Abstract
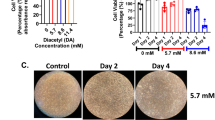
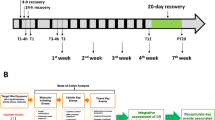
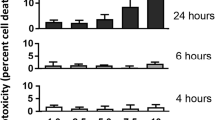
Bronchiolitis obliterans (BO) is a devastating lung disease seen commonly after lung transplant, following severe respiratory tract infection or chemical inhalation exposure. Diacetyl (DA; 2,3-butanedione) is a highly reactive alpha-diketone known to cause BO when inhaled, however, the mechanisms of how inhalation exposure leads to BO development remains poorly understood. In the current work, we combined two clinically relevant models for studying the pathogenesis of DA-induced BO: (1) an in vivo rat model of repetitive DA vapor exposures with recovery and (2) an in vitro model of primary human airway epithelial cells exposed to pure DA vapors. Rats exposed to 5 consecutive days 200 parts-per-million DA 6 h per day had worsening survival, persistent hypoxemia, poor weight gain, and histologic evidence of BO 14 days after DA exposure cessation. At the end of exposure, increased expression of the ubiquitin stress protein ubiquitin-C accumulated within DA-exposed rat lung homogenates and localized primarily to the airway epithelium, the primary site of BO development. Lung proteasome activity increased concurrently with ubiquitin-C expression after DA exposure, supportive of significant proteasome stress. In primary human airway cultures, global proteomics identified 519 significantly modified proteins in DA-exposed samples relative to controls with common pathways of the ubiquitin proteasome system, endosomal reticulum transport, and response to unfolded protein pathways being upregulated and cell–cell adhesion and oxidation–reduction pathways being downregulated. Collectively, these two models suggest that diacetyl inhalation exposure causes abundant protein damage and subsequent ubiquitin proteasome stress prior to the development of chemical-induced BO pathology.







Similar content being viewed by others
Availability of data and material
All proteomics data are uploaded to ftp://massive.ucsd.edu/MSV000086874/, and will be made publically available at the time of publication.
References
Allen JG, Flanigan SS, LeBlanc M, Vallarino J, MacNaughton P, Stewart JH, Christiani DC (2016) Flavoring chemicals in e-cigarettes: diacetyl, 2,3-pentanedione, and acetoin in a sample of 51 products including fruit-, candy-, and cocktail-flavored e-cigarettes. Environ Health Perspect 124:733–739. https://doi.org/10.1289/ehp.1510185
Bailey RL et al (2015) Respiratory morbidity in a coffee processing workplace with sentinel obliterative bronchiolitis cases. Am J Ind Med 58:1235–1245. https://doi.org/10.1002/ajim.22533
Barker AF, Bergeron A, Rom WN, Hertz MI (2014) Obliterative bronchiolitis. N Engl J Med 370:1820–1828. https://doi.org/10.1056/NEJMra1204664
Boylstein R, Piacitelli C, Grote A, Kanwal R, Kullman G, Kreiss K (2006) Diacetyl emissions and airborne dust from butter flavorings used in microwave popcorn production. J Occup Environ Hyg 3:530–535. https://doi.org/10.1080/15459620600909708
Brass DM et al (2017a) The diacetyl-exposed human airway epithelial secretome: new insights into flavoring-induced airways disease. Am J Respir Cell Mol Biol 56:784–795. https://doi.org/10.1165/rcmb.2016-0372OC
Brass DM et al (2017b) The diacetyl-exposed human airway epithelial secretome: new insights into flavoring-induced airways disease. Am J Resp Cell Mol 56:784–795. https://doi.org/10.1165/rcmb.2016-0372OC
Brocker C, Cantore M, Failli P, Vasiliou V (2011) Aldehyde dehydrogenase 7A1 (ALDH7A1) attenuates reactive aldehyde and oxidative stress induced cytotoxicity. Chem Biol Interact 191:269–277. https://doi.org/10.1016/j.cbi.2011.02.016
Chandrakasan G, Hwang CB, Ryder M, Bhatnagar RS (1991) Keratin expression in cultures of adult human epidermal cells. Cell Mol Biol 37:847–852
Clark S, Winter CK (2015) Diacetyl in foods: a review of safety and sensory characteristics. Compr Rev Food Sci F 14:634–643. https://doi.org/10.1111/1541-4337.12150
Colom AJ, Teper AM, Vollmer WM, Diette GB (2006) Risk factors for the development of bronchiolitis obliterans in children with bronchiolitis. Thorax 61:503–506. https://doi.org/10.1136/thx.2005.044909
Cox J, Mann M (2008) MaxQuant enables high peptide identification rates, individualized ppb-range mass accuracies and proteome-wide protein quantification. Nat Biotechnol 26:1367–1372. https://doi.org/10.1038/nbt.1511
Diaz-Villanueva JF, Diaz-Molina R, Garcia-Gonzalez V (2015) Protein folding and mechanisms of proteostasis. Int J Mol Sci 16:17193–17230. https://doi.org/10.3390/ijms160817193
Dudek AZ, Mahaseth H, DeFor TE, Weisdorf DJ (2003) Bronchiolitis obliterans in chronic graft-versus-host disease: analysis of risk factors and treatment outcomes. Biol Blood Marrow Transplant 9:657–666. https://doi.org/10.1016/s1083-8791(03)00242-8
Duling MG, LeBouf RF, Cox-Ganser JM, Kreiss K, Martin SB Jr, Bailey RL (2016) Environmental characterization of a coffee processing workplace with obliterative bronchiolitis in former workers. J Occup Environ Hyg 13:770–781. https://doi.org/10.1080/15459624.2016.1177649
Epler GR (1988) Bronchiolitis obliterans and airways obstruction associated with graft-versus-host disease. Clin Chest Med 9:551–556
Fedan JS, Dowdy JA, Fedan KB, Hubbs AF (2006) Popcorn worker’s lung: in vitro exposure to diacetyl, an ingredient in microwave popcorn butter flavoring, increases reactivity to methacholine. Toxicol Appl Pharmacol 215:17–22. https://doi.org/10.1016/j.taap.2006.02.001
Fennell TR, Morgan DL, Watson SL, Dhungana S, Waidyanatha S (2015) Systemic uptake, albumin and hemoglobin binding of [(14)C]2,3-butanedione administered by intratracheal instillation in male Harlan Sprague Dawley rats and oropharyngeal aspiration in male B6C3F1/N mice. Chem Biol Interact 227:112–119. https://doi.org/10.1016/j.cbi.2014.12.029
Flake GP, Morgan DL (2017) Pathology of diacetyl and 2,3-pentanedione airway lesions in a rat model of obliterative bronchiolitis. Toxicology 388:40–47. https://doi.org/10.1016/j.tox.2016.10.013
Foster MW et al (2017) Proteomic analysis of primary human airway epithelial cells exposed to the respiratory toxicant diacetyl. J Proteome Res 16:538–549. https://doi.org/10.1021/acs.jproteome.6b00672
Ghazanfari T et al (2009) Sardasht-Iran cohort study of chemical warfare victims: design and methods. Arch Iran Med 12:5–14
Goldfarb SB et al (2016) The registry of the international society for heart and lung transplantation: nineteenth pediatric lung and heart-lung transplantation report-2016 focus theme: primary diagnostic indications for transplant. J Heart Lung Transplant 35:1196–1205. https://doi.org/10.1016/j.healun.2016.08.019
Ho HY, Lin YT, Lin G, Wu PR, Cheng ML (2017) Nicotinamide nucleotide transhydrogenase (NNT) deficiency dysregulates mitochondrial retrograde signaling and impedes proliferation. Redox Biol 12:916–928. https://doi.org/10.1016/j.redox.2017.04.035
Hoek JB, Rydstrom J (1988) Physiological roles of nicotinamide nucleotide transhydrogenase. Biochem J 254:1–10. https://doi.org/10.1042/bj2540001
Hubbs AF et al (2002) Necrosis of nasal and airway epithelium in rats inhaling vapors of artificial butter flavoring. Toxicol Appl Pharm 185:128–135. https://doi.org/10.1006/taap.2002.9525
Hubbs AF et al (2008) Respiratory toxicologic pathology of inhaled diacetyl in sprague-dawley rats. Toxicol Pathol 36:330–344. https://doi.org/10.1177/0192623307312694
Hubbs AF et al (2016) Accumulation of ubiquitin and sequestosome-1 implicate protein damage in diacetyl-induced cytotoxicity. Am J Pathol 186:2887–2908. https://doi.org/10.1016/j.ajpath.2016.07.018
Ippolito JA, Palmer L, Spector S, Kane PB, Gorevic PD (1993) Bronchiolitis obliterans organizing pneumonia and rheumatoid arthritis. Semin Arthritis Rheum 23:70–78. https://doi.org/10.1016/s0049-0172(05)80027-7
Kakazu T et al (1990) [A case of bronchiolitis obliterans organizing pneumonia in a patient with rheumatoid arthritis]. Nihon Kyobu Shikkan Gakkai Zasshi 28:493–498
Kelly FL et al (2014) Diacetyl induces amphiregulin shedding in pulmonary epithelial cells and in experimental bronchiolitis obliterans. Am J Respir Cell Mol Biol 51:568–574. https://doi.org/10.1165/rcmb.2013-0339OC
King MS et al (2011) Constrictive bronchiolitis in soldiers returning from Iraq and Afghanistan. N Engl J Med 365:222–230. https://doi.org/10.1056/NEJMoa1101388
Klager S, Vallarino J, MacNaughton P, Christiani DC, Lu Q, Allen JG (2017) Flavoring chemicals and aldehydes in e-cigarette emissions. Environ Sci Technol 51:10806–10813. https://doi.org/10.1021/acs.est.7b02205
Kreiss K, Gomaa A, Kullman G, Fedan K, Simoes EJ, Enright PL (2002) Clinical bronchiolitis obliterans in workers at a microwave-popcorn plant. N Engl J Med 347:330–338. https://doi.org/10.1056/NEJMoa020300
Liu WJ et al (2016) p62 links the autophagy pathway and the ubiqutin-proteasome system upon ubiquitinated protein degradation. Cell Mol Biol Lett 21:29. https://doi.org/10.1186/s11658-016-0031-z
Lund LH et al (2016) The registry of the international society for heart and lung transplantation: thirty-third adult heart transplantation report-2016 focus theme: primary diagnostic indications for transplant. J Heart Lung Transplant 35:1158–1169. https://doi.org/10.1016/j.healun.2016.08.017
Lynch JP 3rd, Weigt SS, DerHovanessian A, Fishbein MC, Gutierrez A, Belperio JA (2012) Obliterative (constrictive) bronchiolitis. Semin Respir Crit Care Med 33:509–532. https://doi.org/10.1055/s-0032-1325161
Marciniak SJ (2017) Endoplasmic reticulum stress in lung disease. Eur Respir Rev 26. https://doi.org/10.1183/16000617.0018-2017
Matsumoto K et al (1996) Bronchiolitis obliterans in a patient with chronic graft-versus-host disease after bone marrow transplantation. Nihon Kyobu Shikkan Gakkai Zasshi 34:345–349
McGraw MD, Rioux JS, Garlick RB, Rancourt RC, White CW, Veress LA (2017) Impaired proliferation and differentiation of the conducting airway epithelium associated with bronchiolitis obliterans after sulfur mustard inhalation injury in rats. Toxicol Sci 157:399–409. https://doi.org/10.1093/toxsci/kfx057
McGraw MD et al (2018) Bronchiolitis obliterans and pulmonary fibrosis after sulfur mustard inhalation in rats. Am J Resp Cell Mol 58:696–705. https://doi.org/10.1165/rcmb.2017-0168OC
McGraw MD, Kim SY, Reed C, Hernady E, Rahman I, Mariani TJ, Finkelstein JN (2020) Airway basal cell injury after acute diacetyl (2,3-butanedione) vapor exposure. Toxicol Lett 325:25–33. https://doi.org/10.1016/j.toxlet.2020.02.012
Meimaridou E et al (2012) Mutations in NNT encoding nicotinamide nucleotide transhydrogenase cause familial glucocorticoid deficiency. Nat Genet 44:740–742. https://doi.org/10.1038/ng.2299
Mercer RR, Russell ML, Roggli VL, Crapo JD (1994) Cell number and distribution in human and rat airways. Am J Respir Cell Mol Biol 10:613–624. https://doi.org/10.1165/ajrcmb.10.6.8003339
Moghieb A et al (2018) Time-resolved proteome profiling of normal lung development. Am J Physiol Lung Cell Mol Physiol 315:L11–L24. https://doi.org/10.1152/ajplung.00316.2017
Moonnumakal SP, Fan LL (2008) Bronchiolitis obliterans in children. Curr Opin Pediatr 20:272–278. https://doi.org/10.1097/MOP.0b013e3282ff62e9
Morgan DL, Flake GP, Kirby PJ, Palmer SM (2008) Respiratory toxicity of diacetyl in C57BL/6 mice. Toxicol Sci 103:169–180. https://doi.org/10.1093/toxsci/kfn016
Morgan DL, Jokinen MP, Price HC, Gwinn WM, Palmer SM, Flake GP (2012) Bronchial and bronchiolar fibrosis in rats exposed to 2,3-pentanedione vapors: implications for bronchiolitis obliterans in humans. Toxicol Pathol 40:448–465. https://doi.org/10.1177/0192623311431946
Morgan DL, Jokinen MP, Johnson CL, Price HC, Gwinn WM, Bousquet RW, Flake GP (2016) Chemical reactivity and respiratory toxicity of the alpha-diketone flavoring agents: 2,3-butanedione, 2,3-pentanedione, and 2,3-hexanedione. Toxicol Pathol 44:763–783. https://doi.org/10.1177/0192623316638962
Morris JB, Hubbs AF (2009) Inhalation dosimetry of diacetyl and butyric acid, two components of butter flavoring vapors. Toxicol Sci 108:173–183. https://doi.org/10.1093/toxsci/kfn222
O’Koren EG, Hogan BL, Gunn MD (2013) Loss of basal cells precedes bronchiolitis obliterans-like pathological changes in a murine model of chlorine gas inhalation. Am J Respir Cell Mol Biol 49:788–797. https://doi.org/10.1165/rcmb.2012-0369OC
Palmer SM et al (2011) Severe airway epithelial injury, aberrant repair and bronchiolitis obliterans develops after diacetyl instillation in rats. PLoS ONE 6:e17644. https://doi.org/10.1371/journal.pone.0017644
Park HR et al (2019) Transcriptomic response of primary human airway epithelial cells to flavoring chemicals in electronic cigarettes. Sci Rep 9:1400. https://doi.org/10.1038/s41598-018-37913-9
Philit F, Wiesendanger T, Archimbaud E, Mornex JF, Brune J, Cordier JF (1995) Post-transplant obstructive lung disease (“bronchiolitis obliterans”): a clinical comparative study of bone marrow and lung transplant patients. Eur Respir J 8:551–558
Piehowski PD, Zhao R, Moore RJ, Clair G, Ansong C (2018) Quantitative proteomic analysis of mass limited tissue samples for spatially resolved tissue profiling. Methods Mol Biol 1788:269–277. https://doi.org/10.1007/7651_2017_78
Pierce JS, Abelmann A, Spicer LJ, Adams RE, Finley BL (2014) Diacetyl and 2,3-pentanedione exposures associated with cigarette smoking: implications for risk assessment of food and flavoring workers. Crit Rev Toxicol 44:420–435. https://doi.org/10.3109/10408444.2014.882292
Rindler TN et al (2017) Alveolar injury and regeneration following deletion of ABCA3. JCI Insight 2(24):e97381. https://doi.org/10.1172/jci.insight.97381
Rock JR et al (2009) Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc Natl Acad Sci USA 106:12771–12775. https://doi.org/10.1073/pnas.0906850106
Rock JR, Randell SH, Hogan BL (2010) Airway basal stem cells: a perspective on their roles in epithelial homeostasis and remodeling. Dis Model Mech 3:545–556. https://doi.org/10.1242/dmm.006031
Rossano JW et al (2016) The registry of the international society for heart and lung transplantation: nineteenth pediatric heart transplantation report-2016; focus theme: primary diagnostic indications for transplant. J Heart Lung Transplant 35:1185–1195. https://doi.org/10.1016/j.healun.2016.08.018
Szema AM, Schmidt MP, Lanzirotti A, Harrington AD, Lyubsky S, Reeder RJ, Schoonen MA (2012) Titanium and iron in lung of a soldier with nonspecific interstitial pneumonitis and bronchiolitis after returning from Iraq. J Occup Environ Med 54:1–2. https://doi.org/10.1097/JOM.0b013e31824327ca
Tyanova S, Temu T, Cox J (2016) The MaxQuant computational platform for mass spectrometry-based shotgun proteomics. Nat Protoc 11:2301–2319. https://doi.org/10.1038/nprot.2016.136
van Thiel RJ, van der Burg S, Groote AD, Nossent GD, Wills SH (1991) Bronchiolitis obliterans organizing pneumonia and rheumatoid arthritis. Eur Respir J 4:905–911
Yusen RD et al (2016) The registry of the international society for heart and lung transplantation: thirty-third adult lung and heart-lung transplant report-2016; focus theme: primary diagnostic indications for transplant. J Heart Lung Transplant 35:1170–1184. https://doi.org/10.1016/j.healun.2016.09.001
Zaccone EJ et al (2015) Diacetyl and 2,3-pentanedione exposure of human cultured airway epithelial cells: ion transport effects and metabolism of butter flavoring agents. Toxicol Appl Pharmacol 289:542–549. https://doi.org/10.1016/j.taap.2015.10.004
Acknowledgements
The authors thank the University of Rochester Medical Center’s Inhalation Exposure Facility, specifically Director Alison Elder, PhD, David Chalupa, MS, and Robert Gelein, MS, for their continued support with diacetyl inhalation exposures. Authors also thank Jon Oldach and Anna Maione with MatTek, Corporation, for support on the project as well as Meghan O’Neil (URMC) for her staining of human samples for Ubq-C. Proteomics experiments were performed in the Environmental Molecular Sciences Laboratory (EMSL), a national scientific user facility sponsored by the Department of Energy (DOE) and located at Pacific Northwest National Laboratory, which is operated by Battelle Memorial Institute for the DOE under Contract DE-AC05-76RL0 1830.
Funding
Grants from the National Institute of Environmental Health Sciences P30 ES001247 (TJM, JNF, MDM) and L40 ES030909-01 (MDM), National Heart Lung and Blood Institute 5R01HL139335 (WJQ, CA, MDM), National Center for Advancing Translational Sciences 2KL2TR001999 (MDM), and the University of Rochester Medical Center’s David H. Smith Fund (MDM).
Author information
Authors and Affiliations
Contributions
Matthew D. McGraw and Jacob N. Finkelstein conceptualized and designed the study; So-Young Kim, Emma House, Heather M. Olson, David Chalupa, and Matthew D. McGraw acquired data for the work; Juan Wang, So-Young Kim, Thomas J Mariani, Wei-Jun Qian, Charles Ansong, Jacob N. Finkelstein and Matthew D. McGraw analyzed and interpreted the findings; Juan Wang and Matthew D. McGraw drafted the manuscript; all authors revised the manuscript for intellectual content, gave final approval for publication and agreed to accountability for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that have no conflicts of interests outside of the funding sources listed on the title page.
Ethics approval
All disclosures of conflicts of interests are listed and verified by each author.
Disclaimer
This article was prepared while Charles Ansong was employed at Pacific Northwest National Laboratory. The opinions expressed in this article are the author's own and do not reflect the view of the National Institutes of Health, the Department of Health and Human Services, or the United States government.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Kim, SY., House, E. et al. Repetitive diacetyl vapor exposure promotes ubiquitin proteasome stress and precedes bronchiolitis obliterans pathology. Arch Toxicol 95, 2469–2483 (2021). https://doi.org/10.1007/s00204-021-03076-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-021-03076-2