Abstract
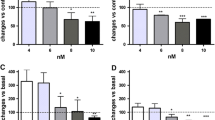
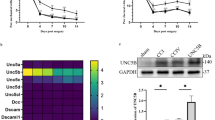
Bortezomib (BTZ) is a proteasome inhibitor serves as a first-line drug for multiple myeloma treatment. BTZ-induced peripheral neuropathy (BIPN) is the most common adverse effect of BTZ with an incidence as high as 40–60%. However, the pathological mechanisms underlying BIPN remain largely unclear. BTZ leads to dramatic Schwann cell demyelination in sciatic nerves. Previous studies implied that myelin debris was predominantly degraded via autophagy-lysosome pathway in Schwann cells. However, the association of autophagy with BIPN has not been made. Mice were treated with BTZ (2 mg/kg, i.v.) on Day1 and Day4 each week for continuous 4 weeks. BTZ-treated mice showed enhanced mechanical hyperalgesia, decreased tail nerve conduction and sciatic nerve demyelination. Unexpectedly, BTZ led to the accumulation of autophagic vesicles, LC3-II and p62 in the sciatic nerve. Moreover, BTZ blocked autophagic flux in RSC96 Schwann cells as determined by mcherry-GFP-LC3 assay, suggesting BTZ may impair lysosomal function rather than inducing autophagy in Schwann cells. BTZ significantly reduced the lysosomal activity in Schwann cells as determined by reduced LysoTracker Red and DQ-Red-BSA staining and increased the level of immature Cathepsin B (CTSB). Remarkably, lysosomal activators PP242 and Torin1, significantly reversed the blockage of autophagic flux by BTZ. We further verified that Torin1 rescued the demyelination, nerve conduction and reduced the mechanical hyperalgesia in BIPN mice. Additionally, Torin1 did not compromise the efficacy of BTZ in suppressing multiple myeloma RPMI8226 cell. Taken together, we identified that lysosomal dysfunction in Schwann cells caused by BTZ is involved in the BIPN pathology. Improved lysosomal function in Schwann cells can be a promising strategy for BIPN treatment.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aguilar LE, Jang SR, Park CH, Lee KM (2020) Supramolecular caffeic acid and bortezomib nanomedicine: prodrug inducing reactive oxygen species and inhibiting cancer cell survival. Pharmaceutics 12(11). https://doi.org/10.3390/pharmaceutics12111082
Arastu-Kapur S, Anderl JL, Kraus M et al (2011) Nonproteasomal targets of the proteasome inhibitors bortezomib and carfilzomib: a link to clinical adverse events. Clin Cancer Res 17(9):2734–2743. https://doi.org/10.1158/1078-0432.CCR-10-1950
Baiz D, Pozzato G, Dapas B et al (2009) Bortezomib arrests the proliferation of hepatocellular carcinoma cells HepG2 and JHH6 by differentially affecting E2F1, p21 and p27 levels. Biochimie 91(3):373–382. https://doi.org/10.1016/j.biochi.2008.10.015
Ballabio A, Bonifacino JS (2020) Lysosomes as dynamic regulators of cell and organismal homeostasis. Nat Rev Mol Cell Bio 21(2):101–118. https://doi.org/10.1038/s41580-019-0185-4
Belgrad J, De Pace R, Fields RD (2020) Autophagy in myelinating glia. J Neurosci 40(2):256–266. https://doi.org/10.1523/JNEUROSCI.1066-19.2019
Bloomingdale P, Meregalli C, Pollard K et al (2021) Systems pharmacology modeling identifies a novel treatment strategy for bortezomib-induced neuropathic pain. Front Pharmacol 12:817236. https://doi.org/10.3389/fphar.2021.817236
Bruna J, Alberti P, Calls-Cobos A, Caillaud M, Damaj MI, Navarro X (2020) Methods for in vivo studies in rodents of chemotherapy induced peripheral neuropathy. Exp Neurol 325:113154. https://doi.org/10.1016/j.expneurol.2019.113154
Bruna J, Udina E, Ale A et al (2010) Neurophysiological, histological and immunohistochemical characterization of bortezomib-induced neuropathy in mice. Exp Neurol 223(2):599–608. https://doi.org/10.1016/j.expneurol.2010.02.006
Carozzi VA, Canta A, Oggioni N et al (2010) Neurophysiological and neuropathological characterization of new murine models of chemotherapy-induced chronic peripheral neuropathies. Exp Neurol 226(2):301–309. https://doi.org/10.1016/j.expneurol.2010.09.004
Castillo K, Valenzuela V, Matus S et al (2013) Measurement of autophagy flux in the nervous system in vivo. Cell Death Dis 4:e917. https://doi.org/10.1038/cddis.2013.421
Cavaletti G, Gilardini A, Canta A et al (2007) Bortezomib-induced peripheral neurotoxicity: a neurophysiological and pathological study in the rat. Exp Neurol 204(1):317–325. https://doi.org/10.1016/j.expneurol.2006.11.010
Chen G, Zhang Z, Wei Z et al (2012) Lysosomal exocytosis in Schwann cells contributes to axon remyelination. Glia 60(2):295–305. https://doi.org/10.1002/glia.21263
Chen YF, Zhou XP (2020) Research progress of mTOR inhibitors. Eur J Med Chem 208:112820. https://doi.org/10.1016/j.ejmech.2020.112820
Frost P, Shi Y, Hoang B, Lichtenstein A (2007) AKT activity regulates the ability of mTOR inhibitors to prevent angiogenesis and VEGF expression in multiple myeloma cells. Oncogene 26(16):2255–2262. https://doi.org/10.1038/sj.onc.1210019
Hideshima T, Anderson KC (2002) Molecular mechanisms of novel therapeutic approaches for multiple myeloma. Nat Rev Cancer 2(12):927–937. https://doi.org/10.1038/nrc952
Holtzman E, Novikoff AB (1965) Lysomes in the rat sciatic nerve following crush. J Cell Biol 27(3):651–669. https://doi.org/10.1083/jcb.27.3.651
Jang SY, Shin YK, Park SY et al (2016) Autophagic myelin destruction by Schwann cells during Wallerian degeneration and segmental demyelination. Glia 64(5):730–742. https://doi.org/10.1002/glia.22957
Jang SY, Yoon BA, Shin YK et al (2017) Schwann cell dedifferentiation-associated demyelination leads to exocytotic myelin clearance in inflammatory segmental demyelination. Glia 65(11):1848–1862. https://doi.org/10.1002/glia.23200
Jessen KR, Mirsky R (2016) The repair Schwann cell and its function in regenerating nerves. J Physiol 594(13):3521–3531. https://doi.org/10.1113/JP270874
Kao C, Chao A, Tsai CL et al (2014) Bortezomib enhances cancer cell death by blocking the autophagic flux through stimulating ERK phosphorylation. Cell Death Dis 5. https://doi.org/10.1038/cddis.2014.468
Klionsky DJ, Abdel-Aziz AK, Abdelfatah S et al (2021) Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition). Autophagy 17(1):1–382. https://doi.org/10.1080/15548627.2020.1797280
Lao Y, Xu N (2016) autophagy in cancer chemoprevention: identification of novel autophagy modulators with anticancer potential. Methods Mol Biol 1379:151–163. https://doi.org/10.1007/978-1-4939-3191-0_14
Laussmann MA, Passante E, Dussmann H et al (2011) Proteasome inhibition can induce an autophagy-dependent apical activation of caspase-8. Cell Death Differ 18(10):1584–1597. https://doi.org/10.1038/cdd.2011.27
Liu XJ, Yin MX, Dong JW et al (2021) Tubeimoside-1 induces TFEB-dependent lysosomal degradation of PD-L1 and promotes antitumor immunity by targeting mTOR. Acta Pharm Sin B 11(10):3134–3149. https://doi.org/10.1016/j.apsb.2021.03.039
Mauthe M, Orhon I, Rocchi C et al (2018) Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion. Autophagy 14(8):1435–1455. https://doi.org/10.1080/15548627.2018.1474314
McCormack PL (2012) Carfilzomib: in relapsed, or relapsed and refractory, multiple myeloma. Drugs 72(15):2023–2032. https://doi.org/10.2165/11209010-000000000-00000
Nakano A, Abe M, Oda A et al (2011) Delayed treatment with vitamin C and N-acetyl-l-cysteine protects Schwann cells without compromising the anti-myeloma activity of bortezomib. Int J Hematol 93(6):727–735. https://doi.org/10.1007/s12185-011-0850-7
Pan B, Li J, Parajuli N et al (2020) The calcineurin-TFEB-p62 pathway mediates the activation of cardiac macroautophagy by proteasomal malfunction. Circ Res 127(4):502–518. https://doi.org/10.1161/Circresaha.119.316007
Park JT, Lee YS, Park SC (2019) Quantification of autophagy during senescence. Methods Mol Biol 1896:149–157. https://doi.org/10.1007/978-1-4939-8931-7_14
Pohl C, Dikic I (2019) Cellular quality control by the ubiquitin-proteasome system and autophagy. Science 366(6467):818–822. https://doi.org/10.1126/science.aax3769
Price OT, Lau C, Zucker RM (2003) Quantitative fluorescence of 5-FU-treated fetal rat limbs using confocal laser scanning microscopy and LysoTracker Red. Cytom Part A 53a(1):9–21. https://doi.org/10.1002/cyto.a.10036
Raab MS, Podar K, Breitkreutz I, Richardson PG, Anderson KC (2009) Multiple myeloma. Lancet 374(9686):324–339. https://doi.org/10.1016/S0140-6736(09)60221-X
Reed CB, Frick LR, Weaver A et al (2020) Deletion of calcineurin in Schwann cells does not affect developmental myelination, but reduces autophagy and delays myelin clearance after peripheral nerve injury. J Neurosci 40(32):6165–6176. https://doi.org/10.1523/JNEUROSCI.0951-20.2020
Richardson PG, Briemberg H, Jagannath S et al (2006) Frequency, characteristics, and reversibility of peripheral neuropathy during treatment of advanced multiple myeloma with bortezomib. J Clin Oncol 24(19):3113–3120. https://doi.org/10.1200/JCO.2005.04.7779
Settembre C, Di Malta C, Polito VA et al (2011) TFEB links autophagy to lysosomal biogenesis. Science 332(6036):1429–1433. https://doi.org/10.1126/science.1204592
Stubgen JP (2011) Drug-induced dysimmune demyelinating neuropathies. J Neurol Sci 307(1–2):1–8. https://doi.org/10.1016/j.jns.2011.05.010
Su WF, Wu F, Jin ZH et al (2019) Overexpression of P2X4 receptor in Schwann cells promotes motor and sensory functional recovery and remyelination via BDNF secretion after nerve injury. Glia 67(1):78–90. https://doi.org/10.1002/glia.23527
Thawani SP, Tanji K, De Sousa EA, Weimer LH, Brannagan TH 3rd (2015) Bortezomib-associated demyelinating neuropathy–clinical and pathologic features. J Clin Neuromuscul Dis 16(4):202–209. https://doi.org/10.1097/CND.0000000000000077
Velasco R, Petit J, Clapes V, Verdu E, Navarro X, Bruna J (2010) Neurological monitoring reduces the incidence of bortezomib-induced peripheral neuropathy in multiple myeloma patients. J Peripher Nerv Syst 15(1):17–25. https://doi.org/10.1111/j.1529-8027.2010.00248.x
Wang L, Zhao C, Zheng T et al (2022) Torin 1 alleviates impairment of TFEB-mediated lysosomal biogenesis and autophagy in TGFBI (p.G623_H626del)-linked Thiel-Behnke corneal dystrophy. Autophagy 18(4):765–782. https://doi.org/10.1080/15548627.2021.1955469
Xiang RF, Wang Y, Zhang N et al (2017) MK2206 enhances the cytocidal effects of bufalin in multiple myeloma by inhibiting the AKT/mTOR pathway. Cell Death Dis 8(5):e2776. https://doi.org/10.1038/cddis.2017.188
Yeung BH, Huang DC, Sinicrope FA (2006) PS-341 (bortezomib) induces lysosomal cathepsin B release and a caspase-2-dependent mitochondrial permeabilization and apoptosis in human pancreatic cancer cells. J Biol Chem 281(17):11923–11932. https://doi.org/10.1074/jbc.M508533200
Yu L, Chen Y, Tooze SA (2018) Autophagy pathway: Cellular and molecular mechanisms. Autophagy 14(2):207–215. https://doi.org/10.1080/15548627.2017.1378838
Zhang ZX, Chen C, Yang F et al (2022) Itaconate is a lysosomal inducer that promotes antibacterial innate immunity. Mol Cell 82(15):2844. https://doi.org/10.1016/j.molcel.2022.05.009
Zhou J, Tan SH, Nicolas V et al (2013) Activation of lysosomal function in the course of autophagy via mTORC1 suppression and autophagosome-lysosome fusion. Cell Res 23(4):508–523. https://doi.org/10.1038/cr.2013.11
Funding
This work was funded by the National Natural Science Foundation of China (81973402 and 82173792), Zhejiang Provincial Natural Science Foundation (LZ21H310001 and LYY22H310009) and the Starry Night Science Fund of Zhejiang University Shanghai Institute for Advanced Study (SN-ZJU-SIAS-0011).
Author information
Authors and Affiliations
Contributions
Correspondence to XZ and JW. ZW and WY contributed equally to this work. All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ZW, WY, XZ, JW, KW, GX, DZ, XL, HW and MY. The first draft of the manuscript was written by ZW, WY, XZ and JW. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
All procedures were performed in accordance with the guidelines of the Zhejiang University Animal Experimentation Committee and were in complete compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, Z., Yan, W., Wang, K. et al. Lysosomal dysfunction in Schwann cells is involved in bortezomib-induced peripheral neurotoxicity. Arch Toxicol 97, 1385–1396 (2023). https://doi.org/10.1007/s00204-023-03468-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03468-6