Abstract
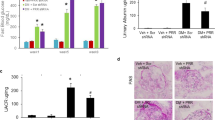
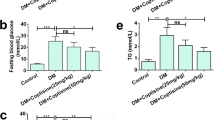
Poly(ADP-ribose) polymerase (PARP) enzyme contributes to nephropathy, a serious diabetic complication which may lead to end-stage renal disease. The study aims to investigate the effect of PARP over-activation on kidney functions in a type 2 diabetic rat model. The study also tests the therapeutic use of PARP inhibitors in diabetic nephropathy. Type 2 diabetes was induced in adult male rats by high-fructose/high-fat diet and low streptozotocin dose. Then, the PARP inhibitor 4-aminobenzamide (4-AB) was administered daily for 10 weeks. At the end, urine samples were collected to measure urine creatinine, albumin, and total proteins. PARP activity, superoxide dismutase (SOD) activity, and nitrite content were measured in kidney tissue homogenate. Glucose, fructosamine, insulin, and tumor necrosis factor-alpha (TNF-α) were measured in serum. Furthermore, histological studies, collagen deposition, and immunofluorescence of nuclear factor kappa B (NFκB) and transforming growth factor beta1 (TGF-β1) were carried out. PARP enzyme activity was significantly higher in the diabetic group and was significantly reduced by 4-AB administration. Diabetic animals had clear nephropathy indicated by proteinuria and increased albumin excretion rate (AER) which were significantly decreased by PARP inhibition. In addition, PARP inhibition increased creatinine clearance in diabetic animals and reduced renal TGF-β1 and glomerular fibrosis. Moreover, PARP inhibition alleviated the elevated serum TNF-α level, renal NFκB, nitrite, and the decrease in SOD activity in diabetic animals. However, PARP inhibition did not significantly affect neither hyperglycemia nor insulin sensitivity. PARP enzyme inhibition alleviates diabetic nephropathy through decreasing inflammation, oxidative stress, and renal fibrosis.






Similar content being viewed by others
References
Afsar B, Elsurer R (2012) Comparison of renal resistive index among patients with Type 2 diabetes with different levels of creatinine clearance and urinary albumin excretion. Diabet Med 29(8):1043–1046. doi:10.1111/j.1464-5491.2012.03593.x
Arora MK, Singh UK (2013) Molecular mechanisms in the pathogenesis of diabetic nephropathy: an update. Vasc Pharmacol 58(4):259–271. doi:10.1016/j.vph.2013.01.001
Beneke S (2008) Poly(ADP-ribose) polymerase activity in different pathologies—the link to inflammation and infarction. Exp Gerontol 43(7):605–614. doi:10.1016/j.exger.2008.04.008
Ben-Hur E, Chen CC, Elkind MM (1985) Inhibitors of poly(adenosine diphosphoribose) synthetase, examination of metabolic perturbations, and enhancement of radiation response in Chinese hamster cells. Cancer Res 45(5):2123–2127
Bouwmeester T, Bauch A, Ruffner H, Angrand PO, Bergamini G, Croughton K, Cruciat C, Eberhard D, Gagneur J, Ghidelli S, Hopf C, Huhse B, Mangano R, Michon AM, Schirle M, Schlegl J, Schwab M, Stein MA, Bauer A, Casari G, Drewes G, Gavin AC, Jackson DB, Joberty G, Neubauer G, Rick J, Kuster B, Superti-Furga G (2004) A physical and functional map of the human TNF-alpha/NF-kappa B signal transduction pathway. Nat Cell Biol 6(2):97–105. doi:10.1038/ncb1086
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414(6865):813–820. doi:10.1038/414813a
Burkart V, Wang ZQ, Radons J, Heller B, Herceg Z, Stingl L, Wagner EF, Kolb H (1999) Mice lacking the poly(ADP-ribose) polymerase gene are resistant to pancreatic beta-cell destruction and diabetes development induced by streptozocin. Nat Med 5(3):314–319. doi:10.1038/6535
Cacho J, Sevillano J, de Castro J, Herrera E, Ramos MP (2008) Validation of simple indexes to assess insulin sensitivity during pregnancy in Wistar and Sprague-Dawley rats. Am J Physiol Endocrinol Metab 295(5):E1269–E1276. doi:10.1152/ajpendo.90207.2008
Cutroneo KR (2007) TGF-beta-induced fibrosis and SMAD signaling: oligo decoys as natural therapeutics for inhibition of tissue fibrosis and scarring. Wound Repair Regen 15(Suppl 1):S54–S60. doi:10.1111/j.1524-475X.2007.00226.x
Dai S, McNeill JH (1995) Fructose-induced hypertension in rats is concentration- and duration-dependent. J Pharmacol Toxicol Methods 33(2):101–107. doi:10.1016/1056-8719(94)00063-A
Drel VR, Xu W, Zhang J, Pavlov IA, Shevalye H, Slusher B, Obrosova IG (2009) Poly(adenosine 5'-diphosphate-ribose) polymerase inhibition counteracts multiple manifestations of experimental type 1 diabetic nephropathy. Endocrinology 150(12):5273–5283. doi:10.1210/en.2009-0628
Drel VR, Pacher P, Stavniichuk R, Xu W, Zhang J, Kuchmerovska TM, Slusher B, Obrosova IG (2011) Poly(ADP-ribose)polymerase inhibition counteracts renal hypertrophy and multiple manifestations of peripheral neuropathy in diabetic Akita mice. Int J Mol Med 28(4):629–635. doi:10.3892/ijmm.2011.709
Dronavalli S, Duka I, Bakris GL (2008) The pathogenesis of diabetic nephropathy. Nat Clin Pract Endocrinol Metab 4(8):444–452. doi:10.1038/ncpendmet0894
Eijkelkamp WB, Zhang Z, Remuzzi G, Parving HH, Cooper ME, Keane WF, Shahinfar S, Gleim GW, Weir MR, Brenner BM, de Zeeuw D (2007) Albuminuria is a target for renoprotective therapy independent from blood pressure in patients with type 2 diabetic nephropathy: post hoc analysis from the reduction of endpoints in NIDDM with the angiotensin II antagonist losartan (RENAAL) trial. J Am Soc Nephrol 18(5):1540–1546. doi:10.1681/ASN.2006050445
El-Bassossy HM, Shaltout HA (2015) Allopurinol alleviates hypertension and proteinuria in high fructose, high salt and high fat induced model of metabolic syndrome. Transl Res 165(5):621–630. doi:10.1016/j.trsl.2014.11.008
El-Bassossy HM, Watson ML (2015) Xanthine oxidase inhibition alleviates the cardiac complications of insulin resistance: effect on low grade inflammation and the angiotensin system. J Transl Med 13(1):82. doi:10.1186/s12967-015-0445-9
Gauldie J, Bonniaud P, Sime P, Ask K, Kolb M (2007) TGF-beta, Smad3 and the process of progressive fibrosis. Biochem Soc Trans 35(Pt 4):661–664. doi:10.1042/BST0350661
Gorin Y, Block K, Hernandez J, Bhandari B, Wagner B, Barnes JL, Abboud HE (2005) Nox4 NAD(P)H oxidase mediates hypertrophy and fibronectin expression in the diabetic kidney. J Biol Chem 280(47):39616–39626. doi:10.1074/jbc.M502412200
Ha HC, Hester LD, Snyder SH (2002) Poly(ADP-ribose) polymerase-1 dependence of stress-induced transcription factors and associated gene expression in glia. Proc Natl Acad Sci U S A 99(5):3270–3275. doi:10.1073/pnas.052712399
Heilig CW, Brosius FC 3rd, Cunningham C (2006) Role for GLUT1 in diabetic glomerulosclerosis. Expert Rev Mol Med 8(4):1–18. doi:10.1017/S1462399406010490
Herceg Z, Wang ZQ (2001) Functions of poly(ADP-ribose) polymerase (PARP) in DNA repair, genomic integrity and cell death. Mutat Res 477(1–2):97–110. doi:10.1016/S0027-5107(01)00111-7
Hirata K, Shikata K, Matsuda M, Akiyama K, Sugimoto H, Kushiro M, Makino H (1998) Increased expression of selectins in kidneys of patients with diabetic nephropathy. Diabetologia 41(2):185–192. doi:10.1007/s001250050888
Khan TA, Ruel M, Bianchi C, Voisine P, Komjáti K, Szabo C, Sellke FW (2003) Poly(ADP-ribose) polymerase inhibition improves postischemic myocardial function after cardioplegia-cardiopulmonary bypass. J Am Coll Surg 197(2):270–277. doi:10.1016/S1072-7515(03)00538-6
Khanra R, Dewanjee S, D TK, Sahu R, Gangopadhyay M, De Feo V, Zia-Ul-Haq M (2015) Abroma Augusta L. (Malvaceae) leaf extract attenuates diabetes induced nephropathy and cardiomyopathy via inhibition of oxidative stress and inflammatory response. J Transl Med 13(1):6. doi:10.1186/s12967-014-0364-1
McCluskey JD, Sava D, Harbison SC, Muro-Cacho CA, Giffe JT, Ping X, Harbison RD (2011) Hepatoprotective effects of select water-soluble PARP inhibitors in a carbon tetrachloride model. Int J Crit Illn Inj Sci 1(2):97–103. doi:10.4103/2229-5151.84788
Minchenko AG, Stevens MJ, White L, Abatan OI, Komjati K, Pacher P, Szabó C, Obrosova IG (2003) Diabetes-induced overexpression of endothelin-1 and endothelin receptors in the rat renal cortex is mediated via poly(ADP-ribose) polymerase activation. FASEB J 17(11):1514–1516. doi:10.1096/fj.03-0013fje
Oeckinghaus A, Ghosh S (2009) The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol 1(4):a000034. doi:10.1101/cshperspect.a000034
Peralta-Leal A, Rodríguez-Vargas JM, Aguilar-Quesada R, Rodríguez MI, Linares JL, de Almodóvar MR, Oliver FJ (2009) PARP inhibitors: new partners in the therapy of cancer and inflammatory diseases. Free Radic Biol Med 47(1):13–26. doi:10.1016/j.freeradbiomed.2009.04.008
Ray SD, Balasubramanian G, Bagchi D, Reddy CS (2001) Ca(2+)-calmodulin antagonist chlorpromazine and poly(ADP-ribose) polymerase modulators 4-aminobenzamide and nicotinamide influence hepatic expression of BCL-XL and P53 and protect against acetaminophen-induced programmed and unprogrammed cell death in mice. Free Radic Biol Med 31(3):277–291. doi:10.1016/S0891-5849(01)00562-7
Reusch JE (2003) Diabetes, microvascular complications, and cardiovascular complications: what is it about glucose? J Clin Invest 112(7):986–988. doi:10.1172/JCI19902
Shevalye H, Maksimchyk Y, Watcho P, Obrosova IG (2010a) Poly(ADP-ribose) polymerase-1 (PARP-1) gene deficiency alleviates diabetic kidney disease. Biochim Biophys Acta 1802(11):1020–1027. doi:10.1016/j.bbadis.2010.07.004
Shevalye H, Stavniichuk R, Xu W, Zhang J, Lupachyk S, Maksimchyk Y, Drel VR, Floyd EZ, Slusher B, Obrosova IG (2010b) Poly(ADP-ribose) polymerase (PARP) inhibition counteracts multiple manifestations of kidney disease in long-term streptozotocin-diabetic rat model. Biochem Pharmacol 79(7):1007–1014. doi:10.1016/j.bcp.2009.11.018
Srinivasan K, Ramarao P (2007) Animal models in type 2 diabetes research: an overview. Indian J Med Res 125(3):451–472
Sun SF, Zhao TT, Zhang HJ, Huang XR, Zhang WK, Zhang L, Yan MH, Dong X, Wang H, Wen YM, Pan XP, Lan HY, Li P (2015) Renoprotective effect of berberine on type 2 diabetic nephropathy in rats. Clin Exp Pharmacol Physiol 42(6):662–670. doi:10.1111/1440-1681.12402
Szabo C, Biser A, Benko R, Böttinger E, Suszták K (2006a) Poly(ADP-ribose) polymerase inhibitors ameliorate nephropathy of type 2 diabetic Leprdb/db mice. Diabetes 55(11):3004–3012. doi:10.2337/db06-0147
Szabo C, Pacher P, Swanson RA (2006b) Novel modulators of poly(ADP-ribose) polymerase. Trends Pharmacol Sci 27(12):626–630. doi:10.1016/j.tips.2006.10.003
Tas Hekimoglu A, Toprak G, Akkoc H, Evliyaoglu O, Tas T, Kelle I, Colpan L (2014) Protective effect of 3-aminobenzamide, an inhibitor of poly (ADP-ribose) polymerase in distant liver injury induced by renal ischemia-reperfusion in rats. Eur Rev Med Pharmacol Sci 18(1):34–38
Theiss AL, Simmons JG, Jobin C, Lund PK (2005) Tumor necrosis factor (TNF) alpha increases collagen accumulation and proliferation in intestinal myofibroblasts via TNF receptor 2. J Biol Chem 280(43):36099–36109. doi:10.1074/jbc.M505291200
Thiemermann C, Bowes J, Myint FP, Vane JR (1997) Inhibition of the activity of poly(ADP ribose) synthetase reduces ischemia-reperfusion injury in the heart and skeletal muscle. Proc Natl Acad Sci U S A 94(2):679–683
Werth S, Lehnert H, Steinhoff J (2015) Diabetic nephropathy: current diagnostics and treatment. Internist (Berl) 56(5):513–519. doi:10.1007/s00108-014-3629-0
Wolf G, Ziyadeh FN (1999) Molecular mechanisms of diabetic renal hypertrophy. Kidney Int 56(2):393–405. doi:10.1046/j.1523-1755.1999.00590.x
Xu B, Chiu J, Feng B, Chen S, Chakrabarti S (2008) PARP activation and the alteration of vasoactive factors and extracellular matrix protein in retina and kidney in diabetes. Diabetes Metab Res Rev 24(5):404–412. doi:10.1002/dmrr.842
Ye DZ, Tai MH, Linning KD, Szabo C, Olson LK (2006) MafA expression and insulin promoter activity are induced by nicotinamide and related compounds in INS-1 pancreatic beta-cells. Diabetes 55(3):742–750. doi:10.2337/diabetes.55.03.06.db05-0653
Yuan F, Liu YH, Liu FY, Peng YM, Tian JW (2014) Intraperitoneal administration of the globular adiponectin gene ameliorates diabetic nephropathy in Wistar rats. Mol Med Rep 9(6):2293–3000. doi:10.3892/mmr.2014.2133
Zhu Y, Usui HK, Sharma K (2007) Regulation of transforming growth factor beta in diabetic nephropathy: implications for treatment. Semin Nephrol 27(2):153–160. doi:10.1016/j.semnephrol.2007.01.008
Acknowledgments
This work was funded by a research fund from the Egyptian Ministry for Higher Education Funding Agency; Science and Technology Development (STDF, www.stdf.org.eg), Fund ID 1024.
Authors’ contributions statement
EMZ carried out laboratory experiments, animal handling procedures, data and statistical analysis, collected field data, and participated in drafting the manuscript; HME-B raised the idea, designed the experiments, and participated in statistical analysis and revising the manuscript; NNE-M participated in the study conception, design, and coordination; AAA carried out histological examination; and AFA participated in study coordination. All authors gave final approval for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded by a research fund from the Egyptian Ministry for Higher Education Funding Agency; Science and Technology Development (STDF, www.stdf.org.eg), Fund ID 1024.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All experimental procedures were performed in accordance with the guidelines of the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023, revised 1978).
Rights and permissions
About this article
Cite this article
Zakaria, E.M., El-Maraghy, N.N., Ahmed, A.F. et al. PARP inhibition ameliorates nephropathy in an animal model of type 2 diabetes: focus on oxidative stress, inflammation, and fibrosis. Naunyn-Schmiedeberg's Arch Pharmacol 390, 621–631 (2017). https://doi.org/10.1007/s00210-017-1360-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-017-1360-9