Abstract
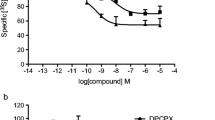
9-Ethyladenine was used as the basis for a series of non-xanthine adenosine receptor antagonists at human adenosine receptors. The adenine-based compounds were substituted in 2- or 8-position with a variety of side chains including some aryl or arylalkynyl groups previously tested as 2-substituents in adenosine and 5'-N-ethylcarboxamidoadenosine (NECA) for their effect on agonist affinity. The affinity of the novel compounds was tested in radioligand binding assays (A1, A2A and A3) and inhibition of NECA-stimulated adenylyl cyclase activity (A2B) in membranes prepared from CHO cells stably transfected with the respective human receptor subtype. High affinity antagonists were identified for A1 (9-ethyl-8-phenyl-9H-adenine, compound 2; 6-(1-butylamino)-9-ethyl-8-phenyl-9H-purine, compound 3), A2A (8-ethoxy-9-ethyladenine; compound 8) and A3 (9-ethyl-8-phenylethynyl-9H-adenine, compound 5) with selectivities versus other receptor subtypes in the range of 10 to 600. These results demonstrate that adenine is a useful template for further development of high-affinity antagonists with distinct receptor selectivity profiles.


Similar content being viewed by others
References
Baraldi PG, Cacciari B, Romagnoli R, Spalluto G, Klotz K-N, Leung E, Varani K, Gessi S, Merighi S, Borea PA (1999) Pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]-pyrimidine derivatives as highly potent and selective human A3 adenosine receptor antagonists. J Med Chem 42:4473–4478
Baraldi PG, Cacciari B, Romagnoli R, Spalluto G, Moro S, Klotz K-N, Leung E, Varani K, Gessi S, Merighi S, Borea PA (2000) Pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]-pyrimidine derivatives as highly potent and selective human A3 adenosine receptor antagonists: Influence of the chain at the N8 pyrazole nitrogen. J Med Chem 43:4768–4780
Camaioni E, Costanzi S, Vittori S, Volpini R, Klotz K-N, Cristalli G (1998) New substituted 9-alkylpurines as adenosine receptor ligands. Bioorg Med Chem 6:523–533
Cheng Y-C, Prusoff WH (1973) Relationship between the inhibition constant (KI) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108
De Lean A, Hancock AA, Lefkowitz RJ (1982) Validation and statistical analysis of a computer modeling method for quantitative analysis of radioligand binding data for mixtures of pharmacological receptor subtypes. Mol Pharmacol 21:5–16
Fredholm BB, IJzerman AP, Klotz K-N, Linden J (2001) International Union of Pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol Rev 53:1–26
Jiang J, van Rhee AM, Chang L, Patchornik A, Ji X, Evans P, Melman N, Jacobson KA (1997) Structure-activity relationships of 4-(phenylethynyl)-6-phenyl-1,4-dihydropyridines as highly selective A3 adenosine receptor antagonists. J Med Chem 40:2596–2608
Kim Y-C, Ji X, Jacobson KA (1996) Derivatives of the triazoloquinazoline adenosine antagonist (CGS15943) are selective for the human A3 receptor subtype. J Med Chem 39:4142–4148
Klotz K-N (2000) Adenosine receptors and their ligands. Naunyn-Schmiedebergs Arch Pharmacol 362:382–391
Klotz K-N, Hessling J, Hegler J, Owman C, Kull B, Fredholm BB, Lohse MJ (1998) Comparative pharmacology of human adenosine receptor subtypes—characterization of stably transfected receptors in CHO cells. Naunyn-Schmiedebergs Arch Pharmacol 357:1–9
Klotz K-N, Camaioni E, Volpini R, Kachler S, Vittori S, Cristalli G (1999) 2-Substituted N-ethylcarboxamidoadenosine derivatives as high-affinity agonists at human A3 adenosine receptors. Naunyn-Schmiedebergs Arch Pharmacol 360:103–108
Lohse MJ, Klotz K-N, Diekmann E, Friedrich K, Schwabe U (1988) 2',3'-Dideoxy-N6-cyclohexyladenosine: an adenosine derivative with antagonist properties at adenosine receptors. Eur J Pharmacol 156:157–160
Martin PL, Ueeda M, Olsson RA (1993) 2-Phenylethoxy-9-methyladenine: an adenosine receptor antagonist that discriminates between A2 adenosine receptors in the aorta and the coronary vessels from the guinea pig. J Pharmacol Exp Ther 265:248–253
Müller CE (2000) Adenosine receptor ligands—recent developments. I. Agonists. Curr Med Chem 7:1269–1288
Müller CE, Stein B (1996) Adenosine receptor antagonists: structure and potential therapeutic applications. Curr Pharm Des 2:501–530
Poulsen S-A, Quinn RJ (1998) Adenosine receptors: new opportunities for future drugs. Bioorg Med Chem 6:619–641
Shryock JC, Travagli HC, Belardinelli L (1992) Evaluation of N-0861, (±)-N 6-endonorbornan-2-yl-9-methyladenine, as an A1 subtype-selective adenosine receptor antagonist in the guinea pig isolated heart. J Pharmacol Exp Ther 260:1292–1299
Thompson RD, Secuda S, Daly JW, Olsson RA (1991) N6,9-Disubstituted adenines: potent, selective antagonists at the A1 adenosine receptor. J Med Chem 34:2877–2882
Van Calenbergh S, von Frijtag Drabbe Künzel J, Blaton NM, Peeters OM, Rozenski J, van Aerschot A, De Bruyn A, De Keukeleire D, IJzerman AP, Herdewijn P (1997) N 6-Cyclopentyl-3'-substituted-xylofuranosyladenosines: a new class of non-xanthine adenosine A1 receptor antagonists. J Med Chem 40:3765–3772
Van der Wenden EM, von Frijtag Drabbe Künzel J, Mathot RA, Danhof M, IJzerman AP, Soudijn W (1995) Ribose-modified adenosine analogues as potential partial agonists for the adenosine receptor. J Med Chem 38:4000–4006
Vittori S, Lorenzen A, Stannek C, Costanzi S, Volpini R, Ijzerman AP, von Frijtag Drabbe Künzel J, Cristalli G (2000) N-Cycloalkyl derivatives of adenosine and 1-deazaadenosine as agonists and partial agonists of the A1 adenosine receptor. J Med Chem 43:250–260
Volpini R, Costanzi S, Lambertucci C, Taffi S, Vittori S, Klotz K-N, Cristalli G (2002) N6-alkyl-2-alkynyl derivatives of adenosine as potent and selective agonists at the human adenosine A3 receptor and a starting point for searching A2B ligands. J Med Chem 45:3271–3279
Williams M, Francis J, Ghai G, Braunwalder A, Psychoyos S, Stone GA, Cash WD (1987) Biochemical characterization of the triazoloquinazoline, CGS 15943, a novel, non-xanthine adenosine antagonist. J Pharmacol Exp Ther 241:415–420
Zocchi C, Ongini E, Conti A, Monopoli A, Negretti A, Baraldi PG, Dionisotti S (1996) The non-xanthine heterocyclic compound SCH 58261 is a new potent and selective A2a adenosine receptor antagonist. J Pharmacol Exp Ther 276:398–404
Acknowledgement
This study was supported by the BIOMED 2 program EURCAR (BMH4-CT98-3474).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klotz, KN., Kachler, S., Lambertucci, C. et al. 9-Ethyladenine derivatives as adenosine receptor antagonists: 2- and 8-substitution results in distinct selectivities. Naunyn-Schmiedeberg's Arch Pharmacol 367, 629–634 (2003). https://doi.org/10.1007/s00210-003-0749-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-003-0749-9