Abstract
The aim was to assess the influence of local application of curcumin-loaded nanoparticles on an experimental model of periodontal repair. Periodontitis was induced by ligatures on both lower first molars of rats. After 15 days, ligatures were removed (“treatment”) and animals were randomly allocated to three experimental groups (n = 8/group): (i) 0.05 mg/ml curcumin-loaded nanoparticles, (ii) empty nanoparticles (vehicle control), and (iii) sterile saline (negative control). Experimental treatments were administered locally on days 0, 3, 5, 7, 9, and 11 after ligature removal. Animals were euthanized at 7 and 14 days. Bone repair was assessed by microcomputer tomography (µCT). Histological sections were stained with hematoxylin/eosin (H/E), Picrosirius Red, and Masson’s trichrome. Expression of Runx-2 was studied by immunohistochemistry. Gene expression of Itgam, Arg1, and Inos was assessed by RT-qPCR. At 7 days, there was increased gene expression of Itgam and Arg1 and of the relative expression of Arg1/Inos in curcumin-treated animals, but no difference in any other outcomes. At 14 days, curcumin-loaded nanoparticles significantly increased bone repair and collagen content, as well as the number of osteocytes, percentage of extracellular matrix, and expression of Runx2. The results demonstrate that local administration of curcumin-loaded nanoparticles enhanced tissue repair in an experimental model of periodontal repair. Nanoparticle-encapsulated curcumin enhances early post-treatment repair of periodontal tissues.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abdollahi E, Momtazi AA, Johnston TPSA (2018) Therapeutic effects of curcumin in inflammatory and immune-mediated diseases: a nature-made jack-of-all-trades? J Cell Physiol 233(2):830–848. https://doi.org/10.1002/jcp.25778
Abolaji AO et al (2020) Curcumin attenuates copper-induced oxidative stress and neurotoxicity in Drosophila melanogaster. Toxicol Reports 7(January):261–268. https://doi.org/10.1016/j.toxrep.2020.01.015
Anuradha BR et al (2015) Evaluation of anti-inflammatory effects of curcumin gel as an adjunct to scaling and root planing: a clinical study. J Int Oral Heal 7(7):90–93
Banaudha KK et al (1999) Curcumin enhances wound healing in streptozotocin induced diabetic rats and genetically diabetic mice. Wound Repair Regen 7:362–374. https://doi.org/10.1046/j.1524-475x.1999.00362.x
Banerjee M et al (2003) Modulation of inflammatory mediators by ibuprofen and curcumin treatment during chronic inflammation in rat #. Immunopharmacol Immunotoxicol 25(2):213–224. https://doi.org/10.1081/IPH-120020471
Bonferoni MC et al (2017) Nanoparticle formulations to enhance tumor targeting of poorly soluble polyphenols with potential anticancer properties. Semin Cancer Biol 46:205–214. https://doi.org/10.1016/j.semcancer.2017.06.010
Borges JS et al (2020) Does systemic oral administration of curcumin effectively reduce alveolar bone loss associated with periodontal disease? A systematic review and meta-analysis of preclinical in vivo studies. J Funct Foods 75(September):104226. https://doi.org/10.1016/j.jff.2020.104226
Bose S et al (2018) Effects of PCL, PEG and PLGA polymers on curcumin release from calcium phosphate matrix for in vitro and in vivo bone regeneration. Mater Today Chem 8:110–120. https://doi.org/10.1016/j.mtchem.2018.03.005
Busari ZA et al (2017) Antiplasmodial activity and toxicological assessment of curcumin PLGA-encapsulated nanoparticles. Front Pharmacol 8(SEP):1–9. https://doi.org/10.3389/fphar.2017.00622
Campbell L et al (2013) Local arginase 1 activity is required for cutaneous wound healing. J Invest Dermatol 133(10):2461–2470. https://doi.org/10.1038/jid.2013.164
Cobb CM (2002) Clinical significance of non-surgical periodontal therapy: an evidence-based perspective of scaling and root planing. J Clin Periodontol 29(SUPPL. 2):22–32. https://doi.org/10.1034/j.1600-051x.29.s2.4.x
Dai M et al (2009) Chitosan-alginate sponge : preparation and application in curcumin delivery for dermal wound healing in rat. J Biomed Biotechnol 2009:595126. https://doi.org/10.1155/2009/595126
Das A et al (2015) Monocyte and macrophage plasticity in tissue repair and regeneration. Am J Pathol 185(10):2596–2606. https://doi.org/10.1016/j.ajpath.2015.06.001
Deas DE et al (2016) Scaling and root planing vs. conservative surgery in the treatment of chronic periodontitis. Periodontology 2000 71(1):128–139. https://doi.org/10.1111/prd.12114
Deonarine K et al (2007) Gene expression profiling of cutaneous wound healing. J Transl Med 5:1–11. https://doi.org/10.1186/1479-5876-5-11
Donos N et al (2020) The adjunctive use of host modulators in non-surgical periodontal therapy. A systematic review of randomized, placebo-controlled clinical studies. J Clin Periodontol 47(S22):199–238. https://doi.org/10.1111/jcpe.13232
Dovigo LN et al (2011) Investigation of the photodynamic effects of curcumin against Candida albicans. Photochem Photobiol 87(4):895–903. https://doi.org/10.1111/j.1751-1097.2011.00937.x
Durgaprasad S et al (2011) Effect of a topical curcumin preparation (BIOCURCUMAX) on burn wound healing in rats. J Pharm Biomed Sci 08(08):1–3. https://doi.org/10.1007/s10735-012-9452-9
Edwards RL et al (2017) The anti-inflammatory activity of curcumin is mediated by its oxidative metabolites. J Biol Chem 292(52):21243–21252. https://doi.org/10.1074/jbc.RA117.000123
Fernandes NAR et al (2021) Chacone T4, a novel chalconic compound, inhibits inflammatory bone resorption in vivo and supresses osteoclastogenesis in vitro. J Periodontal Res 00:1–10. https://doi.org/10.1111/jre.12857
Gandhi KK et al (2019) Effectiveness of the adjunctive use of ozone and chlorhexidine in patients with chronic periodontitis. BDJ Open 5(1):3–6. https://doi.org/10.1038/s41405-019-0025-9
Gao S et al (2015) Curcumin induces M2 macrophage polarization by secretion IL-4 and/or IL-13. J Mol Cell Cardiol 85:131–139. https://doi.org/10.1016/j.yjmcc.2015.04.025
Giannelli M et al (2018) Treatment of severe periodontitis with a laser and light-emitting diode (LED) procedure adjunctive to scaling and root planing: a double-blind, randomized, single-center, split-mouth clinical trial investigating its efficacy and patient-reported outcomes. Lasers Med Sci 33(5):991–1002. https://doi.org/10.1007/s10103-018-2441-9
Gu Q et al (2012) Curcumin increases rat mesenchymal stem cell osteoblast differentiation but inhibits adipocyte differentiation. Pharmacogn Mag 8(31):202–208. https://doi.org/10.4103/0973-1296.99285
Guimaraes MR, Coimbra LS, de Aquino SGSL, Kirkwood KLRCJ (2012) Potent anti-inflammatory effects of systemically-administered curcumin modulates periodontal disease in vivo. J Periodontal Res 46(2):269–279. https://doi.org/10.1111/j.1600-0765.2010.01342.x.Potent
Guimaraes-Stabili MR et al (2019) Systemic administration of curcumin or piperine enhances the periodontal repair: a preliminary study in rats. Clin Oral Investig 23(8):3297–3306. https://doi.org/10.1007/s00784-018-2755-9
Ibrahim SG et al (2020) Dimethyl fumarate and curcumin attenuate hepatic ischemia/reperfusion injury via Nrf2/HO-1 activation and anti-inflammatory properties. Int Immunopharmacol 80(December 2019):106131. https://doi.org/10.1016/j.intimp.2019.106131
Jagetia GC, Aggarwal BB (2007) “ Spicing up ” of the immune system by curcumin. J Clin Immunol 27(1):19–35. https://doi.org/10.1007/s10875-006-9066-7
Joe B et al (2004) Biological properties of curcumin-cellular and molecular mechanisms of action biological properties of curcumin-cellular and molecular. Crit Rev Food Sci Nutr 44(2):97–111. https://doi.org/10.1080/10408690490424702
Kant V et al (2017) Combined effect of substance P and curcumin on cutaneous wound healing in diabetic rats. J Surg Res 212:130–145. https://doi.org/10.1016/j.jss.2017.01.011
Khalil NM et al (2013) Pharmacokinetics of curcumin-loaded PLGA and PLGA-PEG blend nanoparticles after oral administration in rats. Colloids Surfaces B Biointerfaces 101:353–360. https://doi.org/10.1016/j.colsurfb.2012.06.024
Kulac M et al (2013) The effects of topical treatment with curcumin on burn wound healing in rats. J Mol Hist 44:83–90. https://doi.org/10.1007/s10735-012-9452-9
Mani H et al (2002) Curcumin differentially regulates TGF- β 1, its receptors and nitric oxide synthase during impaired wound healing. BioFactors 16:29–43. https://doi.org/10.1002/biof.5520160104
Marcos-Garcés V et al (2017) Comparative measurement of collagen bundle orientation by Fourier analysis and semiquantitative evaluation: reliability and agreement in Masson’s trichrome, Picrosirius red and confocal microscopy techniques. J Microsc 267(2):130–142. https://doi.org/10.1111/jmi.12553
Merrell JG et al (2009) Curcumin-loaded poly(e-caprolactone) nanofibres: diabetic wound dressing with anti-oxidant and anti-inflammatory properties. Clin Exp Pharmacol Physiol 36:1149–1156. https://doi.org/10.1111/j.1440-1681.2009.05216.x
Mohanty C et al (2012) Sustained wound healing activity of curcumin loaded oleic acid based polymeric bandage in a rat model. Mol Pharm 9:2801–2811. https://doi.org/10.1021/mp300075u
Molon RS et al (2018) Characterization of ligature-induced experimental periodontitis. Microsc Res Tech 81(12):1412–1421. https://doi.org/10.1002/jemt.23101
Momtazi-Borojeni AA et al (2019) Curcumin as a potential modulator of M1 and M2 macrophages: new insights in atherosclerosis therapy. Heart Fail Rev 24(3):399–409. https://doi.org/10.1007/s10741-018-09764-z
Mosser DM, Edwards JP (2009) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8(12):958–969. https://doi.org/10.1038/nri2448.Exploring
Ortega AAC et al (2016) Overexpression of Bcl-2, SOCS 1, 3 and Cdh 1, 2 are associated with the early neoplasic changes in modified 4-nitroquinoline 1-oxide-induced murine oral cancer model. J Oral Pathol Med 45(8):573–580. https://doi.org/10.1111/jop.12413
Panahi Y et al (2018) Effects of curcuminoids plus piperine on glycemic, hepatic and inflammatory biomarkers in patients with type 2 diabetes mellitus: a randomized double-blind placebo-controlled trial. Drug Res (Stuttg) 68(7):403–409. https://doi.org/10.1055/s-0044-101752
Panchatcharam M et al (2006) Curcumin improves wound healing by modulating collagen and decreasing reactive oxygen species. Mol Cell Biochem 290(1–2):87–96. https://doi.org/10.1007/s11010-006-9170-2
Papapanou PN et al (2018) Periodontitis: consensus report of workgroup 2 of the 2017 world workshop on the classification of periodontal and peri-implant diseases and conditions. J Periodontol 89(March):S173–S182. https://doi.org/10.1002/JPER.17-0721
Pérez-Pacheco CG et al (2020) Local application of curcumin-loaded nanoparticles as an adjunct to scaling and root planing in periodontitis: randomized, placebo-controlled, double-blind split-mouth clinical trial. Clin Oral Investig. https://doi.org/10.1007/s00784-020-03652-3
Ravindranath V, Chandrasekhara N (1980) Absorption and tissue distribution of curcumin in rats. Toxicology 16:259–265. https://doi.org/10.1016/0300-483x(80)90122-5
Sarkar N, Bose S (2019) Liposome-encapsulated curcumin-loaded 3D printed scaffold for bone tissue engineering. ACS Appl Mater Interfaces 11(19):17184–17192. https://doi.org/10.1021/acsami.9b01218
Shah A, Amini-Nik S (2017) The role of phytochemicals in the inflammatory phase of wound healing. Int J Mol Sci 18(5):1068. https://doi.org/10.3390/ijms18051068
Sharma AK et al (2016) Overview of biopolymers as carriers of antiphlogistic agents for treatment of diverse ocular inflammations. Mater Sci Eng C Mater Biol Appl 67:779–791. https://doi.org/10.1016/j.msec.2016.05.060
Shearer JD et al (1997) Differential regulation of macrophage arginine metabolism: a proposed role in wound healing. Am J Physiol - Endocrinol Metab 272(2 Pt 1):E181–E190. https://doi.org/10.1152/ajpendo.1997.272.2.e181
Sidhu GS et al (1998) Enhancement of wound healing by curcumin in animals. Wound Repair Regen 6:167–177. https://doi.org/10.1046/j.1524-475x.1998.60211.x
Skyvalidas D et al (2020) Curcumin mediates attenuation of pro-inflammatory interferon γ and interleukin 17 cytokine responses in psoriatic disease, strengthening its role as a dietary immunosuppressant. Nutr Res 75:95–108. https://doi.org/10.1016/j.nutres.2020.01.005
Theodoro LH et al (2017) Curcumin photodynamic effect in the treatment of the induced periodontitis in rats. Lasers Med Sci 32(8):1783–1791. https://doi.org/10.1007/s10103-017-2261-3
Ubeyitogullari A, Ciftci ON (2019) A novel and green nanoparticle formation approach to forming low-crystallinity curcumin nanoparticles to improve curcumin’s bioaccessibility. Sci Rep 9(1):1–11. https://doi.org/10.1038/s41598-019-55619-4
Withers SS et al (2018) In vitro and in vivo activity of liposome-encapsulated curcumin for naturally occurring canine cancers. Vet Comp Oncol 16(4):571–579. https://doi.org/10.1111/vco.12424
Witte MB et al (2002) Upregulation of arginase expression in wound-derived fibroblasts. J Surg Res 105(1):35–42. https://doi.org/10.1006/jsre.2002.6443
Xiao CJ et al (2018) Protective effect and related mechanisms of curcumin in rat experimental periodontitis. Head Face Med 14(1):1–8. https://doi.org/10.1186/s13005-018-0169-1
Zambrano LMG et al (2018) Local administration of curcumin-loaded nanoparticles effectively inhibits inflammation and bone resorption associated with experimental periodontal disease. Sci Rep 8(1):1–11. https://doi.org/10.1038/s41598-018-24866-2
Zhao S et al (2017) Curcumin-loaded redox response of self-assembled micelles for enhanced antitumor and anti-inflammation efficacy. Int J Nanomedicine 12:2489–2504. https://doi.org/10.2147/IJN.S123190
Funding
This work was supported by the São Paulo Research Foundation (FAPESP 2020/11916-0) and Brazilian National Council for Scientific and Technological Development (CNPq 440588/2014-18).
Author information
Authors and Affiliations
Contributions
CGPP and NARF conducted experiments, data analyses, manuscript preparation, and writing—review and editing; ACC, DPF, AFS, and MCZ conducted experiments; CFA and FLP—curcumin-loaded nanoparticles synthesis; MRGS—conceptualization and experimental design; CRJ—conceptualization, experimental design, supervision, manuscript preparation, writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval
The experimental protocol was approved by the Ethical Committee for Animal Use (CEUA) of the School of Dentistry at Araraquara − UNESP (protocol number no. 37/2017) and performed in accordance with the guidelines from the National Council for the Control of Animal Experimentation the Brazilian College for Animal Experimentation (CONCEA).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

SI Fig. 1
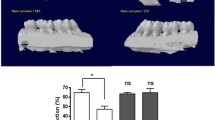
Bone volume at 7 days. (a) Quantification of the ratio of mineralized tissue (BV/TV). (b) Representative images of each experimental condition. The bars indicate the percentage average value and vertical lines the standard error of the mean (SEM) (PNG 36 kb)

SI Fig. 2
Collagen content in the gingival tissues at 7 days. (a) Percentage of collagen content in Picrosirius red-stained samples. (b) Representative images of the picrosirius red staining according to the experimental group (100x magnification). (c) Percentage of collagen content in Masson’s trichrome-stained sections. (d) Representative images of Masson’s trichrome staining according to the experimental group (400x magnification). The bars indicate the percentage average value and vertical lines the standard error of the mean (SEM). (*), (**) and (***) p < 0.05. 1M = first molar; 2M = second molar (PNG 171 kb)

SI Fig. 3
Stereometric analysis of the periodontal tissues according to treatments at 7 days (a) Extracellular matrix percentage. (b) Osteocyte density. (c) Cellular infiltrate. (d) Blood vessels. The bars indicate the percentage average value or osteocyte density (osteocyte/mm2) and vertical lines the standard error of the mean (SEM). (***) p < 0.05 (PNG 29 kb)

SI Fig. 4
Immunohistochemical analysis of Runx2 according to treatments at 7 days. (a) H-score of cells positively stained with antibodies. (b) Representative images of the immunohistochemistry staining (400x magnification). The bars indicate the percentage average value and vertical lines the standard error of the mean (SEM) (PNG 90 kb)

SI Fig. 5
Expression of target genes in the gingival tissues according to treatments at 14 days. Gene expression of (a) Arg1, (b) Inos and (c) Itgam. (d) Relative gene expression of Arg1/Inos. The bars indicate the average value of gene expression and vertical lines the standard error of the mean (SEM) (PNG 34 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Perez-Pacheco, C.G., Fernandes, N.A.R., Camilli, A.C. et al. Local administration of curcumin-loaded nanoparticles enhances periodontal repair in vivo. Naunyn-Schmiedeberg's Arch Pharmacol 396, 311–321 (2023). https://doi.org/10.1007/s00210-022-02310-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02310-7