Abstract
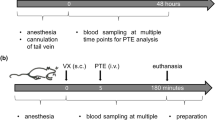
Recent events have shown that organophosphorus nerve agents (OPNAs) are a serious threat. Cholinesterase inhibition by OPNAs results in acetylcholine accumulation, a cholinergic crisis leading to death if untreated. Efficacy assessment of new medical countermeasures against OPNAs relies on translational animal models. We developed a swine model of percutaneous VX intoxication and a simple plate reader-based enzymatic method to quantify plasmatic VX over time. Juvenile pigs anesthetized with sevoflurane were poisoned with a single supralethal (n = 5; 1200 μg/kg) or sublethal (n = 6; 320 μg/kg) percutaneous dose of VX. These intoxicated animals were compared to 7 control animals. Repeated blood sampling was performed up to 6 h post-intoxication. Blood cholinesterase activities were measured using the Ellman assay. Nanomolar plasma concentrations of VX were measured by exogenous butyrylcholinesterase added to an aliquot of plasma. As expected, we observed a steady increase in plasma concentration of VX over time concomitant to a decrease in blood cholinesterase activities for all intoxicated pigs. Despite the simplicity of the enzymatic method, the results obtained are in good agreement with those of the liquid chromatography–mass spectrometry method. This method is also applicable to other OPNAs such as novichoks with minor adaptations.


Similar content being viewed by others
Abbreviations
- AChE:
-
Acetylcholinesterase
- ATC:
-
Acetylthiocholine
- BChE:
-
Butyrylcholinesterase
- BTC:
-
Butyrylthiocholine
- ChE:
-
Total cholinesterase
- DTNB:
-
5,5′-Dithiobis-2-nitrobenzoic acid
- E:
-
Enzyme
- EOPNA:
-
Inhibited enzyme
- hBChE:
-
Human butyrylcholinesterase
- IV:
-
Intravenous
- ki:
-
Bimolecular rate constant of inhibition
- LC–MS/MS:
-
Liquid chromatography–mass spectrometry
- OPNA:
-
Organophosphorus-nerve agents
- PC:
-
Percutaneous
- VX:
-
O-Ethyl S-diisopropylaminomethyl methylphosphonothiolate
References
Bartek MJ, LaBudde JA, Maibach HI (1972) Skin permeability in vivo: comparison in rat, rabbit, pig and man. J Investig Dermatol 58:114–123. https://doi.org/10.1111/1523-1747.ep12538909
Benschop HP, De Jong LPA (2001) Toxicokinetics of nerve agents. Vojen Zdr LISTY 11
Bester SM, Guelta MA, Cheung J et al (2018) Structural insights of stereospecific inhibition of human acetylcholinesterase by VX and subsequent reactivation by HI-6. Chem Res Toxicol 31:1405–1417. https://doi.org/10.1021/acs.chemrestox.8b00294
Bjarnason S, Mikler J, Hill I et al (2008) Comparison of selected skin decontaminant products and regimens against VX in domestic swine. Hum Exp Toxicol 27:253–261. https://doi.org/10.1177/0960327108090269
Black R (2016) Chapter 1—development, historical use and properties of chemical warfare agents. In: Worek F, Jenner J, Thiermann H (eds) Chemical warfare toxicology. Royal Society of Chemistry, pp 1–28
Chilcott RP, Dalton CH, Hill I et al (2005) In vivo skin absorption and distribution of the nerve agent VX (O-ethyl-S-[2(diisopropylamino)ethyl] methylphosphonothioate) in the domestic white pig. Hum Exp Toxicol 24:347–352. https://doi.org/10.1191/0960327105ht537oa
Craig FN, Cummings EG, Sim VM (1977) Environmental temperature and the percutaneous absorption of a cholinesterase inhibitor, VX. J Investig Dermatol 68:357–361. https://doi.org/10.1111/1523-1747.ep12496487
da Fonseca Carvalho LM (2021) Novichok(s) a challenge to the chemical weapons convention. Port J Mil Sci 9:39–62
Dorandeu F, Nachon F (2017) Des contre-mesures médicales face à la menace des neurotoxiques organophosphorés. Biofutur 2017:42–45
Dorandeu F, Mikler JR, Thiermann H et al (2007) Swine models in the design of more effective medical countermeasures against organophosphorus poisoning. Toxicology 233:128–144. https://doi.org/10.1016/j.tox.2006.09.013
Duncan EJS, Brown A, Lundy P et al (2002) Site-specific percutaneous absorption of methyl salicylate and VX in domestic swine. J Appl Toxicol JAT 22:141–148. https://doi.org/10.1002/jat.838
Eddleston M, Buckley NA, Eyer P, Dawson AH (2008) Management of acute organophosphorus pesticide poisoning. Lancet Lond Engl 371:597–607. https://doi.org/10.1016/S0140-6736(07)61202-1
Ellman GL, Courtney KD, Andres V, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Ganesan K, Raza SK, Vijayaraghavan R (2010) Chemical warfare agents. J Pharm Bioallied Sci 2:166–178. https://doi.org/10.4103/0975-7406.68498
Gupta RC (2015) Handbook of toxicology of chemical warfare agents, 2nd edn. Academic Press Elsevier, Amsterdam
Hamilton MG, Hill I, Conley J et al (2004) Clinical aspects of percutaneous poisoning by the chemical warfare agent VX: effects of application site and decontamination. Mil Med 169:856–862. https://doi.org/10.7205/milmed.169.11.856
Harris L, Broomfield C, Adams N, Stitcher D (1984) Detoxification of soman and o-cyclopentyl-s-diethylaminoethyl methylphosphonothioate in vivo. Proc West Pharmacol Soc 27:315–318
Hoenig SL (2007) Compendium of chemical warfare agents. https://link.springer.com/book/10.1007/978-0-387-69260-9
Jacobi U, Kaiser M, Toll R et al (2007) Porcine ear skin: an in vitro model for human skin. Skin Res Technol 13:19–24. https://doi.org/10.1111/j.1600-0846.2006.00179.x
John H, Balszuweit F, Steinritz D et al (2020) Chapter 52—toxicokinetic aspects of nerve agents and vesicants. In: Gupta RC (ed) Handbook of toxicology of chemical warfare agents, 3rd edn. Academic Press, Boston, pp 875–919
Langenberg JP, van Dijk C, Sweeney RE et al (1997) Development of a physiologically based model for the toxicokinetics of C(+/−)P(+/−)-soman in the atropinized guinea pig. Arch Toxicol 71:320–331. https://doi.org/10.1007/s002040050393
Lockridge O (2015) Review of human butyrylcholinesterase structure, function, genetic variants, history of use in the clinic, and potential therapeutic uses. Pharmacol Ther 148:34–46. https://doi.org/10.1016/j.pharmthera.2014.11.011
Mann TM, Price ME, Whitmore CL et al (2018) Bioscavenger is effective as a delayed therapeutic intervention following percutaneous VX poisoning in the guinea-pig. Toxicol Lett 293:198–206. https://doi.org/10.1016/j.toxlet.2017.11.029
Marrs TC, Maynard RL, Sidell FR (2007) Chemical warfare agents: toxicology and treatment, 2nd edn. Wiley, New York
Mikler J, Tenn C, Worek F et al (2011) Immobilization of Russian VX skin depots by localized cooling: implications for decontamination and medical countermeasures. Toxicol Lett 206:47–53. https://doi.org/10.1016/j.toxlet.2011.05.1047
Mumford H, Docx CJ, Price ME et al (2013) Human plasma-derived BuChE as a stoichiometric bioscavenger for treatment of nerve agent poisoning. Chem Biol Interact 203:160–166. https://doi.org/10.1016/j.cbi.2012.08.018
Munro NB, Talmage SS, Griffin GD et al (1999) The sources, fate, and toxicity of chemical warfare agent degradation products. Environ Health Perspect 107:933–974. https://doi.org/10.1289/ehp.99107933
Reiter G, Mikler J, Hill I et al (2008) Chromatographic resolution, characterisation and quantification of VX enantiomers in hemolysed swine blood samples. J Chromatogr B Anal Technol Biomed Life Sci 873:86–94. https://doi.org/10.1016/j.jchromb.2008.08.001
Reiter G, Mikler J, Hill I et al (2011) Simultaneous quantification of VX and its toxic metabolite in blood and plasma samples and its application for in vivo and in vitro toxicological studies. J Chromatogr B Anal Technol Biomed Life Sci 879:2704–2713. https://doi.org/10.1016/j.jchromb.2011.07.031
Reiter G, Müller S, Hill I et al (2015) In vitro and in vivo toxicological studies of V nerve agents: molecular and stereoselective aspects. Toxicol Lett 232:438–448. https://doi.org/10.1016/j.toxlet.2014.11.010
Reutter SA, Mioduszewski RJ, Thomson SA (2000) Evaluation of airborne exposure limits for VX: worker and general population exposure criteria. ECBC-TR-074. https://www.federalregister.gov/documents/2002/01/08/02-396/airborne-exposure-limits-for-chemical-warfare-agents-ga-tabun-gb-sarin-and-vx
Rolland P, Bolzinger M-A, Cruz C et al (2011) Human scalp permeability to the chemical warfare agent VX. Toxicol Vitro Int J Publ Assoc BIBRA 25:1974–1980. https://doi.org/10.1016/j.tiv.2011.06.021
Sawyer TW, Mikler J, Worek F et al (2011) The therapeutic use of localized cooling in the treatment of VX poisoning. Toxicol Lett 204:52–56. https://doi.org/10.1016/j.toxlet.2011.04.008
Silva GR, Borges I Jr, Figueroa-Villar JD, de Castro AT (2012) Defesa química: histórico, classificação dos agentes de guerra e ação dos neurotóxicos. Quím Nova 35:2083–2091. https://doi.org/10.1590/S0100-40422012001000033
Steindl D, Boehmerle W, Körner R et al (2021) Novichok nerve agent poisoning. Lancet Lond Engl 397:249–252. https://doi.org/10.1016/S0140-6736(20)32644-1
Taylor P, Radic Z, Hosea NA et al (1995) Structural bases for the specificity of cholinesterase catalysis and inhibition. Toxicol Lett 82–83:453–458. https://doi.org/10.1016/0378-4274(95)03575-3
Vale JA, Marrs TC, Maynard RL (2018) Novichok: a murderous nerve agent attack in the UK. Clin Toxicol Phila Pa 56:1093–1097. https://doi.org/10.1080/15563650.2018.1469759
Vallet V, Cruz C, Licausi J et al (2008) Percutaneous penetration and distribution of VX using in vitro pig or human excised skin validation of demeton-S-methyl as adequate simulant for VX skin permeation investigations. Toxicology 246:73–82. https://doi.org/10.1016/j.tox.2007.12.027
van der Schans MJ, Lander BJ, van der Wiel H et al (2003) Toxicokinetics of the nerve agent (+/−)-VX in anesthetized and atropinized hairless guinea pigs and marmosets after intravenous and percutaneous administration. Toxicol Appl Pharmacol 191:48–62. https://doi.org/10.1016/s0041-008x(03)00216-3
Worek F, Mast U, Kiderlen D et al (1999) Improved determination of acetylcholinesterase activity in human whole blood. Clin Chim Acta Int J Clin Chem 288:73–90. https://doi.org/10.1016/s0009-8981(99)00144-8
Acknowledgements
We are thankful to the French Ministry of Armed Forces for sustained funding of biomedical research under grants by the Direction Générale de l’Armement and Service de Santé des Armées (Grant PDH-2-NBC-4-C-4209 and PDH-2-NBC-4-C-4212).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fémy, F., Meesemaecker, G., Belverge, N. et al. Toxicokinetics of plasmatic VX in a swine model: comparison of a simple enzymatic titration method with a mass spectrometry method. Arch Toxicol 97, 421–428 (2023). https://doi.org/10.1007/s00204-022-03408-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03408-w