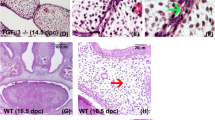
Abstract
While exposure to high levels of all-trans retinoic acid (atRA) during pregnancy is known to suppress murine embryonic palate mesenchymal (MEPM) cells proliferation and to result in cleft palate (CP) development, the underlying mechanisms are poorly understood. Accordingly, this study was designed with the goal of clarifying the etiological basis for atRA-induced CP. A murine model of CP was established via the oral administration of atRA to pregnant mice on gestational day (GD) 10.5, after which transcriptomic and metabolomic analyses were performed with the goal of clarifying the critical genes and metabolites associated with CP development through an integrated multi-omics approach. MEPM cells proliferation was altered by atRA exposure as expected, contributing to CP incidence. In total, 110 genes were differentially expressed in the atRA treatment groups, suggesting that atRA may influence key biological processes including stimulus, adhesion, and signaling-related activities. In addition, 133 differentially abundant metabolites were identified including molecules associated with ABC transporters, protein digestion and absorption, mTOR signaling pathway, and the TCA cycle, suggesting a link between these mechanisms and CP. Overall, combined analyses of these transcriptomic and metabolomic results suggested that the MAPK, calcium, PI3K-Akt, Wnt, and mTOR signaling pathways are particularly important pathways enriched in the palatal cleft under conditions of atRA exposure. Together, these integrated transcriptomic and metabolomic approaches provided new evidence with respect to the mechanisms underlying altered MEPM cells proliferation and signal transduction associated with atRA-induced CP, revealing a possible link between oxidative stress and these pathological changes.







Similar content being viewed by others
Data availability
All datasets generated and analyzed for this work can be found in published Article and supplementary material.
References
Ackermans MM, Zhou H, Carels CE, Wagener FA, Von den Hoff JW (2011) Vitamin A and clefting: putative biological mechanisms. Nutr Rev 69:613–624. https://doi.org/10.1111/j.1753-4887.2011.00425.x
Bastos Maia S, Rolland Souza AS, Costa Caminha MF, Lins da Silva S, Callou Cruz R, Carvalho dos Santos C et al (2019x) Vitamin A and pregnancy: a narrative review. Nutrients 11:681. https://doi.org/10.3390/nu11030681
Clagett-Dame M, Knutson D (2011) Vitamin A in reproduction and development. Nutrients 3:385–428. https://doi.org/10.3390/nu3040385
Dash S, Trainor PA (2020) The development, patterning and evolution of neural crest cell differentiation into cartilage and bone. Bone 137:115409. https://doi.org/10.1016/j.bone.2020.115409
Denu RA, Hematti P (2016) Effects of oxidative stress on mesenchymal stem cell biology. Oxid Med Cell Longev 2016:2989076. https://doi.org/10.1155/2016/2989076
Forman HJ, Zhang H (2021) Author correction: Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nat Rev Drug Discov 20:652. https://doi.org/10.1038/s41573-021-00267-5
Gurley JM, Wamsley MS, Sandell LJ (2004) Alterations in apoptosis and epithelial-mesenchymal transformation in an in vitro cleft palate model. Plast Reconstr Surg 113:907–914. https://doi.org/10.1097/01.Prs.0000105342.08168.13
Hu X, Gao J, Liao Y, Tang S, Lu F. Retinoic acid alters the proliferation and survival of the epithelium and mesenchyme and suppresses Wnt/beta-catenin signaling in developing cleft palate. Cell Death Dis 2013; 4: e898. http://doi.org/https://doi.org/10.1038/cddis.2013.424
Johnson CH, Ivanisevic J, Siuzdak G (2016) Metabolomics: beyond biomarkers and towards mechanisms. Nat Rev Mol Cell Biol 17:451–459. https://doi.org/10.1038/nrm.2016.25
Kim JK, Zhang J, Hwang S, Cho S, Yu WJ, Jeong JS et al (2022) Transcriptome-metabolome-wide association study (TMWAS) in rats revealed a potential carcinogenic effect of DEHP in thyroid associated with eicosanoids. Environ Res 214:113805. https://doi.org/10.1016/j.envres.2022.113805
Lan Y, Xu J, Jiang R (2015) Cellular and molecular mechanisms of palatogenesis. Curr Top Dev Biol 115:59–84. https://doi.org/10.1016/bs.ctdb.2015.07.002
Li J, Rodriguez G, Han X, Janeckova E, Kahng S, Song B et al (2019) Regulatory mechanisms of soft palate development and malformations. J Dent Res 98:959–967. https://doi.org/10.1177/0022034519851786
Liang L, Wang X, Zheng Y, Liu Y (2019) All-trans-retinoic acid modulates TGF-β-induced apoptosis, proliferation, migration and extracellular matrix synthesis of conjunctival fibroblasts by inhibiting PI3K/AKT signaling. Mol Med Rep. https://doi.org/10.3892/mmr.2019.10507
Liang C, Qiao G, Liu Y, Tian L, Hui N, Li J et al (2021) Overview of all-trans-retinoic acid (ATRA) and its analogues: structures, activities, and mechanisms in acute promyelocytic leukaemia. Eur J Med Chem 220:113451. https://doi.org/10.1016/j.ejmech.2021.113451
Liu X, Qi J, Tao Y, Zhang H, Yin J, Ji M et al (2016) Correlation of proliferation, TGF-beta3 promoter methylation, and Smad signaling in MEPM cells during the development of ATRA-induced cleft palate. Reprod Toxicol 61:1–9. https://doi.org/10.1016/j.reprotox.2016.02.010
Liu X, Liu H, Wu Y, He Z, Shen L, Zhang H et al (2021a) The role of lncRNA Meg3 in the proliferation of all-trans retinoic acid-treated mouse embryonic palate mesenchymal cells involves the Smad pathway. Reprod Toxicol 104:1–7. https://doi.org/10.1016/j.reprotox.2021.06.011
Liu X, Zhang Y, Shen L, He Z, Chen Y, Li N et al (2021b) LncRNA Meg3-mediated regulation of the Smad pathway in atRA-induced cleft palate. Toxicol Lett 341:51–58. https://doi.org/10.1016/j.toxlet.2021.01.017
Nielsen J (2017) Systems Biology of Metabolism. Annu Rev Biochem 86:245–275. https://doi.org/10.1146/annurev-biochem-061516-044757
Panahi M, Rahimi B, Rahimi G, Yew Low T, Saraygord-Afshari N, Alizadeh E (2020) Cytoprotective effects of antioxidant supplementation on mesenchymal stem cell therapy. J Cell Physiol 235:6462–6495. https://doi.org/10.1002/jcp.29660
Parada C, Han D, Grimaldi A, Sarrion P, Park SS, Pelikan R et al (2015) Disruption of the ERK/MAPK pathway in neural crest cells as a potential cause of Pierre Robin sequence. Development 142:3734–3745. https://doi.org/10.1242/dev.125328
Radu BM, Banciu A, Banciu DD, Radu M, Cretoiu D, Cretoiu SM (2017) Calcium signaling in interstitial cells: focus on telocytes. Int J Mol Sci 18:397. https://doi.org/10.3390/ijms18020397
Sakuma C, Imura H, Yamada T, Hirata A, Ikeda Y, Ito M et al (2022) Histological and immunohistochemical studies to determine the mechanism of cleft palate induction after palatal fusion in mice exposed to TCDD. Int J Mol Sci 23:2069. https://doi.org/10.3390/ijms23042069
Schubert M, Gibert Y (2020) Retinoids in embryonic development. Biomolecules 10:1278. https://doi.org/10.3390/biom10091278
Shi L, Li B, Zhang B, Zhen C, Zhou J, Tang S (2019) Mouse embryonic palatal mesenchymal cells maintain stemness through the PTEN-Akt-mTOR autophagic pathway. Stem Cell Res Ther 10:217. https://doi.org/10.1186/s13287-019-1340-8
Sultan M, Schulz MH, Richard H, Magen A, Klingenhoff A, Scherf M et al (2008) A global view of gene activity and alternative splicing by deep sequencing of the human transcriptome. Science 321:956–960. https://doi.org/10.1126/science.1160342
Wang Z, Gerstein M, Snyder M (2009) RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10:57–63. https://doi.org/10.1038/nrg2484
Wang X, Zhu X, Li X, Li Z, Mao Y, Zhang S et al (2023) Transcriptomic and metabolomic analyses provide insights into the attenuation of neuroinflammation by nervonic acid in MPTP-stimulated PD model mice. Food Funct 14:277–291. https://doi.org/10.1039/d2fo02595g
You M, Wang K, Pan Y, Tao L, Ma Q, Zhang G et al (2022) Combined royal jelly 10-hydroxydecanoic acid and aspirin has a synergistic effect against memory deficit and neuroinflammation. Food Funct 13:2336–2353. https://doi.org/10.1039/d1fo02397g
Yu Z, Xing Y (2006) atRA-induced apoptosis of mouse embryonic palate mesenchymal cells involves activation of MAPK pathway. Toxicol Appl Pharmacol 215:57–63. https://doi.org/10.1016/j.taap.2006.04.001
Yuan Q, Tang B, Zhang C (2022) Signaling pathways of chronic kidney diseases, implications for therapeutics. Signal Transduct Target Ther 7:182. https://doi.org/10.1038/s41392-022-01036-5
Zhang Y, Bao J, Gong X, Shi W, Liu T, Wang X (2022) Transcriptomics and metabolomics revealed the molecular mechanism of the toxic effect of mancozeb on liver of mice. Ecotoxicol Environ Saf 243:114003. https://doi.org/10.1016/j.ecoenv.2022.114003
Funding
This work was supported by the Natural Science Foundation of China (No. 81801547), Key project of science, technology of Henan province (No. SBGJ202102014), Major Projects of Collaborative Innovation of Zhengzhou (No.18XTZX12009), Special Major Public Welfare Project of Henan Province (No. 201300310800) and Key project of science, technology of Henan province (No. 222102310418).
Author information
Authors and Affiliations
Contributions
ZY: Conceptualization, data curation, formal analysis, investigation, methodology, roles/writing—original draft. SS: data curation, data curation, roles/writing—original draft. GW: conceptualization, writing—review and editing. YZ: conceptualization, methodology, visualization. YZ: conceptualization, project administration. YW: validation, visualization, writing—review and editing. HL: supervision, writing—review and editing. YZ: supervision. XL: conceptualization, data curation, funding acquisition, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflicts of interest were disclosed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Z., Song, S., Wang, G. et al. The mechanisms governing mouse embryonic palate mesenchymal cells’ proliferation associated with atRA-induced cleft palate in mice: insights from integrated transcriptomic and metabolomic analyses. Arch Toxicol 97, 2143–2153 (2023). https://doi.org/10.1007/s00204-023-03534-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03534-z