Abstract
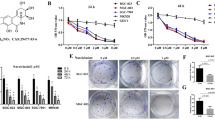
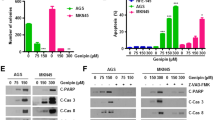
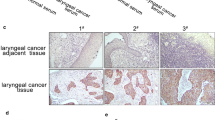
Ochratoxin A (OTA) is a common natural contaminant found in human and animal food worldwide. Our previous work has shown that OTA can cause oxidative DNA damage, G2 arrest and malignant transformation of human gastric epithelium (GES-1) cells. Mitochondria are considered to be target for the action of many cytotoxic agents. However, the role of mitochondria in the cytotoxicity of OTA remains unknown. The aim of this study is to explore the putative role of mitochondria on OTA cytotoxicity by analyzing mitochondrial changes in GES-1 cells. The results showed that OTA treatment (5, 10, 20 µM) for different times caused increases in the production of reactive oxygen species, and induced mitochondrial damage, shown by loss of mitochondrial membrane potential (ΔΨM), and decrease in cellular ATP concentration. Subsequently, the mitochondrial apoptotic pathway was activated, presented by increase of apoptotic rate and activation of apoptotic proteins. Autophagic cell death was also triggered, demonstrated by the conversion of light chain 3B (LC3B)-I to LC3B-II and elevated levels of green fluorescent protein-LC3 (GFP-LC3) puncta. Moreover, Parkin-dependent mitophagy was also activated presented by the colocalization of MitoTracker with LysoTracker or GFP-LC3 puncta. The inhibition of autophagy and mitophagy by inhibitors or siRNA attenuated the toxic effect of OTA on cell growth. Interestingly, OTA treatment also enhanced mitochondrial biogenesis confirmed by activation of AMPK/PGC-1α/TFAM pathway and promoted cell survival. Collectively, the effects of OTA on mitochondria of GES-1 cells are complex. OTA could cause mitochondrial function disturbance, apoptotic and autophagic cell death and also induce mitochondrial biogenesis.






Similar content being viewed by others
References
Adams JM, Cory S (2007) The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 26(9):1324–1337. https://doi.org/10.1038/sj.onc.1210220
Aleo MD, Wyatt RD, Schnellmann RG (1991) Mitochondrial dysfunction is an early event in ochratoxin A but not oosporein toxicity to rat renal proximal tubules. Toxicol Appl Pharmacol 107(1):73–80
Andrzejewski S, Klimcakova E, Johnson RM et al (2017) PGC-1alpha promotes breast cancer metastasis and confers bioenergetic flexibility against metabolic drugs. Cell Metabol 26(5):778–787. https://doi.org/10.1016/j.cmet.2017.09.006 (e5)
Assaf H, Azouri H, Pallardy M (2004) Ochratoxin A induces apoptosis in human lymphocytes through down regulation of Bcl-xL. Toxicol Sci 79(2):335–344. https://doi.org/10.1093/toxsci/kfh123
Atroshi F, Biese I, Saloniemi H et al (2000) Significance of apoptosis and its relationship to antioxidants after ochratoxin A administration in mice. J Pharm Pharm Sci 3(3):281–291
Bhat PV, Pandareesh M, Khanum F, Tamatam A (2016) Cytotoxic effects of Ochratoxin A in neuro-2a cells: role of oxidative stress evidenced by N-acetylcysteine. Front Microbiol 7:1142. https://doi.org/10.3389/fmicb.2016.01142
Bjorkoy G, Lamark T, Johansen T (2006) p62/SQSTM1: a missing link between protein aggregates and the autophagy machinery. Autophagy 2(2):138–139
Bouaziz C, Sharaf el dein O, Martel C et al (2011) Molecular events involved in ochratoxin A induced mitochondrial pathway of apoptosis, modulation by Bcl-2 family members. Environ Toxicol 26(6):579–590. https://doi.org/10.1002/tox.20581
Chen Q, Yu S, Zhang K et al (2018) Exogenous H2S inhibits autophagy in unilateral ureteral obstruction mouse renal tubule cells by regulating the ROS-AMPK signaling pathway. Cell Physiol Biochem 49(6):2200–2213 https://doi.org/10.1159/000493824
Cui J, Xing L, Li Z et al (2010) Ochratoxin A induces G(2) phase arrest in human gastric epithelium GES-1 cells in vitro. Toxicol Lett 193(2):152–158. https://doi.org/10.1016/j.toxlet.2009.12.019
Cui J, Liu J, Wu S et al (2013) Oxidative DNA damage is involved in ochratoxin A-induced G2 arrest through ataxia telangiectasia-mutated (ATM) pathways in human gastric epithelium GES-1 cells in vitro. Arch Toxicol 87(10):1829–1840. https://doi.org/10.1007/s00204-013-1043-3
Deyu H, Luqing C, Xianglian L et al (2018) Protective mechanisms involving enhanced mitochondrial functions and mitophagy against T-2 toxin-induced toxicities in GH3 cells. Toxicol Lett 295:41–53. https://doi.org/10.1016/j.toxlet.2018.05.041
Firdaus F, Zafeer MF, Waseem M, Ullah R, Ahmad M, Afzal M (2018) Thymoquinone alleviates arsenic induced hippocampal toxicity and mitochondrial dysfunction by modulating mPTP in Wistar rats. Biomed Pharmacother 102:1152–1160. https://doi.org/10.1016/j.biopha.2018.03.159
Fulda S, Kogel D (2015) Cell death by autophagy: emerging molecular mechanisms and implications for cancer therapy. Oncogene 34(40):5105–5113. https://doi.org/10.1038/onc.2014.458
Guo X, Dong Y, Yin S et al (2013) Patulin induces pro-survival functions via autophagy inhibition and p62 accumulation. Cell Death Dis 4:e822. https://doi.org/10.1038/cddis.2013.349
Isidoro A, Martinez M, Fernandez PL et al (2004) Alteration of the bioenergetic phenotype of mitochondria is a hallmark of breast, gastric, lung and oesophageal cancer. Biochem J 378(Pt 1):17–20. https://doi.org/10.1042/BJ20031541
Jeon SM, Chandel NS, Hay N (2012) AMPK regulates NADPH homeostasis to promote tumour cell survival during energy stress. Nature 485(7400):661–665. https://doi.org/10.1038/nature11066
Jia X, Cui J, Meng X et al (2016) Malignant transformation of human gastric epithelium cells via reactive oxygen species production and Wnt/beta-catenin pathway activation following 40-week exposure to ochratoxin A. Cancer Lett 372(1):36–47. https://doi.org/10.1016/j.canlet.2015.12.007
Jitschin R, Hofmann AD, Bruns H et al (2014) Mitochondrial metabolism contributes to oxidative stress and reveals therapeutic targets in chronic lymphocytic leukemia. Blood 123(17):2663–2672. https://doi.org/10.1182/blood-2013-10-532200
Ke Y, Ning T, Wang B (1994) Establishment and characterization of a SV40 transformed human fetal gastric epithelial cell line-GES-1. Zhonghua zhong liu za zhi Chin J Oncol 16(1):7–10
Kim SJ, Syed GH, Siddiqui A (2013) Hepatitis C virus induces the mitochondrial translocation of Parkin and subsequent mitophagy. PLoS Pathog 9(3):e1003285. https://doi.org/10.1371/journal.ppat.1003285
Kitamura S, Yanagi T, Imafuku K, Hata H, Abe R, Shimizu H (2017) Drp1 regulates mitochondrial morphology and cell proliferation in cutaneous squamous cell carcinoma. J Dermatol Sci 88(3):298–307. https://doi.org/10.1016/j.jdermsci.2017.08.004
Klionsky DJ, Abdelmohsen K, Abe A et al (2016) Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 12(1):1–222 https://doi.org/10.1080/15548627.2015.1100356
Lee CH, Wu SB, Hong CH et al (2011) Aberrant cell proliferation by enhanced mitochondrial biogenesis via mtTFA in arsenical skin cancers. Am J Pathol 178(5):2066–2076. https://doi.org/10.1016/j.ajpath.2011.01.056
Lemasters JJ (2005) Selective mitochondrial autophagy, or mitophagy, as a targeted defense against oxidative stress, mitochondrial dysfunction, and aging. Rejuvenation Res 8(1):3–5. https://doi.org/10.1089/rej.2005.8.3
Li Z, Zhang X, Cui J, Kang W (2012) Assessment on pollution of Ochratoxin A in grain in China and its apoptosis effect on vitro-cultured human tubular kidney cells. J Biochem Mol Toxicol 26(4):139–146. https://doi.org/10.1002/jbt.20420
Lin CS, Lee HT, Lee SY et al (2012) High mitochondrial DNA copy number and bioenergetic function are associated with tumor invasion of esophageal squamous cell carcinoma cell lines. Int J Mol Sci 13(9):11228–11246. https://doi.org/10.3390/ijms130911228
Liu S, Hartleben B, Kretz O et al (2012) Autophagy plays a critical role in kidney tubule maintenance, aging and ischemia-reperfusion injury. Autophagy 8(5):826–837. https://doi.org/10.4161/auto.19419
Luo Z, Xu X, Sho T et al (2019) ROS-induced autophagy regulates porcine trophectoderm cell apoptosis, proliferation, and differentiation. Am J Physiol Cell Physiol 316(2):C198–C209. https://doi.org/10.1152/ajpcell.00256.2018
Malir F, Ostry V, Pfohl-Leszkowicz A, Malir J, Toman J (2016) Ochratoxin A: 50 years of research. Toxins. https://doi.org/10.3390/toxins8070191
McLaughlin JE, Bin-Umer MA, Tortora A, Mendez N, McCormick S, Tumer NE (2009) A genome-wide screen in Saccharomyces cerevisiae reveals a critical role for the mitochondria in the toxicity of a trichothecene mycotoxin. Proc Natl Acad Sci USA 106(51):21883–21888. https://doi.org/10.1073/pnas.0909777106
Meyer JN, Leuthner TC, Luz AL (2017) Mitochondrial fusion, fission, and mitochondrial toxicity. Toxicology 391:42–53. https://doi.org/10.1016/j.tox.2017.07.019
Narendra D, Tanaka A, Suen DF, Youle RJ (2008) Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J Cell Biol 183(5):795–803. https://doi.org/10.1083/jcb.200809125
Ong SB, Kalkhoran SB, Cabrera-Fuentes HA, Hausenloy DJ (2015) Mitochondrial fusion and fission proteins as novel therapeutic targets for treating cardiovascular disease. Eur J Pharmacol 763(Pt A):104–114. https://doi.org/10.1016/j.ejphar.2015.04.056
Paul S, Jakhar R, Bhardwaj M, Kang SC (2015) Glutathione-S-transferase omega 1 (GSTO1-1) acts as mediator of signaling pathways involved in aflatoxin B1-induced apoptosis-autophagy crosstalk in macrophages. Free Radic Biol Med 89:1218–1230. https://doi.org/10.1016/j.freeradbiomed.2015.11.006
Philley JV, Kannan A, Qin W et al (2016) Complex-I alteration and enhanced mitochondrial fusion are associated with prostate cancer progression. J Cell Physiol 231(6):1364–1374. https://doi.org/10.1002/jcp.25240
Pi H, Xu S, Zhang L et al (2013) Dynamin 1-like-dependent mitochondrial fission initiates overactive mitophagy in the hepatotoxicity of cadmium. Autophagy 9(11):1780–1800. https://doi.org/10.4161/auto.25665
Piao MJ, Ahn MJ, Kang KA et al (2018) Particulate matter 2.5 damages skin cells by inducing oxidative stress, subcellular organelle dysfunction, and apoptosis. Arch Toxicol 92(6):2077–2091. https://doi.org/10.1007/s00204-018-2197-9
Prieto J, Leon M, Ponsoda X et al (2016) Early ERK1/2 activation promotes DRP1-dependent mitochondrial fission necessary for cell reprogramming. Nat Commun 7:11124. https://doi.org/10.1038/ncomms11124
Pryde KR, Smith HL, Chau KY, Schapira AH (2016) PINK1 disables the anti-fission machinery to segregate damaged mitochondria for mitophagy. J Cell Biol 213(2):163–171. https://doi.org/10.1083/jcb.201509003
Qian W, Choi S, Gibson GA, Watkins SC, Bakkenist CJ, Van Houten B (2012) Mitochondrial hyperfusion induced by loss of the fission protein Drp1 causes ATM-dependent G2/M arrest and aneuploidy through DNA replication stress. J Cell Sci 125(23):5745–5757. https://doi.org/10.1242/jcs.109769
Ranaldi G, Caprini V, Sambuy Y, Perozzi G, Murgia C (2009) Intracellular zinc stores protect the intestinal epithelium from Ochratoxin A toxicity. Toxicol In vitro 23(8):1516–1521. https://doi.org/10.1016/j.tiv.2009.08.012
Seibenhener ML, Babu JR, Geetha T, Wong HC, Krishna NR, Wooten MW (2004) Sequestosome 1/p62 is a polyubiquitin chain binding protein involved in ubiquitin proteasome degradation. Mol Cell Biol 24(18):8055–8068. https://doi.org/10.1128/MCB.24.18.8055-8068.2004
Sentelle RD, Senkal CE, Jiang W et al (2012) Ceramide targets autophagosomes to mitochondria and induces lethal mitophagy. Nat Chem Biol 8(10):831–838. https://doi.org/10.1038/nchembio.1059
Shen XL, Zhang B, Liang R et al (2014) Central role of Nix in the autophagic response to ochratoxin A. Food Chem Toxicol 69:202–209. https://doi.org/10.1016/j.fct.2014.04.017
Sheu ML, Shen CC, Chen YS, Chiang CK (2017) Ochratoxin A induces ER stress and apoptosis in mesangial cells via a NADPH oxidase-derived reactive oxygen species-mediated calpain activation pathway. Oncotarget 8(12):19376–19388. https://doi.org/10.18632/oncotarget.14270
Singh KP, Kumari R, Treas J, DuMond JW (2011) Chronic exposure to arsenic causes increased cell survival, DNA damage, and increased expression of mitochondrial transcription factor A (mtTFA) in human prostate epithelial cells. Chem Res Toxicol 24(3):340–349. https://doi.org/10.1021/tx1003112
Suliman HB, Piantadosi CA (2016) Mitochondrial quality control as a therapeutic target. Pharmacol Rev 68(1):20–48. https://doi.org/10.1124/pr.115.011502
Sun Q, Long Z, Wu H et al (2015) Effect of alcohol on diethylnitrosamine-induced hepatic toxicity: Critical role of ROS, lipid accumulation, and mitochondrial dysfunction. Exp Toxicol Pathol 67(10):491–498. https://doi.org/10.1016/j.etp.2015.07.003
Tang Y, Li J, Li F et al (2015) Autophagy protects intestinal epithelial cells against deoxynivalenol toxicity by alleviating oxidative stress via IKK signaling pathway. Free Radic Biol Med 89:944–951. https://doi.org/10.1016/j.freeradbiomed.2015.09.012
van der Merwe KJ, Steyn PS, Fourie L, Scott DB, Theron JJ (1965) Ochratoxin A, a toxic metabolite produced by Aspergillus ochraceus Wilh. Nature 205(976):1112–1113
Wang Y, Zheng W, Bian X et al (2014) Zearalenone induces apoptosis and cytoprotective autophagy in primary Leydig cells. Toxicol Lett 226(2):182–191. https://doi.org/10.1016/j.toxlet.2014.02.003
Wang H, Chen Y, Zhai N et al (2017) Ochratoxin A-induced apoptosis of IPEC-J2 cells through ROS-mediated mitochondrial permeability transition pore opening pathway. J Agric Food Chem 65(48):10630–10637. https://doi.org/10.1021/acs.jafc.7b04434
Wang B, Nie J, Wu L et al (2018) AMPKalpha2 protects against the development of heart failure by enhancing mitophagy via PINK1 phosphorylation. Circ Res 122(5):712–729. https://doi.org/10.1161/CIRCRESAHA.117.312317
Wei YH, Lu CY, Lin TN, Wei RD (1985) Effect of ochratoxin A on rat liver mitochondrial respiration and oxidative phosphorylation. Toxicology 36(2–3):119–130
Wurstle ML, Laussmann MA, Rehm M (2012) The central role of initiator caspase-9 in apoptosis signal transduction and the regulation of its activation and activity on the apoptosome. Exp Cell Res 318(11):1213–1220. https://doi.org/10.1016/j.yexcr.2012.02.013
Xu S, Pi H, Zhang L et al (2016) Melatonin prevents abnormal mitochondrial dynamics resulting from the neurotoxicity of cadmium by blocking calcium-dependent translocation of Drp1 to the mitochondria. J Pineal Res 60(3):291–302. https://doi.org/10.1111/jpi.12310
Yoo SH, Kim HY, Rho JH et al (2015) Targeted inhibition of mitochondrial Hsp90 induces mitochondrial elongation in Hep3B hepatocellular carcinoma cells undergoing apoptosis by increasing the ROS level. Int J Oncol 47(5):1783–1792. https://doi.org/10.3892/ijo.2015.3150
Youle RJ, Narendra DP (2011) Mechanisms of mitophagy. Nat Rev Mol Cell Biol 12(1):9–14. https://doi.org/10.1038/nrm3028
Zhang CS, Lin SC (2016) AMPK promotes autophagy by facilitating mitochondrial fission. Cell Metabol 23(3):399–401. https://doi.org/10.1016/j.cmet.2016.02.017
Zhao CY, Zhang XH, Xue LY et al (2008) Analysis of the changing trends of frequency and localization of gastric cancers arising from different sites of the stomach in population of the high incidence area of esophageal and gastric cancers in Hebei province. Zhonghua zhong liu za zhi Chin J Oncol 30(11):817–820
Acknowledgements
Thanks very much for the support of Engineering Research Center for Cancer Biomedical and Translational Medicine in Southwest University and Chongqing Engineering and Technology Research Center for Silk Biomaterials and Regenerative Medicine in Southwest University. This work was supported by the Key Research and Development Program of Hebei Provence (Grant no. 17277784D), the Natural Science Foundation of Hebei Provence for the Youth (Grant no. H2012206053), and the Major Medical Research Projects in Hebei Province (Grant no. 20180412).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Q., Dong, Z., Lian, W. et al. Ochratoxin A causes mitochondrial dysfunction, apoptotic and autophagic cell death and also induces mitochondrial biogenesis in human gastric epithelium cells. Arch Toxicol 93, 1141–1155 (2019). https://doi.org/10.1007/s00204-019-02433-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-019-02433-6