Abstract
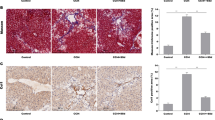
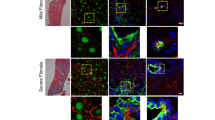
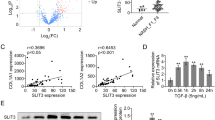
Current researches have confirmed that Smads, mediators of TGF-β signaling, are strictly controlled by domain-specific site phosphorylation in the process of hepatic disease. Usually, Smad3 phospho-isoform pSmad3L and pSmad3C are reversible and antagonistic; pSmad2L/C could act together with pSmad3L by stimulating PAI-1 expression and ECM synthesis to transmit fibrogenic signals. Our recent study found that pSmad3C mutation is supposed to perform a vigorous role on the early phase of liver injury and abates salvianolic acid B’s anti-hepatic fibrotic-carcinogenesis. However, whether pSmad3C mutation expedites pSmad2L/C-mediated signaling transduction during hepatic fibrogenesis remains vague. Presently, Smad3 gene C-terminal phosphorylation site mutation heterozygote (pSmad3C+/-) mice were constructed to probe if and how pSmad3C retards CCl4-induced hepatic fibrogenesis by inhibiting pSmad2L/C-mediated signaling transduction. Twelve 6-week-old pSmad3C+/- C57BL/6J mice were intraperitoneally injection with CCl4 for 6 weeks to induce liver fibrogenesis. Results showed that pSmad3C mutation aggravates the relative liver weight, biochemical parameters, collagenous fibers and fibrotic septa formation, contributes to fibrogenesis in HT-CCl4 mice. Furthermore, fibrotic-related proteins TGF-β1, pSmad2C, pSmad2L, and PAI-1 were also increased in CCl4-induced pSmad3C+/- mice. These results suggest that pSmad3C mutation exacerbates hepatic fibrogenesis which relates to intensifying pSmad2L/C-mediated signaling transduction.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article.
References
Attisano L, Wrana JL (2002) Signal transduction by the TGF-beta superfamily. Science 296:1646–1647. https://doi.org/10.1126/science.1071809
Bouabe H, Okkenhaug K (2013) Gene targeting in mice: a review. Methods Mol Biol 1064:315–336. https://doi.org/10.1007/978-1-62703-601-6_23
Chen D, Le TH, Shahidipour H, Read SA, Ahlenstiel G (2019) The Role of gut-derived microbial antigens on liver fibrosis initiation and progression. Cells 8(11):1324. https://doi.org/10.3390/cells8111324
Dewidar B, Meyer C, Dooley S, Meindl-Beinker AN (2019) TGF-β in hepatic stellate cell activation and liver fibrogenesis-updated 2019. Cells 8(11):1419. https://doi.org/10.3390/cells8111419
Ding H, Fang M, Gong Y, Li D, Zhang C, Wen G, Wu C, Yang J, Yang Y (2020) Smad3 gene C-terminal phosphorylation site mutation aggravates CCl(4) -induced inflammation in mice. J Cell Mol Med 24:7044–7054. https://doi.org/10.1111/jcmm.15385
Friedman SL (2003) Liver fibrosis – from bench to bedside. J Hepatol 38(Suppl 1):S38-53. https://doi.org/10.1016/s0168-8278(02)00429-4
Fujii W (2017) Generation of knock-in mouse by genome editing. Methods Mol Biol 1630:91–100. https://doi.org/10.1007/978-1-4939-7128-2_8
Furukawa F, Matsuzaki K, Mori S, Tahashi Y, Yoshida K, Sugano Y, Yamagata H, Matsushita M, Seki T, Inagaki Y, Nishizawa M, Fujisawa J, Inoue K (2003) p38 MAPK mediates fibrogenic signal through Smad3 phosphorylation in rat myofibroblasts. Hepatology 38:879–889. https://doi.org/10.1053/jhep.2003.50384
Gong Y, Li D, Li L, Yang J, Ding H, Zhang C, Wen G, Wu C, Fang Z, Hou S, Yang Y (2021) Smad3 C-terminal phosphorylation site mutation attenuates the hepatoprotective effect of salvianolic acid B against hepatocarcinogenesis. Food Chem Toxicol 147:111912. https://doi.org/10.1016/j.fct.2020.111912
Hayashi H, Sakai T (2011) Animal models for the study of liver fibrosis: new insights from knockout mouse models. Am J Physiol Gastrointest Liver Physiol 300:G729–G738. https://doi.org/10.1152/ajpgi.00013.2011
Hu HH, Chen DQ, Wang YN, Feng YL, Cao G, Vaziri ND, Zhao YY (2018) New insights into TGF-β/Smad signaling in tissue fibrosis. Chem Biol Interact 292:76–83. https://doi.org/10.1016/j.cbi.2018.07.008
Inagaki Y, Okazaki I (2007) Emerging insights into transforming growth factor beta Smad signal in hepatic fibrogenesis. Gut 56:284–292. https://doi.org/10.1136/gut.2005.088690
Indra AK, Warot X, Brocard J, Bornert JM, Xiao JH, Chambon P, Metzger D (1999) Temporally-controlled site-specific mutagenesis in the basal layer of the epidermis: comparison of the recombinase activity of the tamoxifen-inducible Cre-ER(T) and Cre-ER(T2) recombinases. Nucleic Acids Res 27:4324–4327. https://doi.org/10.1093/nar/27.22.4324
Kesteloot F, Desmoulière A, Leclercq I, Thiry M, Arrese JE, Prockop DJ, Lapière CM, Nusgens BV, Colige A (2007) ADAM metallopeptidase with thrombospondin type 1 motif 2 inactivation reduces the extent and stability of carbon tetrachloride-induced hepatic fibrosis in mice. Hepatology 46:1620–1631. https://doi.org/10.1002/hep.21868
Lambrecht J, van Grunsven LA, Tacke F (2020) Current and emerging pharmacotherapeutic interventions for the treatment of liver fibrosis. Expert Opin Pharmacother 21:1637–1650. https://doi.org/10.1080/14656566.2020.1774553
Liu X, Brenner DA (2016) Liver: DNA methylation controls liver fibrogenesis. Nat Rev Gastroenterol Hepatol 13:126–128. https://doi.org/10.1038/nrgastro.2016.16
Matsuzaki K, Kitano C, Murata M, Sekimoto G, Yoshida K, Uemura Y, Seki T, Taketani S, Fujisawa J, Okazaki K (2009) Smad2 and Smad3 phosphorylated at both linker and COOH-terminal regions transmit malignant TGF-beta signal in later stages of human colorectal cancer. Cancer Res 69:5321–5330. https://doi.org/10.1158/0008-5472.Can-08-4203
Mishina M, Sakimura K (2007) Conditional gene targeting on the pure C57BL/6 genetic background. Neurosci Res 58:105–112. https://doi.org/10.1016/j.neures.2007.01.004
Recknagel RO, Glende EA Jr, Dolak JA, Waller RL (1989) Mechanisms of carbon tetrachloride toxicity. Pharmacol Ther 43:139–154. https://doi.org/10.1016/0163-7258(89)90050-8
Schuppan D (2015) Liver fibrosis: Common mechanisms and antifibrotic therapies. Clin Res Hepatol Gastroenterol 39(Suppl 1):S51–S59. https://doi.org/10.1016/j.clinre.2015.05.005
Seifert WF, Bosma A, Brouwer A, Hendriks HF, Roholl PJ, van Leeuwen RE, van Thiel-de Ruiter GC, Seifert-Bock I, Knook DL (1994) Vitamin A deficiency potentiates carbon tetrachloride-induced liver fibrosis in rats. Hepatology 19:193–201
Sigmund CD (1993) Major approaches for generating and analyzing transgenic mice. An overview. Hypertension 22:599–607. https://doi.org/10.1161/01.hyp.22.4.599
Tacke F, Trautwein C (2015) Mechanisms of liver fibrosis resolution. J Hepatol 63:1038–1039. https://doi.org/10.1016/j.jhep.2015.03.039
Walton KL, Johnson KE, Harrison CA (2017) Targeting TGF-β Mediated SMAD signaling for the prevention of fibrosis. Front Pharmacol 8:461. https://doi.org/10.3389/fphar.2017.00461
Wang R, Yu X-Y, Guo Z-Y, Wang Y-J, Wu Y, Yuan Y-F (2012) Inhibitory effects of salvianolic acid B on CCl4-induced hepatic fibrosis through regulating NF-κB/IκBα signaling. J Ethnopharmacol 144:592–598. https://doi.org/10.1016/j.jep.2012.09.048
Wu S, Yue Y, Tian H, Li Z, Li X, He W, Ding H (2013) Carthamus red from Carthamus tinctorius L. exerts antioxidant and hepatoprotective effect against CCl(4)-induced liver damage in rats via the Nrf2 pathway. J Ethnopharmacol 148:570–578. https://doi.org/10.1016/j.jep.2013.04.054
Yamaguchi T, Matsuzaki K, Inokuchi R, Kawamura R, Yoshida K, Murata M, Fujisawa J, Fukushima N, Sata M, Kage M, Nakashima O, Tamori A, Kawada N, Tsuneyama K, Dooley S, Seki T, Okazaki K (2013) Phosphorylated Smad2 and Smad3 signaling: Shifting between tumor suppression and fibro-carcinogenesis in chronic hepatitis C. Hepatol Res 43:1327–1342. https://doi.org/10.1111/hepr.12082
Yanguas SC, Cogliati B, Willebrords J, Maes M, Colle I, van den Bossche B, de Oliveira C, Andraus W, Alves VAF, Leclercq I, Vinken M (2016) Experimental models of liver fibrosis. Arch Toxicol 90:1025–1048. https://doi.org/10.1007/s00204-015-1543-4
Yoshida K, Matsuzaki K (2012) Differential regulation of TGF-β/Smad signaling in hepatic stellate cells between acute and chronic liver injuries. Front Physiol 3:53. https://doi.org/10.3389/fphys.2012.00053
Yoshida K, Matsuzaki K, Murata M, Yamaguchi T, Suwa K, Okazaki K (2018) Clinico-pathological importance of TGF-β/phospho-Smad signaling during Human hepatic fibrocarcinogenesis. Cancers (basel) 10(6):183. https://doi.org/10.3390/cancers10060183
Yoshida K, Murata M, Yamaguchi T, Matsuzaki K, Okazaki K (2016) Reversible human TGF-β signal shifting between tumor suppression and fibro-carcinogenesis: implications of Smad phospho-isoforms for hepatic epithelial-mesenchymal transitions. J Clin Med 5(1):7. https://doi.org/10.3390/jcm5010007
Zeng B, Su M, Chen Q, Chang Q, Wang W, Li H (2017) Protective effect of a polysaccharide from Anoectochilus roxburghii against carbon tetrachloride-induced acute liver injury in mice. J Ethnopharmacol 200:124–135. https://doi.org/10.1016/j.jep.2017.02.018
Acknowledgements
We thank Prof. Koichi Matsuzaki for providing us with rabbit polyclonal anti-pSmad2L (Ser250/255) antibody.
Funding
This work was supported by the National Natural Science Foundation of China (No. 82074073, No. 81874354, and No. 81573652).
Author information
Authors and Affiliations
Contributions
Contributors JY and YFG were responsible for conceptualization, investigation, and writing the original draft. WJX was responsible for resources and investigation and revised the manuscript. LLL was responsible for methodology, visualization, software, and analyzation of the data. ZHS was responsible for resources and investigation. QW and YHH were responsible for review and editing the manuscript. CZ, CCL, and ZRF were responsible for writing, review, and editing the manuscript. YY was responsible for the supervision and funding acquisition. All authors read and approved the manuscript, and all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval
Experimental Animal Ethics Committee of Anhui Medical University approved all experiments and all procedures, and processes in this study were carried out according to the guidelines for the Ethical Care of Experimental Animals (Number: LLSC20190753).
Consent to participate
Not applicable.
Consent for publication
All authors approved the manuscript to be published.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, J., Gong, Y., Xu, W. et al. Smad3 gene C-terminal phosphorylation site mutation exacerbates CCl4-induced hepatic fibrogenesis by promoting pSmad2L/C-mediated signaling transduction . Naunyn-Schmiedeberg's Arch Pharmacol 394, 1779–1786 (2021). https://doi.org/10.1007/s00210-021-02114-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-021-02114-1