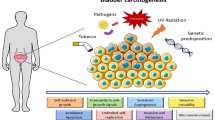
Abstract
Arsenic is highly toxic to the human bladder. In the present study, we established a human bladder epithelial cell line that closely mimics normal human bladder epithelial cells by immortalizing primary uroplakin 1B-positive human bladder epithelial cells with human telomerase reverse transcriptase (HBladEC-T). The uroplakin 1B-positive human bladder epithelial cell line was then used to evaluate the toxicity of seven arsenicals (iAsV, iAsIII, MMAV, MMAIII, DMAV, DMAIII, and DMMTAV). The cellular uptake and metabolism of each arsenical was different. Trivalent arsenicals and DMMTAV exhibited higher cellular uptake than pentavalent arsenicals. Except for MMAV, arsenicals were transported into cells by aquaglyceroporin 9 (AQP9). In addition to AQP9, DMAIII and DMMTAV were also taken up by glucose transporter 5. Microarray analysis demonstrated that arsenical treatment commonly activated the NRF2-mediated oxidative stress response pathway. ROS production increased with all arsenicals, except for MMAV. The activating transcription factor 3 (ATF3) was commonly upregulated in response to oxidative stress in HBladEC-T cells: ATF3 is an important regulator of necroptosis, which is crucial in arsenical-induced bladder carcinogenesis. Inorganic arsenics induced apoptosis while MMAV and DMAIII induced necroptosis. MMAIII, DMAV, and DMMTAV induced both cell death pathways. In summary, MMAIII exhibited the strongest cytotoxicity, followed by DMMTAV, iAsIII, DMAIII, iAsV, DMAV, and MMAV. The cytotoxicity of the tested arsenicals on HBladEC-T cells correlated with their cellular uptake and ROS generation. The ROS/NRF2/ATF3/CHOP signaling pathway emerged as a common mechanism mediating the cytotoxicity and carcinogenicity of arsenicals in HBladEC-T cells.




Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Arnold LL, Eldan M, van Gemert M, Capen CC, Cohen SM (2003) Chronic studies evaluating the carcinogenicity of monomethylarsonic acid in rats and mice. Toxicology 190(3):197–219. https://doi.org/10.1016/s0300-483x(03)00165-3
Bi Z, Fu Y, Wadgaonkar P et al (2021) New discoveries and ambiguities of Nrf2 and ATF3 signaling in environmental arsenic-induced carcinogenesis. Antioxidants (basel) 11(1):77. https://doi.org/10.3390/antiox11010077
Calatayud M, Barrios JA, Vélez D, Devesa V (2012) In vitro study of transporters involved in intestinal absorption of inorganic arsenic. Chem Res Toxicol 25(2):446–453. https://doi.org/10.1021/tx200491f
Chan FK-M, Luz NF, Moriwaki K (2015) Programmed necrosis in the cross talk of cell death and inflammation. Annu Rev Immunol 33(1):79–106. https://doi.org/10.1146/annurev-immunol-032414-112248
Clewell HJ, Thomas RS, Kenyon EM et al (2011) Concentration- and time-dependent genomic changes in the mouse urinary bladder following exposure to arsenate in drinking water for up to 12 weeks. Toxicol Sci 123(2):421–432. https://doi.org/10.1093/toxsci/kfr199
Cohen SM, Arnold LL, Eldan M, Lewis AS, Beck BD (2006) Methylated arsenicals: the implications of metabolism and carcinogenicity studies in rodents to human risk assessment. Crit Rev Toxicol 36(2):99–133. https://doi.org/10.1080/10408440500534230
Cohen SM, Ohnishi T, Arnold LL, Le XC (2007) Arsenic-induced bladder cancer in an animal model. Toxicol Appl Pharmacol 222(3):258–263. https://doi.org/10.1016/j.taap.2006.10.010
Cohen SM, Arnold LL, Beck BD, Lewis AS, Eldan M (2013) Evaluation of the carcinogenicity of inorganic arsenic. Crit Rev Toxicol 43(9):711–752. https://doi.org/10.3109/10408444.2013.827152
Cohen SM, Arnold LL, Tsuji JS (2019) Inorganic arsenic: a nongenotoxic threshold carcinogen. Curr Opin Toxicol 14:8–13. https://doi.org/10.1016/j.cotox.2019.05.002
Córdova EJ, Martínez-Hernández A, Uribe-Figueroa L et al (2014) The NRF2-KEAP1 pathway is an early responsive gene network in arsenic exposed lymphoblastoid cells. PLoS ONE 9(2):e88069. https://doi.org/10.1371/journal.pone.0088069
David CE (2015) Arsenic, reactive oxygen, and endothelial dysfunction. J Pharmacol Exp Ther 353(3):458. https://doi.org/10.1124/jpet.115.223289
Dhuriya YK, Sharma D (2018) Necroptosis: a regulated inflammatory mode of cell death. J Neuroinflammation 15(1):199. https://doi.org/10.1186/s12974-018-1235-0
Dodmane PR, Arnold LL, Kakiuchi-Kiyota S et al (2013) Cytotoxicity and gene expression changes induced by inorganic and organic trivalent arsenicals in human cells. Toxicology 312:18–29. https://doi.org/10.1016/j.tox.2013.07.008
Dopp E, von Recklinghausen U, Diaz-Bone R, Hirner AV, Rettenmeier AW (2010) Cellular uptake, subcellular distribution and toxicity of arsenic compounds in methylating and non-methylating cells. Environ Res 110(5):435–442. https://doi.org/10.1016/j.envres.2009.08.012
El-Ghiaty MA, El-Kadi AOS (2021) Arsenic: various species with different effects on cytochrome P450 regulation in humans. EXCLI J 20:1184–1242. https://doi.org/10.17179/excli2021-3890
Fan C, Liu G, Long Y, Rosen B, Cai Y (2018) Thiolation in arsenic metabolism: a chemical perspective. Metallomics 10(10):1368–1382. https://doi.org/10.1039/C8MT00231B
Hu H, Tian M, Ding C, Yu S (2018) The C/EBP homologous protein (CHOP) transcription factor functions in endoplasmic reticulum stress-induced apoptosis and microbial infection. Front Immunol 9:3083. https://doi.org/10.3389/fimmu.2018.03083
Hu Y, Li J, Lou B et al (2020) The role of reactive oxygen species in arsenic toxicity. Biomolecules 10(2):240. https://doi.org/10.3390/biom10020240
IARC (2012) Arsenic, metals, fibres and dusts. Lyon (FR): International Agency for Research on Cancer (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 100C.)
Inagawa Y, Yamada K, Yugawa T et al (2014) A human cancer xenograft model utilizing normal pancreatic duct epithelial cells conditionally transformed with defined oncogenes. Carcinogenesis 35(8):1840–1846. https://doi.org/10.1093/carcin/bgu112
Kim KH, Jeong JY, Surh YJ, Kim KW (2010) Expression of stress-response ATF3 is mediated by Nrf2 in astrocytes. Nucleic Acids Res 38(1):48–59. https://doi.org/10.1093/nar/gkp865
Liu Z, Carbrey JM, Agre P, Rosen BP (2004) Arsenic trioxide uptake by human and rat aquaglyceroporins. Biochem Biophys Res Commun 316(4):1178–1185. https://doi.org/10.1016/j.bbrc.2004.03.003
Liu Z, Sanchez MA, Jiang X, Boles E, Landfear SM, Rosen BP (2006a) Mammalian glucose permease GLUT1 facilitates transport of arsenic trioxide and methylarsonous acid. Biochem Biophys Res Commun 351(2):424–430. https://doi.org/10.1016/j.bbrc.2006.10.054
Liu Z, Styblo M, Rosen Barry P (2006b) Methylarsonous acid transport by aquaglyceroporins. Environ Health Perspect 114(4):527–531. https://doi.org/10.1289/ehp.8600
Liu X, Ory V, Chapman S et al (2012) ROCK inhibitor and feeder cells induce the conditional reprogramming of epithelial cells. Am J Pathol 180(2):599–607. https://doi.org/10.1016/j.ajpath.2011.10.036
Liu Z, Shi Q, Song X et al (2016) Activating transcription factor 4 (ATF4)-ATF3-C/EBP homologous protein (CHOP) cascade shows an essential role in the ER stress-induced sensitization of tetrachlorobenzoquinone-challenged PC12 cells to ROS-mediated apoptosis via death receptor 5 (DR5) signaling. Chem Res Toxicol 29(9):1510–1518. https://doi.org/10.1021/acs.chemrestox.6b00181
Ma Q (2013) Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53:401–426. https://doi.org/10.1146/annurev-pharmtox-011112-140320
Matsue T, Gi M, Shiota M et al (2022) The carbonic anhydrase inhibitor acetazolamide inhibits urinary bladder cancers via suppression of β-catenin signaling. Cancer Sci 113(8):2642–2653. https://doi.org/10.1111/cas.15467
McDermott JR, Jiang X, Beene LC, Rosen BP, Liu Z (2010) Pentavalent methylated arsenicals are substrates of human AQP9. Biometals 23(1):119–127. https://doi.org/10.1007/s10534-009-9273-9
Medda N, De SK, Maiti S (2021) Different mechanisms of arsenic related signaling in cellular proliferation, apoptosis and neo-plastic transformation. Ecotoxicol Environ Saf 208:111752. https://doi.org/10.1016/j.ecoenv.2020.111752
Moe B, Peng H, Lu X et al (2016) Comparative cytotoxicity of fourteen trivalent and pentavalent arsenic species determined using real-time cell sensing. J Environ Sci 49:113–124. https://doi.org/10.1016/j.jes.2016.10.004
Naranmandura H, Ogra Y, Iwata K et al (2009) Evidence for toxicity differences between inorganic arsenite and thioarsenicals in human bladder cancer cells. Toxicol Appl Pharmacol 238(2):133–140. https://doi.org/10.1016/j.taap.2009.05.006
Naranmandura H, Carew MW, Xu S et al (2011) Comparative toxicity of arsenic metabolites in human bladder cancer EJ-1 cells. Chem Res Toxicol 24(9):1586–1596. https://doi.org/10.1021/tx200291p
Nishiwaki M, Toyoda M, Oishi Y et al (2020) Immortalization of human hepatocytes from biliary atresia with CDK4(R24C), cyclin D1, and TERT for cytochrome P450 induction testing. Sci Rep 10(1):17503. https://doi.org/10.1038/s41598-020-73992-3
Okina M, Yoshida K, Kuroda K, Wanibuchi H, Fukushima S, Endo G (2004) Determination of trivalent methylated arsenicals in rat urine by liquid chromatography–inductively coupled plasma mass spectrometry after solvent extraction. J Chromatogr B 799(2):209–215. https://doi.org/10.1016/j.jchromb.2003.10.028
Okuno T, Gi M, Fujioka M et al (2019) Acetoaceto-o-toluidide enhances cellular proliferative activity in the urinary bladder of rats. Toxicol Sci 169(2):456–464. https://doi.org/10.1093/toxsci/kfz051
Reiswich V, Akdeniz G, Lennartz M et al (2022) Large-scale human tissue analysis identifies Uroplakin 1b as a putative diagnostic marker in surgical pathology. Hum Pathol 126:108–120. https://doi.org/10.1016/j.humpath.2022.05.002
Roggenbeck BA, Banerjee M, Leslie EM (2016) Cellular arsenic transport pathways in mammals. J Environ Sci (china) 49:38–58. https://doi.org/10.1016/j.jes.2016.10.001
Rohini M, Haritha Menon A, Selvamurugan N (2018) Role of activating transcription factor 3 and its interacting proteins under physiological and pathological conditions. Int J Biol Macromol 120:310–317. https://doi.org/10.1016/j.ijbiomac.2018.08.107
Shen J, Wanibuchi H, Waalkes MP et al (2006) A comparative study of the sub-chronic toxic effects of three organic arsenical compounds on the urothelium in F344 rats; gender-based differences in response. Toxicol Appl Pharmacol 210(3):171–180. https://doi.org/10.1016/j.taap.2005.04.018
Shi Q, Hu B, Yang C, Zhao L, Wu J, Qi N (2021) ATF3 promotes arsenic-induced apoptosis and oppositely regulates DR5 and Bcl-xL expression in human bronchial epithelial cells. Int J Mol Sci 22(8):4223. https://doi.org/10.3390/ijms22084223
Styblo M, Del Razo LM, Vega L et al (2000) Comparative toxicity of trivalent and pentavalent inorganic and methylated arsenicals in rat and human cells. Arch Toxicol 74(6):289–299. https://doi.org/10.1007/s002040000134
Sumi D, Tsurumoto M, Yoshino Y et al (2015) High accumulation of arsenic in the esophagus of mice after exposure to arsenite. Arch Toxicol 89(10):1751–1758. https://doi.org/10.1007/s00204-014-1326-3
Sun H-J, Ding S, Guan D-X, Ma LQ (2022) Nrf2/Keap1 pathway in countering arsenic-induced oxidative stress in mice after chronic exposure at environmentally-relevant concentrations. Chemosphere 303:135256. https://doi.org/10.1016/j.chemosphere.2022.135256
Suzuki S, Arnold LL, Pennington KL et al (2010) Dietary administration of sodium arsenite to rats: relations between dose and urinary concentrations of methylated and thio-metabolites and effects on the rat urinary bladder epithelium. Toxicol Appl Pharmacol 244(2):99–105. https://doi.org/10.1016/j.taap.2009.12.026
Thomas DJ (2016) The chemistry and metabolism of arsenic. In: States JC (ed) Arsenic: exposure sources, health risks, and mechanisms of toxicity. John Wiley & Sons Inc, Hoboken, New Jersey, pp 81–109
Wang L, Kou M-C, Weng C-Y, Hu L-W, Wang Y-J, Wu M-J (2012) Arsenic modulates heme oxygenase-1, interleukin-6, and vascular endothelial growth factor expression in endothelial cells: roles of ROS, NF-κB, and MAPK pathways. Arch Toxicol 86(6):879–896. https://doi.org/10.1007/s00204-012-0845-z
Wei M, Wanibuchi H, Morimura K et al (2002) Carcinogenicity of dimethylarsinic acid in male F344 rats and genetic alterations in induced urinary bladder tumors. Carcinogenesis 23(8):1387–1397. https://doi.org/10.1093/carcin/23.8.1387
Ye K, Chen Z, Xu Y (2023) The double-edged functions of necroptosis. Cell Death Dis 14(2):163. https://doi.org/10.1038/s41419-023-05691-6
Zhong L, Hao H, Chen D et al (2019) Arsenic trioxide inhibits the differentiation of fibroblasts to myofibroblasts through nuclear factor erythroid 2-like 2 (NFE2L2) protein and the Smad2/3 pathway. J Cell Physiol 234(3):2606–2617. https://doi.org/10.1002/jcp.27073
Acknowledgements
This work was supported by a grant from the Food Safety Commission, Cabinet Office, Government of Japan (Research Program for Risk Assessment Study on Food Safety, No. JPCAFSC20212102), Health and Labour Sciences Research Grants from the Ministry of Health, Labour and Welfare of Japan (No. 22KD1003), and grants from Japan Society for the Promotion of Science (No. 23K09652). The authors gratefully acknowledge the technical assistance of Rie Onodera, Keiko Sakata, Yuko Hisabayashi, and Yukiko Iura (Department of Molecular Pathology, Osaka Metropolitan University Graduate School of Medicine, Osaka, Japan), and Yoriko Yabunaka, and Yukimi Kira (Research Support Platform, Osaka Metropolitan University Graduate School of Medicine, Osaka, Japan). We would like to thank Dr. Hiroyuki Miyoshi and RIKEN Bioresource center for providing the lentivirus and packaging plasmids.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vachiraarunwong, A., Gi, M., Kiyono, T. et al. Characterizing the toxicological responses to inorganic arsenicals and their metabolites in immortalized human bladder epithelial cells. Arch Toxicol (2024). https://doi.org/10.1007/s00204-024-03750-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00204-024-03750-1