Abstract
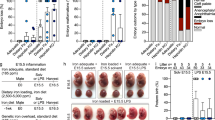
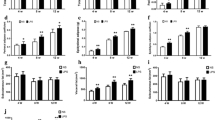
Maternal lipopolysaccharide (LPS) exposure during pregnancy has been related to IUGR. Here, we explored whether paternal LPS exposure before mating impaired fetal development. All male mice except controls were intraperitoneally injected with LPS every other day for a total of five injections. The next day after the last LPS, male mice were mated with untreated female mice. Interestingly, fetal weight and crown-rump length were reduced, while the incidence of IUGR was increased in paternal LPS exposure group. Additionally, paternal LPS exposure leaded to poor placental development through causing cell proliferation inhibition and apoptosis. Additional experiment demonstrated that the inactivation of placental PI3K/AKT pathway might be involved in paternal LPS-induced cell proliferation inhibition and apoptosis of trophoblast cells. Furthermore, the mRNA and protein levels of mesoderm specific transcript (MEST), a maternally imprinted gene with paternal expression, were significantly decreased in mouse placentas from paternal LPS exposure. Further analysis showed that paternal LPS exposure caused the inactivation of placental PI3K/AKT pathway and then cell proliferation inhibition and apoptosis might be via down-regulating placental MEST. Overall, our results provide evidence that paternal LPS exposure causes poor placental development and subsequently IUGR may be via down-regulating MEST/PI3K/AKT pathway, and then inducing cell proliferation inhibition and apoptosis in placentas.









Similar content being viewed by others
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Abbreviations
- LPS:
-
Lipopolysaccharide
- IUGR:
-
Intrauterine growth restriction
- MEST:
-
Mesoderm specific transcript
- GD:
-
Gestational day
- CCND1:
-
Cyclin D1
- PCNA:
-
Proliferating cell nuclear antigen
References
Argyraki M, Damdimopoulou P, Chatzimeletiou K et al (2019) In-utero stress and mode of conception: impact on regulation of imprinted genes, fetal development and future health. Hum Reprod Update 25:777–801
Barrio E, Calvo MT, Miramar MD et al (2009) Study of apoptosis and related proteins, CRH and hpGH in placentas of newborns small for gestational age (SGA). Pediatr Endocrinol Rev 6:337–342
Berends LM, Dearden L, Tung YCL et al (2018) Programming of central and peripheral insulin resistance by low birthweight and postnatal catch-up growth in male mice. Diabetologia 61:2225–2234
Burton GJ, Jauniaux E (2018) Pathophysiology of placental-derived fetal growth restriction. Am J Obstet Gynecol 218:S745–S761
Ceasrine AM, Devlin BA, Bolton JL et al (2022) Maternal diet disrupts the placenta-brain axis in a sex-specific manner. Nat Metab 4:1732–1745
Chen YH, Yu Z, Fu L et al (2015a) Supplementation with vitamin D3 during pregnancy protects against lipopolysaccharide-induced neural tube defects through improving placental folate transportation. Toxicol Sci 145:90–97
Chen YH, Yu Z, Fu L et al (2015b) Vitamin D3 inhibits lipopolysaccharide-induced placental inflammation through reinforcing interaction between vitamin D receptor and nuclear factor kappa B p65 subunit. Sci Rep 5:10871
Chen YH, Hu XG, Zhou Y et al (2016) Obeticholic acid protects against lipopolysaccharide-induced fetal death and intrauterine growth restriction through its anti-inflammatory activity. J Immunol 197:4762–4770
Chen YH, Liu ZB, Ma L et al (2020) Gestational vitamin D deficiency causes placental insufficiency and fetal intrauterine growth restriction partially through inducing placental inflammation. J Steroid Biochem Mol Biol 203:105733
Chew LC, Verma RP (2023) Fetal growth restriction. In: StatPearls. StatPearls Publishing, Treasure Island
Claycombe-Larson K, Singh B, Bundy AN et al (2023) Effects of maternal HF diet and absence of TRPC1 gene on mouse placental growth and fetal intrauterine growth retardation (IUGR). J Nutr Biochem 114:109162
Cotechini T, Komisarenko M, Sperou A et al (2014) Inflammation in rat pregnancy inhibits spiral artery remodeling leading to fetal growth restriction and features of preeclampsia. J Exp Med 211:165–179
Crispi F, Miranda J, Gratacós E (2018) Long-term cardiovascular consequences of fetal growth restriction: biology, clinical implications, and opportunities for prevention of adult disease. Am J Obstet Gynecol 218:S869–S879
Dall’Asta A, Stampalija T, Mecacci F et al (2022) Incidence, clinical features and perinatal outcome in anomalous fetuses with late-onset growth restriction: cohort study. Ultrasound Obstet Gynecol 60:632–639
Darendeliler F (2019) IUGR: genetic influences, metabolic problems, environmental associations/triggers, current and future management. Best Pract Res Clin Endocrinol Metab 33:101260
Gao XX, Lin S, Jiang PY et al (2022) Gestational cholestasis induced intrauterine growth restriction through triggering IRE1alpha mediated apoptosis of placental trophoblast cells. FASEB J 36:e22388
Green DR, Reed JC (1998) Mitochondria and apoptosis. Science 281:1309–1312
Harris LK, Pantham P, Yong HEJ et al (2019) The role of insulin-like growth factor 2 receptor-mediated homeobox gene expression in human placental apoptosis, and its implications in idiopathic fetal growth restriction. Mol Hum Reprod 25:572–585
Jacob AI, Goldberg PK, Bloom N, Degenshein GA, Kozinn PJ (1977) Endotoxin and bacteria in portal blood. Gastroenterology 72:1268–1270
Jaddoe VW, de Jonge LL, Hofman A et al (2014) First trimester fetal growth restriction and cardiovascular risk factors in school age children: population based cohort study. BMJ 348:g14
Kaiser AM, Forsthuber M, Widhalm R et al (2023) Prenatal exposure to per- and polyfluoroalkyl substances and pregnancy outcome in Austria. Ecotoxicol Environ Saf 259:115006
Kennedy LM, Tong S, Robinson AJ et al (2020) Reduced growth velocity from the mid-trimester is associated with placental insufficiency in fetuses born at a normal birthweight. BMC Med 18:395
Kowalska K, Kozieł MJ, Habrowska-Górczyńska DE et al (2022) Deoxynivalenol induces apoptosis and autophagy in human prostate epithelial cells via PI3K/Akt signaling pathway. Arch Toxicol 96:231–241
Kozai K, Iqbal K, Moreno-Irusta A et al (2021) Protective role of IL33 signaling in negative pregnancy outcomes associated with lipopolysaccharide exposure. FASEB J 35:e21272
Liebers V, Brüning T, Raulf M (2020) Occupational endotoxin exposure and health effects. Arch Toxicol 94:3629–3644
Lin S, Ye MY, Fu QY et al (2023) Cholic acid exposure during late pregnancy causes placental dysfunction and fetal growth restriction by reactive oxygen species-mediated activation of placental GCN2/eIF2α pathway. FASEB J 37:e22820
Moore GS, Kneitel AW, Walker CK et al (2012) Autism risk in small- and large-for-gestational-age infants. Am J Obstet Gynecol 206:314.e1–9
Nagata S (2018) Apoptosis and clearance of apoptotic cells. Annu Rev Immunol 36:489–517
Olga L, Sovio U, Wong H et al (2023) Association between antenatal diagnosis of late fetal growth restriction and educational outcomes in mid-childhood: a UK prospective cohort study with long-term data linkage study. PLoS Med 20:e1004225
Parlesak A, Schäfer C, Schütz T et al (2000) Increased intestinal permeability to macromolecules and endotoxemia in patients with chronic alcohol abuse in different stages of alcohol-induced liver disease. J Hepatol 32:742–747
Sandovici I, Georgopoulou A, Pérez-García V et al (2022) The imprinted Igf2-Igf2r axis is critical for matching placental microvasculature expansion to fetal growth. Dev Cell 57:63-79.e8
Sapehia D, Mahajan A, Singh P et al (2023) High dietary folate and low vitamin B12 in the parental diet disturbed the epigenetics of imprinted genes MEST and PHLDA2 in mice placenta. J Nutr Biochem 118:109354
Shen P, Ji S, Li X et al (2022) LPS-induced systemic inflammation caused mPOA-FSH/LH disturbance and impaired testicular function. Front Endocrinol (Lausanne) 13:886085
Tokoro S, Koshida S, Tsuji S et al (2023) Insufficient antenatal identification of fetal growth restriction leading to intrauterine fetal death: a regional population-based study in Japan. J Matern Fetal Neonatal Med 36:2167075
Vaughan OR, Maksym K, Silva E et al (2021) Placenta-specific Slc38a2/SNAT2 knockdown causes fetal growth restriction in mice. Clin Sci (Lond) 135(17):2049–2066
Wang X, Jian W, Luo Q et al (2022) CircSEMA4B inhibits the progression of breast cancer by encoding a novel protein SEMA4B-211aa and regulating AKT phosphorylation. Cell Death Dis 13:794
Wu J, Liu Y, Dou Z et al (2020) Black garlic melanoidins prevent obesity, reduce serum LPS levels and modulate the gut microbiota composition in high-fat diet-induced obese C57BL/6J mice. Food Funct 11(11):9585–9598
Yamazaki T, Galluzzi L (2022) BAX and BAK dynamics control mitochondrial DNA release during apoptosis. Cell Death Differ 29:1296–1298
Yang D, Wei Y, Lu Q et al (2021) Melatonin alleviates LPS-induced endoplasmic reticulum stress and inflammation in spermatogonial stem cells. J Cell Physiol 236:3536–3551
Yin Q, Bi R, Li H et al (2023) Regulatory role of human fibrocartilage stem cells in condyle osteochondroma. Cell Prolif 56:e13342
Zhong G, Wan F, Lan J et al (2021) Arsenic exposure induces intestinal barrier damage and consequent activation of gut-liver axis leading to inflammation and pyroptosis of liver in ducks. Sci Total Environ 788:147780
Zhou Y, Chen YH, Fu L et al (2017) Vitamin D3 pretreatment protects against lipopolysaccharide-induced early embryo loss through its anti-inflammatory effects. Am J Reprod Immunol 77:e12620
Zhu YD, Han Y, Huang K et al (2018) The impact of isolated maternal hypothyroxinaemia on the incidence of large-for-gestational-age infants: the Ma’anshan Birth Cohort study. BJOG 125:1118–1125
Zhu HL, Dai LM, Xiong YW et al (2022) Gestational exposure to environmental cadmium induces placental apoptosis and fetal growth restriction via Parkin-modulated MCL-1 degradation. J Hazard Mater 424:127268
Funding
This study was supported by the National Natural Science Foundation of China (82173559 and 31902225), Key Research and Development Projects of Anhui Province (202104j07020035), Top Talent Funding Project of Anhui Province in Colleges and Universities (gxbjZD2022011), Natural Science Foundation of Anhui Province (2208085MH205) and Promotion project of basic and clinical collaborative research from Anhui Medical University (2020xkjT038 and 2021xkjT022).
Author information
Authors and Affiliations
Contributions
Conceptualization: YHC; investigation: PYJ, SL, JRL, YL, LMZ, QH, YJF; data curation and formal analysis: PYJ, SL; funding acquisition: YHC, SL, LMZ, YJF; supervision: YHC; writing-original draft preparation: YHC; writing-review and editing: YHC, DXX.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, PY., Lin, S., Liu, JR. et al. Paternal lipopolysaccharide exposure induced intrauterine growth restriction via the inactivation of placental MEST/PI3K/AKT pathway in mice. Arch Toxicol 97, 2929–2941 (2023). https://doi.org/10.1007/s00204-023-03584-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03584-3