Abstract
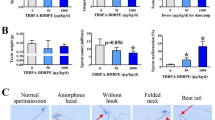
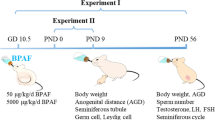
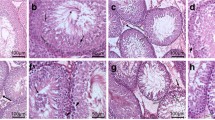
Whether or not tetrabromobisphenol A (TBBPA) has reproductive developmental toxicity remains controversial. Here, we evaluated the effects of postnatal TBBPA exposure of dams (before weaning) and pups through drinking water (15, 150, 1500 ng/mL) on testis development in mice. On postnatal day (PND) 56, we found that TBBPA exerted little effects on testis weight, anogenital distance, sperm parameters, and the serum testosterone level, but resulted in dose-dependent reductions in the seminiferous tubule area coupled with decreased Sertoli cells and spermatogonia and the number of stage VII–VIII seminiferous tubules, and cytoskeleton damage in Sertoli cells, along with down-regulated expression of marker genes for Sertoli cells, spermatogonia and spermatocyte. Further study revealed that the reduced tubule area coupled decreased Sertoli cell and germ cell numbers and marker gene expression also occurred in TBBPA-treated testes on PND 7, along with reduced cell proliferation and disordered arrangement of Sertoli cell nuclei. On PND 15, most of these testicular alterations were still observed in TBBPA-treated males, and cytoskeleton damage in Sertoli cells became observable. All observations convincingly demonstrate that postnatal exposure to TBBPA disturbed testis development in early life and ultimately caused adverse outcomes in adult testes, and that cell proliferation inhibition, the reduction in the seminiferous tubule area coupled decreased Sertoli cell and germ cell numbers and marker gene expression, and cytoskeleton damage in Sertoli cells, are early events contributing to adverse outcomes in adult testes. Our study improves the understanding of reproductive developmental toxicity of TBBPA, highlighting its risk for human health.







Similar content being viewed by others
References
Brown PR, Gillera SEA, Fenton SE, Yao HH (2020) Developmental exposure to tetrabromobisphenol A has minimal impact on male rat reproductive health. Reprod Toxicol 95:59–65. https://doi.org/10.1016/j.reprotox.2020.05.003
Cariou R, Antignac JP, Zalko D et al (2008) Exposure assessment of French women and their newborns to tetrabromobisphenol-A: occurrence measurements in maternal adipose tissue, serum, breast milk and cord serum. Chemosphere 73(7):1036–1041. https://doi.org/10.1016/j.chemosphere.2008.07.084
Chain EPoCitF (2011) Scientific opinion on tetrabromobisphenol A (TBBPA) and its derivatives in food. EFSA J 9(12):2477. https://doi.org/10.2903/j.efsa.2011.2477
Chojnacka K, Bilinska B, Mruk DD (2016) Interleukin 1alpha-induced disruption of the Sertoli cell cytoskeleton affects gap junctional communication. Cell Signal 28(5):469–480. https://doi.org/10.1016/j.cellsig.2016.02.003
Cope RB, Kacew S, Dourson M (2015) A reproductive, developmental and neurobehavioral study following oral exposure of tetrabromobisphenol A on Sprague-Dawley rats. Toxicology 329:49–59. https://doi.org/10.1016/j.tox.2014.12.013
Dirami G, Ravindranath N, Achi MV, Dym M (2001) Expression of Notch pathway components in spermatogonia and Sertoli cells of neonatal mice. J Androl 22(6):944–952. https://doi.org/10.1002/j.1939-4640.2001.tb03434.x
Dong M, Li Y, Zhu M, Qin Z (2021) Tetrabromobisphenol A: a neurotoxicant or not? Environ Sci Pollut Res Int 28(39):54466–54476. https://doi.org/10.1007/s11356-021-15166-w
European Union (2008) Risk Assessment of 2,2’,6,6’-tetrabtomo-4,4’-isopropylidene diphenol (tetrabromobisphenol-A) CAS Number: 79-947. http://europaenuint
França LR, Hess RA, Dufour JM, Hofmann MC, Griswold MD (2016) The Sertoli cell: one hundred fifty years of beauty and plasticity. Andrology 4(2):189–212. https://doi.org/10.1111/andr.12165
Garcia TX, DeFalco T, Capel B, Hofmann MC (2013) Constitutive activation of NOTCH1 signaling in Sertoli cells causes gonocyte exit from quiescence. Dev Biol 377(1):188–201. https://doi.org/10.1016/j.ydbio.2013.01.031
Gundersen GG, Worman HJ (2013) Nuclear positioning. Cell 152(6):1376–1389. https://doi.org/10.1016/j.cell.2013.02.031
Hutchison GR, Scott HM, Walker M et al (2008) Sertoli cell development and function in an animal model of testicular dysgenesis syndrome. Biol Reprod 78(2):352–360. https://doi.org/10.1095/biolreprod.107.064006
Kemmlein S, Herzke D, Law RJ (2003) BFR-governmental testing programme. Environ Int 29(6):781–792. https://doi.org/10.1016/s0160-4120(03)00112-0
Li L, Gao Y, Chen H et al (2017) Cell polarity, cell adhesion, and spermatogenesis: role of cytoskeletons. F1000Res 6:1565–1565. https://doi.org/10.12688/f1000research.11421.1
Li Y, Shen Y, Li J, Cai M, Qin Z (2020) Transcriptomic analysis identifies early cellular and molecular events by which estrogen disrupts testis differentiation and causes feminization in Xenopus laevis. Aquat Toxicol 226:105557. https://doi.org/10.1016/j.aquatox.2020.105557
Liu K, Li J, Yan S, Zhang W, Li Y, Han D (2016) A review of status of tetrabromobisphenol A (TBBPA) in China. Chemosphere 148:8–20. https://doi.org/10.1016/j.chemosphere.2016.01.023
Mäkelä J-A, Koskenniemi JJ, Virtanen HE, Toppari J (2018) Testis development. Endocr Rev 40(4):857–905. https://doi.org/10.1210/er.2018-00140
Malkoske T, Tang Y, Xu W, Yu S, Wang H (2016) A review of the environmental distribution, fate, and control of tetrabromobisphenol A released from sources. Sci Total Environ 569–570:1608–1617. https://doi.org/10.1016/j.scitotenv.2016.06.062
Meroni SB, Galardo MN, Rindone G, Gorga A, Riera MF, Cigorraga SB (2019) Molecular mechanisms and signaling pathways involved in Sertoli cell proliferation. Front Endocrinol 10:224. https://doi.org/10.3389/fendo.2019.00224
NTP (2014) NTP technical report on the toxicology studies of tetrabromo-bisphenol a (CAS No. 79-94-7) in F344/NTac rats and B6C3F1/N Mice and toxicology and carcinogenesis studies of tetrabromobisphernol A in Wistar Han (Crl:WI(Han)) rats and B6C3F1/N mice (Gavage studies)
Rebourcet D, O’Shaughnessy PJ, Pitetti JL et al (2014) Sertoli cells control peritubular myoid cell fate and support adult Leydig cell development in the prepubertal testis. Development 141(10):2139–2149. https://doi.org/10.1242/dev.107029
Rebourcet D, Darbey A, Monteiro A et al (2017) Sertoli cell number defines and predicts germ and leydig cell population sizes in the adult mouse testis. Endocrinology 158(9):2955–2969. https://doi.org/10.1210/en.2017-00196
Rebourcet D, Monteiro A, Cruickshanks L et al (2019) Relationship of transcriptional markers to Leydig cell number in the mouse testis. PLoS ONE 14(7):e0219524. https://doi.org/10.1371/journal.pone.0219524
Ren XM, Yao L, Xue Q et al (2020) Binding and activity of tetrabromobisphenol A mono-ether structural analogs to thyroid hormone transport proteins and receptors. Environ Health Perspect 128(10):107008. https://doi.org/10.1289/EHP6498
Saegusa Y, Fujimoto H, Woo G-H et al (2009) Developmental toxicity of brominated flame retardants, tetrabromobisphenol A and 1,2,5,6,9,10-hexabromocyclododecane, in rat offspring after maternal exposure from mid-gestation through lactation. Reprod Toxicol 28(4):456–467. https://doi.org/10.1016/j.reprotox.2009.06.011
Sharpe R, Mckinnell C, Kivlin C, Fisher J (2003) Proliferation and functional maturation of Sertoli cells, and their relevance to disorders of testis function in adulthood. Reproduction 125(6):769–784. https://doi.org/10.1530/rep.0.1250769
Shi Z-X, Wu Y-N, Li J-G, Zhao Y-F, Feng J-F (2009) Dietary exposure assessment of Chinese adults and nursing infants to tetrabromobisphenol-A and hexabromocyclododecanes: occurrence measurements in foods and human milk. Environ Sci Technol 43(12):4314–4319. https://doi.org/10.1021/es8035626
Shi Z, Zhang L, Zhao Y et al (2017) A national survey of tetrabromobisphenol-A, hexabromocyclododecane and decabrominated diphenyl ether in human milk from China: occurrence and exposure assessment. Sci Total Environ 599–600:237–245. https://doi.org/10.1016/j.scitotenv.2017.04.237
Tada Y, Fujitani T, Yano N et al (2006) Effects of tetrabromobisphenol A, brominated flame retardant, in ICR mice after prenatal and postnatal exposure. Food Chem Toxicol 44(8):1408–1413. https://doi.org/10.1016/j.fct.2006.03.006
Van der Ven LT, Van de Kuil T, Verhoef A et al (2008) Endocrine effects of tetrabromobisphenol-A (TBBPA) in Wistar rats as tested in a one-generation reproduction study and a subacute toxicity study. Toxicology 245(1–2):76–89. https://doi.org/10.1016/j.tox.2007.12.009
Wang W, Abualnaja KO, Asimakopoulos AG et al (2015) A comparative assessment of human exposure to tetrabromobisphenol A and eight bisphenols including bisphenol A via indoor dust ingestion in twelve countries. Environ Int 83:183–191. https://doi.org/10.1016/j.envint.2015.06.015
Wang Y, Li Y, Qin Z, Wei W (2017) Re-evaluation of thyroid hormone signaling antagonism of tetrabromobisphenol A for validating the T3-induced Xenopus metamorphosis assay. J Environ Sci 52:325–332. https://doi.org/10.1016/j.jes.2016.09.021
Weber JE, Russell LD, Wong V, Peterson RN (1983) Three-dimensional reconstruction of a rat stage V Sertoli cell: II. Morphometry of Sertoli-Sertoli and Sertoli–germ-cell relationships. Am J Anat 167(2):163–179. https://doi.org/10.1002/aja.1001670203
Yin N, Liang S, Liang S et al (2018) TBBPA and its alternatives disturb the early stages of neural development by interfering with the NOTCH and WNT pathways. Environ Sci Technol 52(9):5459–5468. https://doi.org/10.1021/acs.est.8b00414
Yu Y, Yu Z, Chen H et al (2019) Tetrabromobisphenol A: disposition, kinetics and toxicity in animals and humans. Environ Pollut 253:909–917. https://doi.org/10.1016/j.envpol.2019.07.067
Zatecka E, Ded L, Elzeinova F et al (2013) Effect of tetrabrombisphenol A on induction of apoptosis in the testes and changes in expression of selected testicular genes in CD1 mice. Reprod Toxicol 35:32–39. https://doi.org/10.1016/j.reprotox.2012.05.095
Zhang YF, Xu W, Lou QQ et al (2014) Tetrabromobisphenol A disrupts vertebrate development via thyroid hormone signaling pathway in a developmental stage-dependent manner. Environ Sci Technol 48(14):8227–8234. https://doi.org/10.1021/es502366g
Zhou X, Guo J, Zhang W, Zhou P, Deng J, Lin K (2014) Tetrabromobisphenol A contamination and emission in printed circuit board production and implications for human exposure. J Hazard Mater 273:27–35. https://doi.org/10.1016/j.jhazmat.2014.03.003
Zhou H, Yin N, Faiola F (2020) Tetrabromobisphenol A (TBBPA): a controversial environmental pollutant. J Environ Sci (china) 97:54–66. https://doi.org/10.1016/j.jes.2020.04.039
Zhu M, Niu Y, Li Y et al (2020) Low concentrations of tetrabromobisphenol A disrupt Notch signaling and intestinal development in in Vitro and in Vivo models. Chem Res Toxicol. https://doi.org/10.1021/acs.chemrestox.9b00528
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2018YFA0901103) and National Natural Science Foundation of China (21906178; 21876196).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Dong, M., Xiong, Y. et al. Effects of postnatal exposure to tetrabromobisphenol A on testis development in mice and early key events. Arch Toxicol 96, 1881–1892 (2022). https://doi.org/10.1007/s00204-022-03259-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03259-5