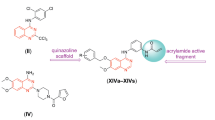
Abstract
In order to search for new antitumor drugs with high efficiency and low toxicity, a series of novel 4-aminoquinazoline derivatives containing 1,3,4-thiadiazole group were designed, synthesized and evaluated for antiproliferative activity against four human cancer cell lines (H1975, PC-3, MCF-7 and HGC-27) using MTT assay in vitro. Among them, compound N-(5-((3,5-dichlorobenzyl)thio)-1,3,4-thiadiazol-2-yl)-2-((4-(phenylamino)quinazolin-2-yl)thio)acetamide (15o) showed good anti-tumor proliferation activity against four tested cancer cell lines, with IC50 values of 1.96 ± 0.15 μM to PC-3 cell. The antitumor activity was significantly better than that of gefitinib. Further mechanism studies showed that compound 15o inhibited the migration ability of PC-3 tumor cells in a concentration dependent and time-dependent manner, and blocked the cell cycle in S phase. At the same time, cell cloning experiment further proved that compound 15o significantly inhibited the colony formation of PC-3 cells, The inhibition rate was as high as 92% at 2.0 μM.

Graphical abstract





Similar content being viewed by others
References
Bixel K, Vetter M, Davidson B, Berchuck A, Cohn D, Copeland L, et al. Intraperitoneal chemotherapy following neoadjuvant chemotherapy and optimal interval tumor reductive surgery for advanced ovarian cancer. Gynecol Oncol. 2020;156:530–4. https://doi.org/10.1016/j.ygyno.2019.12.016.
Bonet M, Garcia V, Farre N, Algara M, Farrús B, Fernandez J, et al. Radiation therapy for bone-only metastases in breast cancer patients: a Goco survey of current clinical practice. Rep. Pr Oncol Radiother. 2020;25:113–6. https://doi.org/10.1016/j.rpor.2019.12.019.
Fu RG, Sun Y, Sheng WB, Liao DF. Designing multi-targeted agents: an emerging anticancer drug discovery paradigm. Eur J Med Chem. 2017;136:195–211. https://doi.org/10.1016/j.ejmech.2017.05.016.
Hagemans J, Van Rees JM, Alberda WJ, Rothbarth J, Nuyttens JJME, van Meerten E, et al. Locally recurrent rectal cancer: long-term outcome of curative surgical and non-surgical treatment of 447 consecutive patients in a tertiary referral centre. Eur J Surg Oncol. 2020;46:448–54. https://doi.org/10.1016/j.ejso.2019.10.037.
Rettenmaier MA, Micha JP, Bohart R, Goldstein BH. A retrospective study comparing the efficacy of dose-dense chemotherapy, intraperitoneal chemotherapy and dose-dense chemotherapy with hyperthermic intraperitoneal chemotherapy in the treatment of advanced stage ovarian carcinoma. Eur J Obstet Gynecol Reprod Biol. 2020;244:101–5. https://doi.org/10.1016/j.ejogrb.2019.10.047.
Nurgali K, Jagoe RT, Abalo R. Editorial: adverse effects of cancer chemotherapy: anything new to improve tolerance and reduce sequelae? Front Pharmacol. 2018;9:245 https://doi.org/10.3389/fphar.2018.00245.
Alagarsamy V, Chitra K, Saravanan G, Solomon VR, Sulthana MT, Narendhar B. An overview of quinazolines: pharmacological significance and recent developments. Eur J Medicinal Chem. 2018;151:628–85. https://doi.org/10.1016/j.ejmech.2018.03.076.
Leila E, Zeinab F, Soghra K, Zahra R, Razieh S, Ebrahim H, et al. 2-(Chloromethyl)-3-phenylquinazolin-4(3H) -ones as potent anticancer agents; cytotoxicity, molecular docking and in silico studies. J Iran Chem Soc. 2021;18:1877–89. https://doi.org/10.1007/s13738-021-02168-1.
Zhang Y, Chen L, Xu H, Li X, Zhao L, Wang W, et al. 6,7-Dimorpholinoalkoxy quinazoline derivatives as potent EGFR inhibitors with enhanced antiproliferative activities against tumor cells. Eur J Medicinal Chem. 2018;147:77–89. https://doi.org/10.1016/j.ejmech.2018.01.090.
Desroches J, Kieffer C, Primas N, Hutter S, Gellis A, El-Kashef H, et al. Discovery of new hit-molecules targeting Plasmodium falciparum through a global SAR study of the 4-substituted-2-trichloromethylquinazoline antiplasmodial scaffold. Eur J Medicinal Chem. 2017;125:68–86. https://doi.org/10.1016/j.ejmech.2016.09.029.
Mendoza-Martinez C, Correa-Basurto J, Nieto-Meneses R, Marquez-Navarro A, Aguilar-Suarez R, Dinora Montero-Cortes M, et al. Design, synthesis and biological evaluation of quinazoline derivatives as anti-trypanosomatid and anti-plasmodial agents. Eur J Medicinal Chem. 2015;96:296–307. https://doi.org/10.1016/j.ejmech.2015.04.028.
Nurgali K, Jagoe RT, Abalo R. Editorial: adverse effects of cancer chemotherapy: anything new to improve tolerance and reduce sequelae? Front Pharmacol. 2018;9:245 https://doi.org/10.1016/j.antiviral.2016.08.005.
Kumar KS, Ganguly S, Veerasamy R, De Clercq E. Synthesis, antiviral activity and cytotoxicity evaluation of Schiff bases of some 2-phenyl quinazoline-4(3)H-ones. Eur J Medicinal Chem. 2010;45:5474–9. https://doi.org/10.1016/j.ejmech.2010.07.058.
Piotrowska DG, Andrei G, Schols D, Snoeck R, Lysakowska M. Synthesis, anti-varicella-zoster virus and anti-cytomegalovirus activity of quinazoline-2,4-diones containing isoxazolidine and phosphonate substructures. Eur J Medicinal Chem. 2017;126:84–100. https://doi.org/10.1016/j.ejmech.2016.10.002.
Jatav V, Mishra P, Kashaw S, Stables JP. CNS depressant and anticonvulsant activities of some novel 3-[5-substituted 1,3,4-thiadiazole-2-yl]-2-styryl quinazoline-4(3H)-ones. Eur J Med Chem. 2008;43:1945–54. https://doi.org/10.1016/j.ejmech.2007.12.003.
Abdelmalek CM, Hu Z, Kronenberger T, Küblbeck J, Kinnen FJM, Hesse SS, et al. Gefitinib-tamoxifen hybrid ligands as potent agents against triple-negative breast cancer. J Medicinal Chem. 2022;65:4616–32. https://doi.org/10.1021/acs.jmedchem.1c01646.
Lin P, An F, Xu X, Zhao L, Liu L, Liu N, et al. Chronopharmacodynamics and mechanisms of antitumor effect induced by erlotinib in xenograft-bearing nude mice. Biochem Biophys Res Commun. 2015;460:362–7. https://doi.org/10.1016/j.bbrc.2015.03.039.
Imai H, Kaira K, Suzuki K, Anzai M, Tsuda T, Ishizuka T, et al. A phase II study of afatinib treatment for elderly patients with previously untreated advanced non-small-cell lung cancer harboring EGFR mutations1. Lung Cancer. 2018;126:41–7. https://doi.org/10.1016/j.lungcan.2018.10.014.
Gomez HL, Doval DC, Chavez MA, Ang PCS, Aziz Z, Nag S, et al. Efficacy and safety of lapatinib as first-line therapy for ErbB2-amplified locally advanced or metastatic breast cancer. J Clin Oncol. 2008;26:2999–3005. https://doi.org/10.1200/jco.2007.14.0590.
Yadagiri B, Gurrala S, Bantu R, Nagarapu L, Polepalli S, Srujana G, et al. Synthesis and evaluation of benzosuberone embedded with 1, 3, 4-oxadiazole, 1, 3, 4-thiadiazole and 1, 2, 4-triazole moieties as new potential anti proliferative agents. Bioorg Medicinal Chem Lett. 2015;25:2220–4. https://doi.org/10.1016/j.bmcl.2015.03.032.
Gomha SM, Kheder NA, Abdelaziz MR, Mabkhot YN, Alhajoj AM. A facile synthesis and anticancer activity of some novel thiazoles carrying 1,3,4-thiadiazole moiety. Chem Cent J. 2017;11:25 https://doi.org/10.1186/s13065-017-0255-7.
Shukla K, Ferraris DV, Thomas AG, Stathis M, Duvall B, Delahanty G, et al. Design, synthesis, and pharmacological evaluation of bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl sulfide (BPTES) analogs as glutaminase inhibitors. J Medicinal Chem. 2012;55:10551–63. https://doi.org/10.1021/jm301191p.
Shen L-H, Li H-Y, Shang H-X, Tian S-T, Lai Y-S, Liu L-J. Synthesis and cytotoxic evaluation of new colchicine derivatives bearing 1,3,4-thiadiazole moieties. Chin Chem Lett. 2013;24:299–302. https://doi.org/10.1016/j.cclet.2013.01.052.
Mao R, Shao J, Zhu K, Zhang Y, Ding H, Zhang C, et al. A potent, selective and cell active protein arginine methyltransferase 5 (PRMT5) inhibitor developed by structure-based virtual screening and hit optimization. J Medicinal Chem. 2017;60:6289–304. https://doi.org/10.1021/acs.jmedchem.7b00587.
Komar M, Kraljevi´c TG, Jerkovi´c I, Molnar M. Application of deep eutectic solvents in the synthesis of substituted 2-mercaptoquinazolin-4(3H)-Ones: a comparison of selected green Chemistry Methods. Molecules. 2022;27:558 https://doi.org/10.3390/molecules27020558.
Funding
This work was supported by the the National Natural Science Foundation of China (No. U21A20416) and Opening Fund from State Key Laboratory of Esophageal Cancer Prevention & Treatment (No. K2020000X).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Si, X., Dai, H., Gao, C. et al. Synthesis and antitumor activity evaluation in vitro of 4-aminoquinazoline derivatives containing 1,3,4-thiadiazole. Med Chem Res 31, 1384–1392 (2022). https://doi.org/10.1007/s00044-022-02913-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-022-02913-y