Abstract
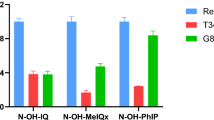
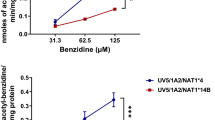
The use of new psychoactive substances (NPS) as drugs of abuse is common and increasingly popular, particularly among youth and neglected communities. Recent studies have reported acute toxic effects from these chemicals; however, their long-term toxicity is unknown. Genetic differences between individuals likely affect the toxicity risk. Arylamine N-acetyltransferase 2 (NAT2) capacity differs among individuals due to genetic inheritance. The goal of the present study is to investigate the gene–environment interaction between NAT2 polymorphism and toxicity after exposure to these chemicals. We measured N-acetylation by human NAT1 and NAT2 and found that N-acetylation of NPS is carried out exclusively by NAT2. Differences in N-acetylation between NAT2*4 (reference allele) and NAT2*5B (common variant allele) were highly significant (p < 0.0001). Using DNA repair-deficient genetically engineered Chinese hamster ovary (CHO cells), expressing human CYP1A2 and either NAT2*4 or NAT2*5B, we measured the induction of DNA double-strand breaks (\(\gamma\)H2Ax) following treatment of the CHO cells with increasing concentrations of NPS. The induction of \(\gamma\)H2Ax showed a NAT2 allele-dependent response, higher in the NAT2*4 vs NAT2*5B alleles (p < 0.05). Induction of oxidative stress (ROS/RNS) was evaluated; we observed NAT2 allele-dependent response for all compounds in concentrations as low as 10 \(\mu\)M, where NAT2*4 showed increased ROS/RNS vs NAT2*5B (p < 0.05). In summary, NPS are N-acetylated by NAT2 at rates higher in cells expressing NAT2*4 than NAT2*5B. Exposure to psychoactive chemicals results in genotoxic and oxidative damage that is modified by the NAT2 genetic polymorphism.



Similar content being viewed by others
References
Adamowicz P, Tokarczyk B, Stanaszek R, Slopianka M (2013) Fatal mephedrone intoxication—a case report. J Anal Toxicol 37(1):37–42. https://doi.org/10.1093/jat/bks085
Andreasen MF, Telving R, Birkler RI, Schumacher B, Johannsen M (2009) A fatal poisoning involving Bromo-Dragonfly. Forensic Sci Int 183(1–3):91–96. https://doi.org/10.1016/j.forsciint.2008.11.001
Audebert M, Riu A, Jacques C et al (2010) Use of the gammaH2AX assay for assessing the genotoxicity of polycyclic aromatic hydrocarbons in human cell lines. Toxicol Lett 199(2):182–192. https://doi.org/10.1016/j.toxlet.2010.08.022
Baldauf KJ, Salazar-Gonzalez RA, Doll MA, Pierce WM Jr, States JC, Hein DW (2020) Role of human N-acetyltransferase 2 genetic polymorphism on aromatic amine carcinogen-induced DNA damage and mutagenicity in a chinese hamster ovary cell mutation assay. Environ Mol Mutagen 61(2):235–245. https://doi.org/10.1002/em.22331
Brooks PJ (1997) DNA damage, DNA repair, and alcohol toxicity—a review. Alcohol Clin Exp Res 21(6):1073–1082
Camporez JP, Wang Y, Faarkrog K, Chukijrungroat N, Petersen KF, Shulman GI (2017) Mechanism by which arylamine N-acetyltransferase 1 ablation causes insulin resistance in mice. Proc Natl Acad Sci USA 114(52):E11285–E11292. https://doi.org/10.1073/pnas.1716990115
Cascorbi I, Drakoulis N, Brockmoller J, Maurer A, Sperling K, Roots I (1995) Arylamine N-acetyltransferase (NAT2) mutations and their allelic linkage in unrelated Caucasian individuals: correlation with phenotypic activity. Am J Hum Genet 57(3):581–592
Coffman BL, King CD, Rios GR, Tephly TR (1998) The glucuronidation of opioids, other xenobiotics, and androgens by human UGT2B7Y(268) and UGT2B7H(268). Drug Metab Dispos 26(1):73–77
Conway LP, Rendo V, Correia MSP, Bergdahl IA, Sjoblom T, Globisch D (2020) Unexpected acetylation of endogenous aliphatic amines by arylamine n-acetyltransferase NAT2. Angew Chem Int Ed Engl 59(34):14342–14346. https://doi.org/10.1002/anie.202005915
Crabbe JC, Jarvik LF, Liston EH, Jenden DJ (1983) Behavioral responses to amphetamines in identical twins. Acta Genet Med Gemellol (roma) 32(2):139–149. https://doi.org/10.1017/s0001566000006425
Dias da Silva D, Silva E, Carmo H (2014) Combination effects of amphetamines under hyperthermia–the role played by oxidative stress. J Appl Toxicol 34(6):637–650. https://doi.org/10.1002/jat.2889
Dias da Silva D, Ferreira B, Roque Bravo R et al (2019) The new psychoactive substance 3-methylmethcathinone (3-MMC or metaphedrone) induces oxidative stress, apoptosis, and autophagy in primary rat hepatocytes at human-relevant concentrations. Arch Toxicol 93(9):2617–2634. https://doi.org/10.1007/s00204-019-02539-x
Doll MA, Hein DW (2017) Genetic heterogeneity among slow acetylator N-acetyltransferase 2 phenotypes in cryopreserved human hepatocytes. Arch Toxicol 91(7):2655–2661. https://doi.org/10.1007/s00204-017-1988-8
Doll MA, Zang Y, Moeller T, Hein DW (2010) Codominant expression of N-acetylation and O-acetylation activities catalyzed by N-acetyltransferase 2 in human hepatocytes. J Pharmacol Exp Ther 334(2):540–544. https://doi.org/10.1124/jpet.110.168567
Doll MA, Salazar-Gonzalez RA, Bodduluri S, Hein DW (2017) Arylamine N-acetyltransferase 2 genotype-dependent N-acetylation of isoniazid in cryopreserved human hepatocytes. Acta Pharmaceutica Sinica B 7(4):517–522. https://doi.org/10.1016/j.apsb.2017.05.003
El Kawak M, Dhaini HR, Jabbour ME, Moussa MA, El Asmar K, Aoun M (2020) Slow N-acetylation as a possible contributor to bladder carcinogenesis. Mol Carcinog 59(9):1017–1027. https://doi.org/10.1002/mc.23232
EMCDDA (2018) European drug report 2018: trends and developments european monitoring centre for drugs and drug addiction. Office of the European Union, Luxemburg
Forrester MB, Merz RD (2007) Risk of selected birth defects with prenatal illicit drug use, Hawaii, 1986–2002. J Toxicol Environ Health A 70(1):7–18. https://doi.org/10.1080/15287390600748799
Fretland AJ, Doll MA, Leff MA, Hein DW (2001) Functional characterization of nucleotide polymorphisms in the coding region of N-acetyltransferase 1. Pharmacogenetics 11(6):511–520
G. B. D. Alcohol Drug Use C (2018) The global burden of disease attributable to alcohol and drug use in 195 countries and territories, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Psychiatry 5(12):987–1012. https://doi.org/10.1016/S2215-0366(18)30337-7
G. B. D. Causes of Death C (2018) Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392(10159):1736–1788. https://doi.org/10.1016/S0140-6736(18)32203-7
Grant DM, Hughes NC, Janezic SA et al (1997) Human acetyltransferase polymorphisms. Mutat Res 376(1–2):61–70
Habil MR, Doll MA, Hein DW (2020) N-acetyltransferase 2 acetylator genotype-dependent N-acetylation of 4-aminobiphenyl in cryopreserved human hepatocytes. Pharmacogenet Genomics 30(3):61–65. https://doi.org/10.1097/fpc.0000000000000394
Hein DW, Millner LM (2021) Arylamine N-acetyltransferase acetylation polymorphisms: paradigm for pharmacogenomic-guided therapy—a focused review. Expert Opin Drug Metab Toxicol 17(1):9–21. https://doi.org/10.1080/17425255.2021.1840551
Hein DW, Doll MA, Fretland AJ et al (2000) Molecular genetics and epidemiology of the NAT1 and NAT2 acetylation polymorphisms. Cancer Epidemiol Biomarkers Prev 9(1):29–42
Hein DW, Doll MA, Nerland DE, Fretland AJ (2006) Tissue distribution of N-acetyltransferase 1 and 2 catalyzing the N-acetylation of 4-aminobiphenyl and O-acetylation of N-hydroxy-4-aminobiphenyl in the congenic rapid and slow acetylator Syrian hamster. Mol Carcinog 45(4):230–238
Hein DW, Millner LM, Leggett CS, Doll MA (2010) Relationship between N-acetyltransferase 2 single-nucleotide polymorphisms and phenotype. Carcinogenesis 31(2):326–327. https://doi.org/10.1093/carcin/bgp290
Hong BL, D’Cunha R, Li P et al (2020a) A systematic review and meta-analysis of isoniazid pharmacokinetics in healthy volunteers and patients with tuberculosis. Clin Ther 42(11):e220–e241. https://doi.org/10.1016/j.clinthera.2020.09.009
Hong KU, Doll MA, Lykoudi A et al (2020b) Acetylator genotype-dependent dyslipidemia in rats congenic for N-acetyltransferase 2. Toxicol Rep 7:1319–1330. https://doi.org/10.1016/j.toxrep.2020.09.011
Jeng W, Ramkissoon A, Parman T, Wells PG (2006) Prostaglandin H synthase-catalyzed bioactivation of amphetamines to free radical intermediates that cause CNS regional DNA oxidation and nerve terminal degeneration. FASEB J 20(6):638–650. https://doi.org/10.1096/fj.05-5271com
Kendler KS, Jacobson KC, Prescott CA, Neale MC (2003) Specificity of genetic and environmental risk factors for use and abuse/dependence of cannabis, cocaine, hallucinogens, sedatives, stimulants, and opiates in male twins. Am J Psychiatry 160(4):687–695. https://doi.org/10.1176/appi.ajp.160.4.687
Knowles JW, Xie W, Zhang Z et al (2015) Identification and validation of N-acetyltransferase 2 as an insulin sensitivity gene. J Clin Investig 125(4):1739–1751. https://doi.org/10.1172/JCI74692
Leff MA, Fretland AJ, Doll MA, Hein DW (1999) Novel human N-acetyltransferase 2 alleles that differ in mechanism for slow acetylator phenotype. J Biol Chem 274(49):34519–34522
Li JH, Hu HC, Chen WB, Lin SK (2003) Genetic toxicity of methamphetamine in vitro and in human abusers. Environ Mol Mutagen 42(4):233–242. https://doi.org/10.1002/em.10198
McDonagh EM, Boukouvala S, Aklillu E, Hein DW, Altman RB, Klein TE (2014) PharmGKB summary: very important pharmacogene information for N-acetyltransferase 2. Pharmacogenet Genomics 24(8):409–425. https://doi.org/10.1097/fpc.0000000000000062
Metry KJ, Zhao S, Neale JR et al (2007) 2-amino-1-methyl-6-phenylimidazo [4,5-b] pyridine-induced DNA adducts and genotoxicity in Chinese hamster ovary (CHO) cells expressing human CYP1A2 and rapid or slow acetylator N-acetyltransferase 2. Mol Carcinog 46(7):553–563. https://doi.org/10.1002/mc.20302
Meyer MR, Maurer HH (2010) Metabolism of designer drugs of abuse: an updated review. Curr Drug Metab 11(5):468–482. https://doi.org/10.2174/138920010791526042
Meyer MR, Lindauer C, Maurer HH (2014a) Dimethocaine, a synthetic cocaine derivative: studies on its in vitro metabolism catalyzed by P450s and NAT2. Toxicol Lett 225(1):139–146. https://doi.org/10.1016/j.toxlet.2013.11.033
Meyer MR, Robert A, Maurer HH (2014b) Toxicokinetics of novel psychoactive substances: characterization of N-acetyltransferase (NAT) isoenzymes involved in the phase II metabolism of 2C designer drugs. Toxicol Lett 227(2):124–128. https://doi.org/10.1016/j.toxlet.2014.03.010
Millner LM, Doll MA, Cai J, States JC, Hein DW (2012a) NATb/NAT1*4 promotes greater arylamine N-acetyltransferase 1 mediated DNA adducts and mutations than NATa/NAT1*4 following exposure to 4-aminobiphenyl. Mol Carcinog 51(8):636–646. https://doi.org/10.1002/mc.20836
Millner LM, Doll MA, Cai J, States JC, Hein DW (2012b) Phenotype of the most common “slow acetylator” arylamine N-acetyltransferase 1 genetic variant (NAT1*14B) is substrate-dependent. Drug Metab Dispos 40(1):198–204. https://doi.org/10.1124/dmd.111.041855
Millner LM, Doll MA, Stepp MW, States JC, Hein DW (2012c) Functional analysis of arylamine N-acetyltransferase 1 (NAT1) NAT1*10 haplotypes in a complete NATb mRNA construct. Carcinogenesis 33(2):348–355. https://doi.org/10.1093/carcin/bgr273
Mitchell SC (2020) N-acetyltransferase: the practical consequences of polymorphic activity in man. Xenobiotica 50(1):77–91. https://doi.org/10.1080/00498254.2019.1618511
Mortensen HM, Froment A, Lema G et al (2011) Characterization of genetic variation and natural selection at the arylamine N-acetyltransferase genes in global human populations. Pharmacogenomics 12(11):1545–1558. https://doi.org/10.2217/pgs.11.88
Nelson ME, Bryant SM, Aks SE (2014) Emerging drugs of abuse. Emerg Med Clin North Am 32(1):1–28. https://doi.org/10.1016/j.emc.2013.09.001
Ritchie H, Roser M (2019) Drug Use. In: Our World in Data. https://ourworldindata.org/drug-use. Accessed 20 July 2022
Rojek S, Klys M, Maciow-Glab M, Kula K, Strona M (2014) Cathinones derivatives-related deaths as exemplified by two fatal cases involving methcathinone with 4-methylmethcathinone and 4-methylethcathinone. Drug Test Anal 6(7–8):770–777. https://doi.org/10.1002/dta.1615
Ropek N, Al-Serori H, Misik M et al (2019) Methamphetamine (“crystal meth”) causes induction of DNA damage and chromosomal aberrations in human derived cells. Food Chem Toxicol 128:1–7. https://doi.org/10.1016/j.fct.2019.03.035
Sabbagh A, Darlu P, Crouau-Roy B, Poloni ES (2011) Arylamine N-acetyltransferase 2 (NAT2) genetic diversity and traditional subsistence: a worldwide population survey. PLoS ONE 6(4):e18507. https://doi.org/10.1371/journal.pone.0018507
Shafi A, Berry AJ, Sumnall H, Wood DM, Tracy DK (2020) New psychoactive substances: a review and updates. Ther Adv Psychopharmacol 10:2045125320967197. https://doi.org/10.1177/2045125320967197
Sim E, Abuhammad A, Ryan A (2014) Arylamine N-acetyltransferases: from drug metabolism and pharmacogenetics to drug discovery. Br J Pharmacol 171(11):2705–2725. https://doi.org/10.1111/bph.12598
Song Y, Qi X, Liu X (2020) N-acetyltransferase 2 polymorphism is associated with bladder cancer risk: an updated meta-analysis based on 54 case-control studies. Gene 757:144924. https://doi.org/10.1016/j.gene.2020.144924
Suarez-Kurtz G, Fuchshuber-Moraes M, Struchiner CJ, Parra EJ (2016) Single nucleotide polymorphism coverage and inference of N-acetyltransferase-2 acetylator phenotypes in wordwide population groups. Pharmacogenet Genomics 26(8):363–369. https://doi.org/10.1097/FPC.0000000000000225
Totah RA, Allen KE, Sheffels P, Whittington D, Kharasch ED (2007) Enantiomeric metabolic interactions and stereoselective human methadone metabolism. J Pharmacol Exp Ther 321(1):389–399. https://doi.org/10.1124/jpet.106.117580
UNODC (2013) The challenge of new psychoactive substances global SMART programme. United Nations Office on Drug and Crime, Vienna, pp 1–122
UNODC (2021) Current NPS threats global SMART programme, vol IV. United Nations Office on Drug and Crime, Vienna, pp 1–6
Wagmann L, Hempel N, Richter LHJ, Brandt SD, Stratford A, Meyer MR (2019) Phenethylamine-derived new psychoactive substances 2C-E-FLY, 2C-EF-FLY, and 2C-T-7-FLY: investigations on their metabolic fate including isoenzyme activities and their toxicological detectability in urine screenings. Drug Test Anal 11(10):1507–1521. https://doi.org/10.1002/dta.2675
Weber WW, Hein DW (1985) N-acetylation pharmacogenetics. Pharmacol Rev 37(1):25–79
Weber CA, Salazar EP, Stewart SA, Thompson LH (1988) Molecular cloning and biological characterization of a human gene, ERCC2, that corrects the nucleotide excision repair defect in CHO UV5 cells. Mol Cell Biol 8(3):1137–1146
Acknowledgements
Research reported in this publication/presentation was supported by the National Institute of Environmental Health Sciences of the National Institutes of Health under Award Number P30ES030283. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
RASG: conceived and designed study, performed research, analyzed data, interpretation of the data, contributed new methods and models, and wrote the manuscript. MAD: performed research, analyzed data, interpretation of the data, contributed new methods and models, and edited the manuscript. DWH: conceived and designed study, interpretation of the data, and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Salazar-González, R.A., Doll, M.A. & Hein, D.W. N-acetyltransferase 2 genetic polymorphism modifies genotoxic and oxidative damage from new psychoactive substances. Arch Toxicol 97, 189–199 (2023). https://doi.org/10.1007/s00204-022-03383-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03383-2