Abstract
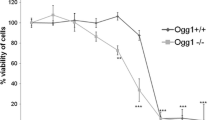
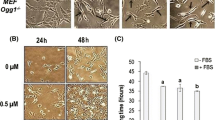
The nanoparticles (NPs) exposure-related oxidative stress is considered among the main causes of the toxic effects induced by these materials. However, the importance of this mechanism has been mostly explored at short term. Previous experience with cells chronically exposed to ZnO and Co NPs hinted to the existence of an adaptative mechanism contributing to the development of oncogenic features. MTH1 is a well-described enzyme expressed exclusively in cancer cells and required to avoid the detrimental consequences of its high prooxidant microenvironment. In the present work, a significantly marked overexpression was found when MTH1 levels were monitored in long-term ZnO and Co NP-exposed cells, a fact that correlates with acquired 2.5-fold and 3.75-fold resistance to the ZnO and Co NPs treatment, respectively. The forced stable inhibition of Mth1 expression by shRNA, followed by 6 additional weeks of exposure, significantly reduced this acquired resistance and sensitized cells to the oxidizing agents H2O2 and KBrO3. When the oncogenic phenotype of Mth1 knock-down cells was evaluated, we found a decrease in several oncogenic markers, including proliferation, anchorage-independent cell growth, and migration and invasion potential. Thus, MTH1 elicits here as a relevant player in the NPs-induced toxicity and carcinogenicity. This study is the first to give a mechanistic explanation for long-term NPs exposure-derived effects. We propose MTH1 as a candidate biomarker to unravel NPs potential genotoxic and carcinogenic effects, as its expression is expected to be elevated only under exposure conditions able to induce DNA damage and the acquisition of an oncogenic phenotype.







Similar content being viewed by others
References
Adamcakova-Dodd A, Stebounova LV, Kim JS, Vorrink SU, Ault AP, O’Shaughnessy PT, Thorne PS (2014) Toxicity assessment of zinc oxide nanoparticles using sub-acute and sub-chronic murine inhalation models. Particle Fibre Toxicol 11(1):1–15. https://doi.org/10.1186/1743-8977-11-15
Alarifi S, Ali D, Suliman AO, Ahamed M, Siddiqui MA, Al-Khedhairy AA (2013) Oxidative stress contributes to cobalt oxide nanoparticles-induced cytotoxicity and DNA damage in human hepatocarcinoma cells. Int J Nanomed 8:189–199. https://doi.org/10.2147/IJN.S37924
Annangi B, Bach J, Vales G, Rubio L, Marcos R, Hernández A (2015) Long-term exposures to low doses of cobalt nanoparticles induce cell transformation enhanced by oxidative damage. Nanotoxicology 9(2):138–147. https://doi.org/10.3109/17435390.2014.900582
Annangi B, Rubio L, Alaraby M, Bach J, Marcos R, Hernández A (2016) Acute and long-term in vitro effects of zinc oxide nanoparticles. Arch Toxicol 90(9):2201–2213. https://doi.org/10.1007/s00204-015-1613-7
Bach J, Peremartí J, Annangi B, Marcos R, Hernández A (2015) Reduced cellular DNA repair capacity after environmentally relevant arsenic exposure. Influence of Ogg1 deficiency. Mutat Res Fundament Mol Mech Mutagen 779:144–151. https://doi.org/10.1016/j.mrfmmm.2015.07.004
Berglund UW, Sanjiv K, Gad H, Kalderén C, Koolmeister T, Pham T, Helleday T (2016) Validation and development of MTH1 inhibitors for treatment of cancer. Ann Oncol 27(12):2275–2283. https://doi.org/10.1093/annonc/mdw429
Buzea C, Pacheco II, Robbie K (2007) Nanomaterials and nanoparticles: sources and toxicity. Biointerphases 2(4):MR17–MR71. https://doi.org/10.1116/1.2815690
Colognato R, Bonelli A, Ponti J, Farina M, Bergamaschi E, Sabbioni E, Migliore L (2008) Comparative genotoxicity of cobalt nanoparticles and ions on human peripheral leukocytes in vitro. Mutagenesis 23(5):377–382. https://doi.org/10.1093/mutage/gen024
Demir E, Akça H, Turna F, Aksakal S, Burgucu D, Kaya B, Marcos R (2015) Genotoxic and cell-transforming effects of titanium dioxide nanoparticles. Environ Res 136:300–308. https://doi.org/10.1016/j.envres.2014.10.032
Eneman JD, Potts RJ, Osier M, Shukla GS, Lee CH, Chiu JF, Hart BA (2000) Suppressed oxidant-induced apoptosis in cadmium adapted alveolar epithelial cells and its potential involvement in cadmium carcinogenesis. Toxicology 147(3):215–228. https://doi.org/10.1016/S0300-483X(00)00215-8
Gad H, Koolmeister T, Jemth AS, Eshtad S, Jacques SA, Ström CE, Helleday T (2014) MTH1 inhibition eradicates cancer by preventing sanitation of the dNTP pool. Nature 508(7495):215–221. https://doi.org/10.1038/nature13181
Ghanbari F, Nasarzadeh P, Seydi E, Ghasemi A, Taghi Joghataei M, Ashtari K, Akbari M (2017) Mitochondrial oxidative stress and dysfunction induced by single- and multiwall carbon nanotubes: a comparative study. J Biomed Mater Res Part A 105(7):2047–2055. https://doi.org/10.1002/jbm.a.36063
Gonzalez L, Lison D, Kirsch-Volders M (2008) Genotoxicity of engineered nanomaterials: a critical review. Nanotoxicology 2(4):252–273. https://doi.org/10.1080/17435390802464986
Guan R, Kang T, Lu F, Zhang Z, Shen H, Liu M (2012) Cytotoxicity, oxidative stress, and genotoxicity in human hepatocyte and embryonic kidney cells exposed to ZnO nanoparticles. Nanoscale Res Lett 7(1):1. https://doi.org/10.1186/1556-276X-7-602
Hirano T (2008) Repair system of 7, 8-dihydro-8-oxoguanine as a defense line against carcinogenesis. J Radiat Res 49(4):329–340. https://doi.org/10.1269/jrr.08049
Johnson BM, Fraietta JA, Gracias DT, Hope JL, Stairiker CJ, Patel PR, Katsikis PD (2015) Acute exposure to ZnO nanoparticles induces autophagic immune cell death. Nanotoxicology 9(6):737–748. https://doi.org/10.3109/17435390.2014.974709
Kettle JG, Alwan H, Bista M, Breed J, Davies NL, Eckersley K, Wylot M (2016) Potent and Selective Inhibitors of MTH1 probe its role in cancer cell survival. J Med Chem 59(6):2346–2361. https://doi.org/10.1021/acs.jmedchem.5b01760
Liu YK, Deng XX, Yang HL (2016) Cytotoxicity and genotoxicity in liver cells induced by cobalt nanoparticles and ions. Bone Joint Res 5(10):461–469. https://doi.org/10.1302/2046-3758.510.bjr-2016-0016.r1
Mateo D, Morales P, Ávalos A, Haza AI (2014) Oxidative stress contributes to gold nanoparticle-induced cytotoxicity in human tumor cells. Toxicol Mech Methods 24(3):161–172. https://doi.org/10.3109/15376516.2013.869783
May S, Hirsch C, Rippl A, Bohmer N, Kaiser JP, Diener L, Wick P (2018) Transient DNA damage following exposure to gold nanoparticles. Nanoscale 10(33):15723–15735. https://doi.org/10.1039/c8nr03612h
Mishra PK, Mishra H, Ekielski A, Talegaonkar S, Vaidya B (2017) Zinc oxide nanoparticles: a promising nanomaterial for biomedical applications. Drug Discovery Today 22(12):1825–1834. https://doi.org/10.1016/j.drudis.2017.08.006
Nakabeppu Y, Ohta E, Abolhassani N (2017) MTH1 as a nucleotide pool sanitizing enzyme: friend or foe? Free Radical Biol Med 107(2016):151–158. https://doi.org/10.1016/j.freeradbiomed.2016.11.002
Oka S, Ohno M, Tsuchimoto D, Sakumi K, Furuichi M, Nakabeppu Y (2008) Two distinct pathways of cell death triggered by oxidative damage to nuclear and mitochondrial DNAs. EMBO J 27(2):421–432. https://doi.org/10.1038/sj.emboj.7601975
Petrocchi A, Leo E, Reyna NJ, Hamilton MM, Shi X, Parker CA, Lewis RT (2016) Identification of potent and selective MTH1 inhibitors. Bioorg Med Chem Lett 26(6):1503–1507. https://doi.org/10.1016/j.bmcl.2016.02.026
Pi J, He Y, Bortner C, Huang J, Liu J, Zhou T, Waalkes MP (2005) Low level, long-term inorganic arsenite exposure causes generalized resistance to apoptosis in cultured human keratinocytes: Potential role in skin co-carcinogenesis. Int J Cancer 116(1):20–26. https://doi.org/10.1002/ijc.20990
Rudramurthy GR, Swamy MK (2018) Potential applications of engineered nanoparticles in medicine and biology: an update. J Biol Inorg Chem. https://doi.org/10.1007/s00775-018-1600-6
Sharma V, Anderson D, Dhawan A (2012) Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells (HepG2). Apoptosis 17(8):852–870. https://doi.org/10.1007/s10495-012-0705-6
Sing T, Shukla S, Kumar P, Wahla V, Bajpai VK, Rather IA (2017) Corrigendum: application of nanotechnology in food science: Perception and overview. Front Microbiol 8:1501. https://doi.org/10.3389/fmicb.2017.01501
Song B, Zhou T, Yang WL, Liu J, Shao LQ (2016) Contribution of oxidative stress to TiO2 nanoparticle-induced toxicity. Environ Toxicol Pharmacol 48:130–140. https://doi.org/10.1016/j.etap.2016.10.013
Srinivasan S, Paknikar K, Bodas D, Gajbhiye V (2018) Applications of cobalt ferrite nanoparticles in biomedical nanotechnology. Nanomedicine. https://doi.org/10.2217/nnm-2017-0379
Tee JK, Ong CN, Bay BH, Ho HK, Leong DT (2016) Oxidative stress by inorganic nanoparticles. Wiley Interdiscip Rev Nanomed Nanobiotechnol 8(3):414–438. https://doi.org/10.1002/wnan.1374
Tu Y, Wang Z, Wang X, Yang H, Zhang P, Johnson M, Cui D (2016) Birth of MTH1 as a therapeutic target for glioblastoma: MTH1 is indispensable for glioma tumorigenesis. Am J Translat Res 8(6):2803–2811
Vales G, Rubio L, Marcos R (2016) Genotoxic and cell-transformation effects of multi-walled carbon nanotubes (MWCNT) following in vitro sub-chronic exposures. J Hazard Mater 306:193–202. https://doi.org/10.1016/j.jhazmat.2015.12.021
Vance ME, Kuiken T, Vejerano EP, McGinnis SP, Hochella MF, Hull DR (2015) Nanotechnology in the real world: redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6(1):1769–1780. https://doi.org/10.3762/bjnano.6.181
Vila L, Marcos R, Hernández A (2017) Long-term effects of silver nanoparticles in caco-2 cells. Nanotoxicology 11(6):771–780. https://doi.org/10.1080/17435390.2017.1355997
Visalli G, Bertuccio MP, Iannazzo D, Piperno A, Pistone A, di Pietro A (2015) Toxicological assessment of multi-walled carbon nanotubes on A549 human lung epithelial cells. Toxicol In Vitro 29(2):352–362. https://doi.org/10.1016/j.tiv.2014.12.004
Yu KN, Yoon TJ, Minai-Tehrani A, Kim JE, Park SJ, Jeong MS, Cho MH (2013) Zinc oxide nanoparticle induced autophagic cell death and mitochondrial damage via reactive oxygen species generation. Toxicol In Vitro 27(4):1187–1195. https://doi.org/10.1016/j.tiv.2013.02.010
Wang JY, Jin L, Yan XG, Sherwin S, Farrelly M, Zhang YY, Zhang XD (2016) Reactive oxygen species dictate the apoptotic response of melanoma cells to TH588. J Investigat Dermatol 136(11):2277–2286. https://doi.org/10.1016/j.jid.2016.06.625
Zhang X, Song W, Zhou Y, Mao F, Lin Y, Guan J, Sun Q (2017) Expression and function of MutT homolog 1 in distinct subtypes of breast cancer. Oncol Lett 13(4):2161–2168. https://doi.org/10.3892/ol.2017.5726
Zhou W, Ma L, Yang J, Qiao H, Li L, Guo Q, Zhao W (2019) Potent and specific MTH1 inhibitors targeting gastric cancer. Cell Death Dis 10(6):434. https://doi.org/10.1038/s41419-019-1665-3
Acknowledgements
We wish to thank Mr. C. Valiente for his technical assistance. This work was supported by the Generalitat de Catalunya [2014SGR-725], Universitat Autònoma de Barcelona [PIF-UAB to I.B. and J.D., Postdoctoral fellowship to B.A.] and the Spanish Ministry of Education and Science [BFU2016-76831-R].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare they have no actual or potential conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barguilla, I., Barszczewska, G., Annangi, B. et al. MTH1 is involved in the toxic and carcinogenic long-term effects induced by zinc oxide and cobalt nanoparticles. Arch Toxicol 94, 1973–1984 (2020). https://doi.org/10.1007/s00204-020-02737-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-020-02737-y