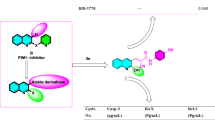
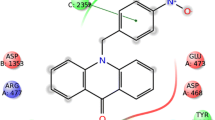
In this study, the anticancer activities of some pyrrolopyrimidine derivatives were evaluated. Compound 3 is the most cytotoxic compound on MCF-7 cancer cells with an IC50 value of 23.42 µM. Also, compound 3 induced apoptosis and the ROS(+) cell population in MCF-7 cells. Moreover, it significantly reduced MMP-9 activity, having 42.16 ± 5.10% and 58.28 ± 1.96% inhibitory activities at 10 µM and 50 µM concentrations, respectively. Molecular docking results supported the activity, showing key hydrogen bonds with the binding site of MMP-9. Therefore, compound 3 might be a lead compound for the development of potent MMP-9 inhibitors.








Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Amawi H, Karthikeyan C, Pathak R, Hussein N, Christman R, Robey R et al (2017) Thienopyrimidine derivatives exert their anticancer efficacy via apoptosis induction, oxidative stress and mitotic catastrophe. Eur J Med Chem 138:1053–1065. https://doi.org/10.1016/j.ejmech.2017.07.028
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Azimi I, Petersen RM, Thompson EW, Roberts-Thomson SJ, Monteith GR (2017) Hypoxia-induced reactive oxygen species mediate N-cadherin and SERPINE1 expression, EGFR signalling and motility in MDA-MB-468 breast cancer cells. Sci Rep 7:15140. https://doi.org/10.1038/s41598-017-15474-7
Brieger K, Schiavone S, Miller FJ Jr, Krause KH (2012) Reactive oxygen species: from health to disease. Swiss Med Wkly 142:w13659. https://doi.org/10.4414/smw.2012.13659
Chen Y, Azad MB, Gibson SB (2009) Superoxide is the major reactive oxygen species regulating autophagy. Cell Death Differ 16:1040–1052. https://doi.org/10.1038/cdd.2009.49
Church DF, Pryor WA (1985) Free-radical chemistry of cigarette smoke and its toxicological implications. Environ Health Perspect 64:111–126. https://doi.org/10.1289/ehp.8564111
Conklin KA (2004) Chemotherapy-associated oxidative stress: impact on chemotherapeutic effectiveness. Integr Cancer Ther 3:294–300. https://doi.org/10.1177/1534735404270335
El Ashry ESH, Awad LF, Teleb M, Ibrahim NA, Abu-Serie MM, Abd Al Moaty MN (2020) Structure-based design and optimization of pyrimidine- and 1,2,4-triazolo[4,3-a]pyrimidine-based matrix metalloproteinase-10/13 inhibitors via Dimroth rearrangement towards targeted polypharmacology. Bioorg Chem 96:103616. https://doi.org/10.1016/j.bioorg.2020.103616
Engel CK, Pirard B, Schimanski S, Kirsch R, Habermann J, Klingler O et al (2005) Structural basis for the highly selective inhibition of MMP-13. Chem Biol 12:181–189. https://doi.org/10.1016/j.chembiol.2004.11.014
Fischer T, Senn N, Riedl R (2019) Design and structural evolution of matrix metalloproteinase inhibitors. Chemistry 25:7960–7980. https://doi.org/10.1002/chem.201805361
Gencer S, Cebeci A, Irmak-Yazicioglu MB (2013) Matrix metalloproteinase gene expressions might be oxidative stress targets in gastric cancer cell lines. Chin J Cancer Res 25:322–333. https://doi.org/10.3978/j.issn.1000-9604.2013.06.05
Gimeno A, Beltrán-Debón R, Mulero M, Pujadas G, Garcia-Vallvé S (2020) Understanding the variability of the S1’ pocket to improve matrix metalloproteinase inhibitor selectivity profiles. Drug Discov Today 25:38–57. https://doi.org/10.1016/j.drudis.2019.07.013
Huang H (2018) Matrix metalloproteinase-9 (MMP-9) as a cancer biomarker and mMP-9 biosensors: recent advances. Sensors 18:3249. https://doi.org/10.3390/s18103249
Jabłońska-Trypuć A, Matejczyk M, Rosochacki S (2016) Matrix metalloproteinases (MMPs), the main extracellular matrix (ECM) enzymes in collagen degradation, as a target for anticancer drugs. J Enzyme Inhib Med Chem 31:177–183. https://doi.org/10.3109/14756366.2016.1161620
Kilic-Kurt Z, Aka Y, Kutuk O (2020) Novel pyrrolopyrimidine derivatives induce p53-independent apoptosis via the mitochondrial pathway in colon cancer cells. Chem Biol Interact 330:109236. https://doi.org/10.1016/j.cbi.2020.109236
Kilic-Kurt Z, Bakar-Ates F, Aka Y, Kutuk O (2019) Design, synthesis and in vitro apoptotic mechanism of novel pyrrolopyrimidine derivatives. Bioorg Chem 83:511–519. https://doi.org/10.1016/j.bioorg.2018.10.060
Kilic-Kurt Z, Bakar-Ates F, Karakas B, Kütük Ö (2018) Cytotoxic and apoptotic effects of novel pyrrolo[2,3-d]pyrimidine derivatives containing urea moieties on cancer cell lines. Anticancer Agents Med Chem 18:1303–1312. https://doi.org/10.2174/1871520618666180605082026
Li N, Ragheb K, Lawler G, Sturgis J, Rajwa B, Melendez JA et al (2003) Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J Biol Chem 278:8516–8525. https://doi.org/10.1074/jbc.M210432200
Lin W, Shen P, Song Y, Huang Y, Tu S (2021) Reactive oxygen species in autoimmune cells: function, differentiation, and metabolism. Front Immunol 12. https://doi.org/10.3389/fimmu.2021.635021
Marullo R, Werner E, Degtyareva N, Moore B, Altavilla G, Ramalingam SS et al (2013) Cisplatin induces a mitochondrial-ROS response that contributes to cytotoxicity depending on mitochondrial redox status and bioenergetic functions. PLoS One 8:e81162. https://doi.org/10.1371/journal.pone.0081162
Mondal S, Adhikari N, Banerjee S, Amin SA, Jha T (2020) Matrix metalloproteinase-9 (MMP-9) and its inhibitors in cancer: a minireview. Eur J Med Chem 194:112260. https://doi.org/10.1016/j.ejmech.2020.112260
Mouret S, Baudouin C, Charveron M, Favier A, Cadet J, Douki T (2006) Cyclobutane pyrimidine dimers are predominant DNA lesions in whole human skin exposed to UVA radiation. Proc Natl Acad Sci U S A 103:13765–13770. https://doi.org/10.1073/pnas.0604213103
Nara H, Sato K, Naito T, Mototani H, Oki H, Yamamoto Y et al (2014) Thieno[2,3-d]pyrimidine-2-carboxamides bearing a carboxybenzene group at 5-position: highly potent, selective, and orally available MMP-13 inhibitors interacting with the S1″ binding site. Bioorg Med Chem 22:5487–5505. https://doi.org/10.1016/j.bmc.2014.07.025
Nicolotti O, Catto M, Giangreco I, Barletta M, Leonetti F, Stefanachi A et al (2012) Design, synthesis and biological evaluation of 5-hydroxy, 5-substituted-pyrimidine-2,4,6-triones as potent inhibitors of gelatinases MMP-2 and MMP-9. Eur J Med Chem 58:368–376. https://doi.org/10.1016/j.ejmech.2012.09.036
Rowsell S, Hawtin P, Minshull CA, Jepson H, Brockbank SM, Barratt DG et al (2002) Crystal structure of human MMP9 in complex with a reverse hydroxamate inhibitor. J Mol Biol 319:173–181. https://doi.org/10.1016/S0022-2836(02)00262-0
Schumacker PT (2015) Reactive oxygen species in cancer: a dance with the devil. Cancer Cell 27:156–157. https://doi.org/10.1016/j.ccell.2015.01.007
Snezhkina AV, Kudryavtseva AV, Kardymon OL, Savvateeva MV, Melnikova NV, Krasnov GS et al (2019) ROS generation and antioxidant defense systems in normal and malignant cells. Oxid Med Cell Longev 2019:6175804. https://doi.org/10.1155/2019/6175804
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31:455–461. https://doi.org/10.1002/jcc.21334
Wang FC, Peng B, Cao SL, Li HY, Yuan XL, Zhang TT et al. (2020) Synthesis and cytotoxic activity of chalcone analogues containing a thieno[2,3-d]pyrimidin-2-yl group as the A-ring or B-ring. Bioorg Chem 94. https://doi.org/10.1016/j.bioorg.2019.103346
Winter E, Pizzol CD, Filippin-Monteiro FB, Brondani P, Silva AMPW, Silva AH et al (2014) Antitumoral activity of a trichloromethyl pyrimidine analogue: molecular cross-talk between intrinsic and extrinsic apoptosis. Chem Res Toxicol 27:1040–1049. https://doi.org/10.1021/tx500094x
Yang H, Villani RM, Wang H, Simpson MJ, Roberts MS, Tang M et al (2018) The role of cellular reactive oxygen species in cancer chemotherapy. J Exp Clin Cancer Res 37:266. https://doi.org/10.1186/s13046-018-0909-x
Funding
This work was supported by Ankara University Scientific Research Council of Ankara University with 18H0237006 project number.
Author information
Authors and Affiliations
Contributions
ZKK and FBA conceived and the designed research. ZKK, FBA, and AC conducted experiments. FBA and AC analyzed data. ZKK and FBA wrote the manuscript. All authors read and approved the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kilic-Kurt, Z., Celik, A. & Bakar-Ates, F. Effects of pyrrolopyrimidine derivatives on cancer cells cultured in vitro and potential mechanism. Naunyn-Schmiedeberg's Arch Pharmacol 397, 3169–3177 (2024). https://doi.org/10.1007/s00210-023-02799-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02799-6