Abstract
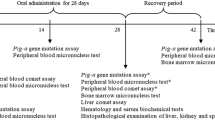
Parabens have historically served as antimicrobial preservatives in a range of consumables such as food, beverages, medications, and personal care products due to their broad-spectrum antibacterial and antifungal properties. Traditionally, these compounds were believed to exhibit low toxicity, causing minimal irritation, and possessing limited sensitization potential. However, recent evidence suggests that parabens might function as endocrine-disrupting chemicals (EDCs). Consequently, extensive research is underway to elucidate potential human health implications arising from exposure to these substances. Among these parabens, particular concerns have been raised regarding the potential adverse effects of iso-butylparaben (IBP). Studies have specifically highlighted its potential for inducing hormonal disruption, significant ocular damage, and allergic skin reactions. This study aimed to evaluate the prolonged systemic toxicity, semen quality, and estrus cycle in relation to endocrine disruption endpoints, alongside assessing the toxicokinetic behavior of IBP in Sprague–Dawley rats following a 13-week repeated subcutaneous administration. The rats were administered either the vehicle (4% Tween 80) or IBP at dosage levels of 2, 10, and 50 mg/kg/day for 13 weeks. Blood collection for toxicokinetic study was conducted on three specified days: day 1 (1st), day 30 (2nd), and day 91 (3rd). Systemic toxicity assessment and potential endocrine effects were based on various parameters including mortality rates, clinical signs, body weights, food and water consumption, ophthalmological findings, urinalysis, hematological and clinical biochemistry tests, organ weights, necropsy and histopathological findings, estrus cycle regularity, semen quality, and toxicokinetic behavior. The findings revealed that IBP induced local irritation at the injection site in males at doses ≥ 10 mg/kg/day and in females at 50 mg/kg/day; however, systemic toxicity was not observed. Consequently, the no-observed-adverse-effect level (NOAEL) for IBP was determined to be 50 mg/kg/day in rats of both sexes, indicating no impact on the endocrine system. The toxicokinetics of IBP exhibited dose-dependent systemic exposure, reaching a maximum dose of 50 mg/kg/day, and repeated administration over 13 weeks showed no signs of accumulation.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anderson JM, Rodriguez A, Chang DT (2008) Foreign body reaction to biomaterials. Semin Immunol 20:86–100
Aubert N, Ameller T, Legrand JJ (2012) Systemic exposure to parabens: pharmacokinetics, tissue distribution, excretion balance and plasma metabolites of [14C]-methyl-, propyl- and butylparaben in rats after oral, topical or subcutaneous administration. Food Chem Toxicol 50:445–454
Bae JS, Lee JD, Song SW (2021) Thirteen-week subcutaneous repeated dose toxicity study of butylparaben and its toxicokinetics in rats. Arch Toxicol 95:2037–2050
Cashman AL, Warshaw EM (2005) Parabens: a review of epidemiology, structure, allergenicity, and hormonal properties. Dermatitis 16:57–66
CIR (Cosmetic Ingredient Review) (2008) Report on the safety assessment of methylparaben, ethylparaben, propylparaben, isopropylparaben, butylparaben, isobutylparaben, and benzylparaben as used in cosmetic products. Int J Toxicol 27(Suppl 4):1–82
Danishi MOE (The Ministry of the Environment) (2011) Statutory order amending the statutory order on cosmetic products (amendment to Annex5)
Darbre PD, Byford JR, Shaw LE et al (2002) Oestrogenic activity of isobutylparaben in vitro and in vivo. J Appl Toxicol 22:219–226
Daston GP (2004) Developmental toxicity evaluation of butylparaben in Sprague–Dawley rats. Birth Defects Res B Dev Reprod Toxicol 71:296–302
Derache R, Gourdon J (1963) Metabolism of a food preservative: phydroxybenzoic acid and its esters. Food Cosmet Toxicol 1:189–195
ECHA (European Chemicals Agency) (2022) Member state committee support document for identification of isobutyl 4-hydroxybenzoate as a substance of very high concern because of its endocrine disrupting properties (article 57(f)—human health) properties. https://echa.europa.eu/documents/10162/d6ed2a8b-1e39-f908-f5f1-8cbf8fdcb40f
ECHA (European Chemicals Agency) (2023) Isobutyl 4-hydroxybenzoate—substance information. https://echa.europa.eu/substance-information/-/substanceinfo/100.022.008
Elder RL (1984) Final report on the safety assessment of methylparaben, ethylparaben, propylparaben, and butylparaben. J Am Coll Toxicol 3:147–209
EMA (European Medicines Agency) (2015) Reflection paper on the use of methyl- and propylparaben as excipients in human medicinal products for oral use. https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-use-methyl-propylparaben-excipients-human-medicinal-products-oral-use_en.pdf
Fransway AF, Fransway PJ, Belsito DV et al (2019) Paraben toxicology. Dermatitis 30:32–45
Genuis SJ, Birkholz D, Curtis L, Sandau C (2013) Paraben levels in an urban community of Western Canada. ISRN Toxicol 2013:507897
Han ZZ, Xu HD, Kim KH et al (2010) Reference data of the main physiological parameters in control Sprague–Dawley rats from pre-clinical toxicity studies. Lab Anim Res 26:153–164
Harvey PW, Everett DJ (2006) Regulation of endocrine-disrupting chemicals: critical overview and deficiencies in toxicology and risk assessment for human health. Best Pract Res Clin Endocrinol Metab 20:145–165
Health Canada (2022) Cosmetic ingredient hotlis. https://www.canada.ca/en/health-canada/services/consumer-product-safety/cosmetics/cosmetic-ingredient-hotlist-prohibited-restricted-ingredients/hotlist.html#t1p
Hu Y, Zhang Z, Sun L et al (2013) The estrogenic effects of benzylparaben at low doses based on uterotrophic assay in immature SD rats. Food Chem Toxicol 53:69–74
Karpuzoglu E, Holladay SD, Gogal RM Jr (2013) Parabens: potential impact of low-affinity estrogen receptor binding chemicals on human health. J Toxicol Environ Health B Crit Rev 16:321–335
Katzin WE, Centeno JA, Feng LJ et al (2005) Pathology of lymph nodes from patients with breast implants: a histologic and spectroscopic evaluation. Am J Surg Pathol 29:506–511
Kawaguchi M, Morohoshi K, Imai H et al (2010) Maternal isobutyl-paraben exposure decreased the plasma corticosterone level in dams and sensitivity to estrogen in female offspring rats. Exp Anim 59:631–635
Kim PG (2014) Toxicokinetic modeling of ethyl paraben administered orally in rats. J Environ Health Sci 40:407–412
Kim MJ, Kwack SJ, Lim SK, Kim YJ, Roh TH, Choi SM, Kim HS, Lee BM (2015) Toxicological evaluation of isopropylparaben and isobutylparaben mixture in Sprague–Dawley rats following 28 days of dermal exposure. Regul Toxicol Pharmacol 73:544–551
Koda T, Umezu T, Kamata R et al (2005) Uterotrophic effects of benzophenone derivatives and a p-hydroxybenzoate used in ultraviolet screens. Environ Res 98:40–45
Krogenæs AK, Ropstad E, Gutleb AC et al (2014) In utero exposure to environmentally relevant concentrations of PCB 153 and PCB 118 disrupts fetal testis development in sheep. J Toxicol Environ Health A 77:628–649
MFDS (The Ministry of Food and Drug Safety) (2013) The regulation on safety standards and others of cosmetics. MFDS Notifcation No. 2013-2 (in Korean) Accessed at https://www.mfds.go.kr/brd/m_207/view.do?seq=6139&srchFr=&srchTo=&srchWord=&srchTp=&itm_seq_1=0&itm_seq_2=0&multi_itm_seq=0&company_cd=&company_nm=&page=107
MFDS (The Ministry of Food and Drug Safety) (2015) Toxicity test standards for medicines. MFDS Notifcation No. 2015-82 (in Korean) Accessed at https://mfds.go.kr/brd/m_211/view.do?seq=10128&srchFr=&srchTo=&srchWord=&srchTp=&itm_seq_1=0&itm_seq_2=0&multi_itm_seq=0&company_cd=&company_nm=&page=51
Moos RK, Koch HM, Angerer J, Apel P, Schröter-Kermani C, Brüning T, Kolossa-Gehring M (2015) Parabens in 24 h urine samples of the German Environmental Specimen Bank from 1995 to 2012. Int J Hyg Environ Health 218:666–674
Moriyama I, Hiraoka K, Yamaguchi R (1975) Teratogenic effects of food additive ethyl-p-hydroxybenzoate studied in pregnant rats. Acta Obstet Gynaecol Jpn 22:94–106
OECD (Organization for Economic Cooperation and Development) (1998) Test Guideline No. 408 ‘Repeated Dose 90-Day Oral Toxicity Study in Rodents’. Available from: https://www.oecd-ilibrary.org/docserver/9789264070707-en.pdf?expires=1712538848&id=id&accname=guest&checksum=701A0C5312FF8675198F08C71E412A18
OECD (Organization for Economic Cooperation and Development) (2010) OECD Test Guideline No. 417 ‘Toxicokinetics’. https://www.oecd-ilibrary.org/docserver/9789264070882-en.pdf?expires=1700751516&id=id&accname=guest&checksum=248E31F35DEE952DE1DC702D3E61AFAF
RIVM (National Institute for Public Health and the Environment) (2018) Review on butylparaben: exposure, toxicity and risk assessment: with a focus on endocrine disrupting properties and cumulative risk assessment. https://www.rivm.nl/bibliotheek/rapporten/2018-0161.pdf
Sakazaki F, Ueno H, Nagase H, Nakamuro K (2009) p-hydroxybenzoate esters enhance mouse contact hypersensitivity. J Health Sci 55:560–566
Satoh K, Nagai F, Aoki N, Nishijima M (2000) Competitive binding of some alkyl p-hydroxybenzoates to human estrogen receptor alpha and beta. Yakugaku Zasshi 120:1429–1433
SCCS (Scientific Committee on Consumer Safety) (2010) Opinion on parabens. SCCS/1348/10. https://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_041.pdf
SCCS (Scientific Committee on Consumer Safety) (2013) Opinion on parabens, updated request on propyl- and butylparaben. SCCS/1514/13. https://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_132.pdf
Shin MY, Shin C, Choi JW et al (2019) Pharmacokinetic profile of propyl paraben in humans after oral administration. Environ Int 130:104917
Shin MY, Choi JW, Lee S (2023) Pharmacokinetics of transdermal methyl-, ethyl-, and propylparaben in humans following single dermal administration. Chemosphere 310:136689
Soni MG, Carabin IG, Burdock GA (2005) Safety assessment of esters of p-hydroxybenzoic acid (parabens). Food Chem Toxicol 43:985–1015
Sternheimer R, Malbin B (1951) Clinical recognition of pyelonephritis, with a new stain for urinary sediments. Am J Med 11(3): 312–323
Tzortzatou F, Hayhoe FG (1974) The effect of folate antagonists on dihydrofolate reductase activity demonstrated cytochemically. Br J Haematol 28:209–216
US FDA (Food and drug administration) (2010) Selected cosmetic ingredients-ingredients prohibited & restricted by FDA regulations
Vo TT, Jeung EB (2009) An evaluation of estrogenic activity of parabens using uterine calbindin-d9k gene in an immature rat model. Toxicol Sci 112:68–77
Ye X, Bishop AM, Reidy JA et al (2006) Parabens as urinary biomarkers of exposure in humans. Environ Health Perspect 114:1843–1846
Yoon K, Kwack SJ, Kim HS et al (2014) Estrogenic endocrine-disrupting chemicals: molecular mechanisms of actions on putative human diseases. J Toxicol Environ Health B Crit Rev 17:127–174
Acknowledgements
This work was supported by grants (14172MFDS975 and 19172MFDS221) from the Ministry of Food and Drug Safety, Korea in 2014 and 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, J.D., Bae, JS., Kim, H.Y. et al. Repeated-dose toxicity and toxicokinetic study of isobutylparaben in rats subcutaneously treated for 13 weeks. Arch Toxicol (2024). https://doi.org/10.1007/s00204-024-03741-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00204-024-03741-2