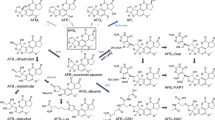
Abstract
Large interspecies differences between rats and mice concerning the hepatotoxicity and carcinogenicity of aflatoxin B1 (AFB1) are known, with mice being more resistant. However, a comprehensive interspecies comparison including subcellular liver tissue compartments has not yet been performed. In this study, we performed spatio-temporal intravital analysis of AFB1 kinetics in the livers of anesthetized mice and rats. This was supported by time-dependent analysis of the parent compound as well as metabolites and adducts in blood, urine, and bile of both species by HPLC–MS/MS. The integrated data from intravital imaging and HPLC–MS/MS analysis revealed major interspecies differences between rats and mice: (1) AFB1-associated fluorescence persisted much longer in the nuclei of rat than mouse hepatocytes; (2) in the sinusoidal blood, AFB1-associated fluorescence was rapidly cleared in mice, while a time-dependent increase was observed in rats in the first three hours after injection followed by a plateau that lasted until the end of the observation period of six hours; (3) this coincided with a far stronger increase of AFB1-lysine adducts in the blood of rats compared to mice; (4) the AFB1-guanine adduct was detected at much higher concentrations in bile and urine of rats than mice. In both species, the AFB1-glutathione conjugate was efficiently excreted via bile, where it reached concentrations at least three orders of magnitude higher compared to blood. In conclusion, major differences between mice and rats were observed, concerning the nuclear persistence, formation of AFB1-lysine adducts, and the AFB1-guanine adducts.






Similar content being viewed by others
Data availability
All data are already provided in the figure.
References
Buetler TM, Eaton DL (1992) Complementary DNA cloning, messenger RNA expression, and induction of alpha-class glutathione S-transferases in mouse tissues. Can Res 52(2):314–318
Cole KE, Jones TW, Lipsky MM, Trump BF, Hsu IC (1988) In vitro binding of aflatoxin B1 and 2-acetylaminofluorene to rat, mouse and human hepatocyte DNA: the relationship of DNA binding to carcinogenicity. Carcinogenesis 9(5):711–716. https://doi.org/10.1093/carcin/9.5.711
Custodio RJP, Hobloss Z, Myllys M et al (2023) Cognitive functions, neurotransmitter alterations, and hippocampal microstructural changes in mice caused by feeding on western diet. Cells 12(18):2331. https://doi.org/10.3390/cells12182331
Degen GH, Neumann HG (1978) The major metabolite of aflatoxin B1 in the rat is a glutathione conjugate. Chem Biol Interact 22(2–3):239–255. https://doi.org/10.1016/0009-2797(78)90129-1
Degen GH, Neumann HG (1981) Differences in aflatoxin B1-susceptibility of rat and mouse are correlated with the capability in vitro to inactivate aflatoxin B1-epoxide. Carcinogenesis 2(4):299–306. https://doi.org/10.1093/carcin/2.4.299
Eaton DL, Gallagher EP (1994) Mechanisms of aflatoxin carcinogenesis. Annu Rev Pharmacol Toxicol 34:135–172. https://doi.org/10.1146/annurev.pa.34.040194.001031
Essigmann JM, Croy RG, Bennett RA, Wogan GN (1982) Metabolic activation of aflatoxin B1: patterns of DNA adduct formation, removal, and excretion in relation to carcinogenesis. Drug Metab Rev 13(4):581–602. https://doi.org/10.3109/03602538209011088
Gerdemann A, Cramer B, Degen GH et al (2023) Comparative metabolism of aflatoxin B(1) in mouse, rat and human primary hepatocytes using HPLC-MS/MS. Arch Toxicol 97(12):3179–3196. https://doi.org/10.1007/s00204-023-03607-z
Ghallab A, Myllys M, Holland CH et al (2019) Influence of liver fibrosis on lobular zonation. Cells 8(12):1556–1556. https://doi.org/10.3390/CELLS8121556
Ghallab A, Hassan R, Myllys M et al (2021a) Subcellular spatio-temporal intravital kinetics of aflatoxin B(1) and ochratoxin A in liver and kidney. Arch Toxicol 95(6):2163–2177. https://doi.org/10.1007/s00204-021-03073-5
Ghallab A, Myllys M, Friebel A et al (2021b) Spatio-temporal multiscale analysis of western diet-fed mice reveals a translationally relevant sequence of events during NAFLD progression. Cells 10(10):2516
Ghallab A, Hassan R, Hofmann U et al (2022) Interruption of bile acid uptake by hepatocytes after acetaminophen overdose ameliorates hepatotoxicity. J Hepatol 77(1):71–83. https://doi.org/10.1016/j.jhep.2022.01.020
Ghallab A, Gonzalez D, Strangberg E et al (2024) Inhibition of the renal apical sodium dependent bile acid transporter prevents cholemic nephropathy in mice with obstructive cholestasis. J Hepatol 80(2):268–281. https://doi.org/10.1016/j.jhep.2023.10.035
Gianmoena K, Gasparoni N, Jashari A et al (2021) Epigenomic and transcriptional profiling identifies impaired glyoxylate detoxification in NAFLD as a risk factor for hyperoxaluria. Cell Rep 36(8):109526. https://doi.org/10.1016/j.celrep.2021.109526
Green CE, Rice DW, Hsieh DP, Byard JL (1982) The comparative metabolism and toxic potency of aflatoxin B1 and aflatoxin M1 in primary cultures of adult-rat hepatocytes. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc 20(1):53–60. https://doi.org/10.1016/s0278-6915(82)80009-4
Guengerich FP, Johnson WW, Shimada T, Ueng YF, Yamazaki H, Langouët S (1998) Activation and detoxication of aflatoxin B1. Mutat Res 402(1–2):121–128. https://doi.org/10.1016/s0027-5107(97)00289-3
Hanigan HM, Laishes BA (1984) Toxicity of aflatoxin B1 in rat and mouse hepatocytes in vivo and in vitro. Toxicology 30(3):185–193. https://doi.org/10.1016/0300-483x(84)90090-8
Hassan R (2016) Possibilities and limitations of intravital imaging. Excli j 15:872–874. https://doi.org/10.17179/excli2016-863
Hassan R, Friebel A, Brackhagen L et al (2022a) Hypoalbuminemia affects the spatio-temporal tissue distribution of ochratoxin A in liver and kidneys: consequences for organ toxicity. Arch Toxicol. https://doi.org/10.1007/s00204-022-03361-8
Hassan R, Gonzalez D, Hobloss Z et al (2022b) Inhibition of cytochrome P450 enhances the nephro- and hepatotoxicity of ochratoxin A. Arch Toxicol 96(12):3349–3361. https://doi.org/10.1007/s00204-022-03395-y
Hengstler JG, Van der Burg B, Steinberg P, Oesch F (1999) Interspecies differences in cancer susceptibility and toxicity. Drug Metab Rev 31(4):917–970. https://doi.org/10.1081/dmr-100101946
Holland CH, Ramirez Flores RO, Myllys M et al (2022) Transcriptomic cross-species analysis of chronic liver disease reveals consistent regulation between humans and mice. Hepatol Commun 6(1):161–177. https://doi.org/10.1002/hep4.1797
IARC (2015) Mycotoxin control in low-and middle-income countries IARC—international agency for research on cancer (Wild CP, Miller JD, Groopman JD, eds), 9th edn IARC, Lyon, France
Ilic Z, Crawford D, Vakharia D, Egner PA, Sell S (2010) Glutathione-S-transferase A3 knockout mice are sensitive to acute cytotoxic and genotoxic effects of aflatoxin B1. Toxicol Appl Pharmacol 242(3):241–246. https://doi.org/10.1016/j.taap.2009.10.008
Kensler TW, Roebuck BD, Wogan GN, Groopman JD (2011) Aflatoxin: a 50-year odyssey of mechanistic and translational toxicology. Toxicol Sci off J Soc Toxicol 120(Suppl 1):S28-48. https://doi.org/10.1093/toxsci/kfq283
Koeppert S, Ghallab A, Peglow S et al (2021) Live imaging of calciprotein particle clearance and receptor mediated uptake: role of calciprotein monomers. Front Cell Dev Biol 9:633925. https://doi.org/10.3389/fcell.2021.633925
Monroe DH, Eaton DL (1987) Comparative effects of butylated hydroxyanisole on hepatic in vivo DNA binding and in vitro biotransformation of aflatoxin B1 in the rat and mouse. Toxicol Appl Pharmacol 90(3):401–409. https://doi.org/10.1016/0041-008x(87)90132-3
Moss EJ, Neal GE, Judah DJ (1985) The mercapturic acid pathway metabolites of a glutathione conjugate of aflatoxin B1. Chem Biol Interact 55(1–2):139–155. https://doi.org/10.1016/s0009-2797(85)80124-1
Neal GE, Colley PJ (1979) The formation of 2,3-dihydro-2,3-dihydroxy aflatoxin B1 by the metabolism of aflatoxin B1 in vitro by rat liver microsomes. FEBS Lett 101(2):382–386. https://doi.org/10.1016/0014-5793(79)81049-2
Neal GE, Eaton DL, Judah DJ, Verma A (1998) Metabolism and toxicity of aflatoxins M1 and B1 in human-derived in vitro systems. Toxicol Appl Pharmacol 151(1):152–158. https://doi.org/10.1006/taap.1998.8440
Rushing BR, Selim MI (2019) Aflatoxin B1: a review on metabolism, toxicity, occurrence in food, occupational exposure, and detoxification methods. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc 124:81–100. https://doi.org/10.1016/j.fct.2018.11.047
Sabbioni G, Skipper PL, Büchi G, Tannenbaum SR (1987) Isolation and characterization of the major serum albumin adduct formed by aflatoxin B1 in vivo in rats. Carcinogenesis 8(6):819–824. https://doi.org/10.1093/carcin/8.6.819
Schneider KM, Candels LS, Hov JR et al (2021) Gut microbiota depletion exacerbates cholestatic liver injury via loss of FXR signalling. Nat Metab 3(9):1228–1241. https://doi.org/10.1038/s42255-021-00452-1
Vucur M, Ghallab A, Schneider AT et al (2023) Sublethal necroptosis signaling promotes inflammation and liver cancer. Immunity 56(7):1578–1595. https://doi.org/10.1016/j.immuni.2023.05.017
Wild CP, Gong YY (2010) Mycotoxins and human disease: a largely ignored global health issue. Carcinogenesis 31(1):71–82. https://doi.org/10.1093/carcin/bgp264
Wogan GN (1973) Aflatoxin carcinogenesis. In: Busch J (ed) Methods in cancer research. Academic Press, New York, pp 309–344
Wu K, Jia S, Zhang J et al (2021) Transcriptomics and flow cytometry reveals the cytotoxicity of aflatoxin B(1) and aflatoxin M(1) in bovine mammary epithelial cells. Ecotoxicol Environ Saf 209:111823. https://doi.org/10.1016/j.ecoenv.2020.111823
Yang C, Rathman JF, Ribeiro JV et al (2023) Update of the cancer potency database (CPDB) to enable derivations of thresholds of toxicological concern (TTC) for cancer potency. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc 182:114182. https://doi.org/10.1016/j.fct.2023.114182
Yourtee DM, Bean TA, Kirk-Yourtee CL (1987) Human aflatoxin B1 metabolism: an investigation of the importance of aflatoxin Q1 as a metabolite of hepatic post-mitochondrial fraction. Toxicol Lett 38(3):213–224. https://doi.org/10.1016/0378-4274(87)90002-6
Author information
Authors and Affiliations
Contributions
AG, JGH, GHD helped in study concept and design, data analysis and interpretation, drafting the manuscript; AG, RH were involved in intravital imaging, data acquisition; RH, ZH, MM, DG contributed to animal experiments, data acquisition and writing of the manuscript; WA performed toxicokinetic analysis of the data and contributed to writing of the manuscript; AF, SH contributed to image analysis and writing of the manuscript; H-UH involved in study concept and design, supervision of the HPLC–MS/MS analysis, data analysis and interpretation and contributed to writing of the manuscript; ME contributed to data analysis and interpretation and writing of the manuscript; JV helped in preparation of reference compounds and HPLC–MS/MS analysis; AG, BC contributed to HPLC–MS/MS analysis and data interpretation, data acquisition and writing of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 38817 KB)
Supplementary file3 (M4V 91980 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hassan, R., Gerdemann, A., Cramer, B. et al. Integrated data from intravital imaging and HPLC–MS/MS analysis reveal large interspecies differences in AFB1 metabolism in mice and rats. Arch Toxicol 98, 1081–1093 (2024). https://doi.org/10.1007/s00204-024-03688-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-024-03688-4