Abstract
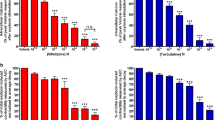
Growing evidence pointed out that guanine-based purines are able to modulate smooth muscle contractile activity of blood vessels and gastrointestinal tract. Since, so far, possible guanine-based purine modulation of uterine musculature is unknown, the aim of the present study was to investigate in vitro, using organ bath technique, guanosine and guanine effects on spontaneous uterine contraction, and uterine contraction induced by K+-depolarization and oxytocin in a non-pregnant rat. Guanosine, but not guanine, reduced the amplitude of spontaneous contraction of the uterine muscle in a dose-dependent manner. The inhibitory response was antagonized by S-(4-nitrobenzyl)-6-thioinosine (NBTI), a membrane nucleoside transporter inhibitor, but persisted in the presence of theophylline, a nonselective adenosine receptor antagonist, or propanolol, β1/β2 adrenoreceptor antagonist or blockers of a nitrergic pathway. In addition, potassium channel blockers did not influence guanosine-induced effects. Guanosine was able to inhibit the external calcium (Ca2+) influx-induced contraction, but it did not affect the contraction induced by high-KCl solution, indicating that guanosine does not interact with L-type voltage-gated calcium channel. Guanosine prevented/reduced uterine contractions induced by oxytocin, even in the absence of external calcium. In conclusion, guanosine is able to reduce both spontaneous and oxytocin-induced contractions of rat myometrium, likely subsequently to its intracellular intake. The blockade of extracellular Ca2+ influx and reduction of Ca2+ release from the intracellular store are the mechanisms involved in the guanosine-induced tocolytic effects.







Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aguilar HN, Mitchell BF (2010) Physiological pathways and molecular mechanisms regulating uterine contractility. Hum Reprod Update 16:725–744. https://doi.org/10.1093/HUMUPD/DMQ016
Benowitz LI, Jing Y, Tabibiazar R et al (1998) Axon outgrowth is regulated by an intracellular purine-sensitive mechanism in retinal ganglion cells. J Biol Chem 273:29626–29634. https://doi.org/10.1074/JBC.273.45.29626
Burnstock G (2014) Purinergic signalling in the reproductive system in health and disease. Purinergic Signal 10:157. https://doi.org/10.1007/S11302-013-9399-7
Chaud M, Franchi AM, Rettori V et al (1997) Nitric oxide in the contractile action of bradykinin, oxytocin, and prostaglandin F2 α in the estrogenized rat uterus. Proc Natl Acad Sci U S A 94:11049–11054. https://doi.org/10.1073/PNAS.94.20.11049
Dabertrand F, Morel JL, Sorrentino V et al (2006) Modulation of calcium signalling by dominant negative splice variant of ryanodine receptor subtype 3 in native smooth muscle cells. Cell Calcium 40:11–21. https://doi.org/10.1016/J.CECA.2006.03.008
Dal-Cim T, Ludka FK, Martins WC et al (2013) Guanosine controls inflammatory pathways to afford neuroprotection of hippocampal slices under oxygen and glucose deprivation conditions. J Neurochem 126:437–450. https://doi.org/10.1111/JNC.12324
Di Liberto V, Mudò G, Garozzo R, et al (2016) The guanine-based purinergic system: the tale of an orphan neuromodulation. Front Pharmacol 7:. https://doi.org/10.3389/FPHAR.2016.00158
Fujiwara T, Itoh T, Kubota Y, Kuriyama H (1989) Effects of guanosine nucleotides on skinned smooth muscle tissue of the rabbit mesenteric artery. J Physiol 408:535–547. https://doi.org/10.1113/JPHYSIOL.1989.SP017474
Gillman TA, Pennefather JN (1998) Evidence for the presence of both P1 and P2 purinoceptors in the rat myometrium. Clin Exp Pharmacol Physiol 25:592–599. https://doi.org/10.1111/J.1440-1681.1998.TB02257.X
Michel MC, Murphy TJ, Motulsky HJ (2020) New author guidelines for displaying data and reporting data analysis and statistical methods in experimental biology. J Pharmacol Exp Ther 372:136–147. https://doi.org/10.1124/jpet.119.264143
Motulsky HJ (2014) Common misconceptions about data analysis and statistics. Naunyn Schmiedebergs Arch Pharmacol 387:1017–1023. https://doi.org/10.1007/S00210-014-1037-6
Oleskovicz SPB, Martins WC, Leal RB, Tasca CI (2008) Mechanism of guanosine-induced neuroprotection in rat hippocampal slices submitted to oxygen-glucose deprivation. Neurochem Int 52:411–418. https://doi.org/10.1016/J.NEUINT.2007.07.017
Rathbone M, Pilutti L, Caciagli F, Jiang S (2008) Neurotrophic effects of extracellular guanosine. 27:666–672. https://doi.org/10.1080/15257770802143913
Tasca CI, Lanznaster D, Oliveira KA, et al (2018) Neuromodulatory effects of guanine-based purines in health and disease. Front Cell Neurosci 12:. https://doi.org/10.3389/FNCEL.2018.00376
Traversa U, Bombi G, Camaioni E et al (2003) Rat brain guanosine binding site. Biological studies and pseudo-receptor construction. Bioorg Med Chem 11:5417–5425. https://doi.org/10.1016/J.BMC.2003.09.043
Trujillo MM, Ausina P, Savineau JP et al (2000) Cellular mechanisms involved in iso-osmotic high K+ solutions-induced contraction of the estrogen-primed rat myometrium. Life Sci 66:2441–2453. https://doi.org/10.1016/S0024-3205(00)80004-1
Volpini R, Marucci G, Buccioni M et al (2011) Evidence for the existence of a specific g protein-coupled receptor activated by guanosine. ChemMedChem 6:1074–1080. https://doi.org/10.1002/CMDC.201100100
Wray S, Shmygol A (2007) Role of the calcium store in uterine contractility. Semin Cell Dev Biol 18:315–320. https://doi.org/10.1016/J.SEMCDB.2007.05.005
Zizzo MG, Auteri M, Amato A, et al (2017) Angiotensin II type II receptors and colonic dysmotility in 2,4-dinitrofluorobenzenesulfonic acid-induced colitis in rats. Neurogastroenterol Motil 29:. https://doi.org/10.1111/nmo.13019
Zizzo MG, Mulè F, Amato A et al (2013) Guanosine negatively modulates the gastric motor function in mouse. Purinergic Signal 9:655–661. https://doi.org/10.1007/S11302-013-9378-Z
Zizzo MG, Mulè F, Mastropaolo M, et al (2011) Can guanine-based purines be considered modulators of intestinal motility in rodents? Eur J Pharmacol 650:. https://doi.org/10.1016/j.ejphar.2010.09.062
Zizzo MG, Mulè F, Mastropaolo M, Serio R (2010) D1 receptors play a major role in the dopamine modulation of mouse ileum contractility. Pharmacol Res 61:. https://doi.org/10.1016/j.phrs.2010.01.015
Funding
This study was funded by a grant (FFR 2018) from Ministero dell’Università e della Ricerca Scientifica (MIUR).
Author information
Authors and Affiliations
Contributions
MGZ designed and performed the study, analyzed and interpreted the data, prepared figures, and drafted and edited the manuscript. AC performed the experiments and analyzed the data. RS participated to supervision, experimental design, interpreted the data, and edited and revised the manuscript. All the authors contributed to the critical revision and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All animal care was approved by the Animal Care and Use Ethics Committee of the University of Palermo and performed in accordance with national.
Consent to participate
Not applicable.
Consent for publication
The authors authorize the submission and publication of this article in Naunyn–Schmiedeberg’s Archives of Pharmacology.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zizzo, M.G., Cicio, A. & Serio, R. Inhibition of uterine contractility by guanine-based purines in non-pregnant rats. Naunyn-Schmiedeberg's Arch Pharmacol 396, 963–972 (2023). https://doi.org/10.1007/s00210-022-02366-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02366-5