Abstract
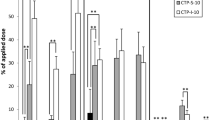
Polycyclic aromatic hydrocarbons (PAH) are ubiquitous pollutants, among which benzo[a]pyrene (B[a]P) is the only compound classified carcinogenic to humans. Besides pulmonary uptake, skin is the major route of PAH absorption during occupational exposure. Health risk due to PAH exposure is commonly assessed among workers using biomonitoring. A realistic human ex vivo skin model was developed to explore B[a]P diffusion and metabolism to determine the most relevant biomarker following dermal exposure. Three realistic doses (0.88, 8.85 and 22.11 nmol/cm2) were topically applied for 8, 24, and 48 h. B[a]P and its metabolites were quantified by liquid chromatography coupled with fluorimetric detection. The impact of time, applied dose, and donor age were estimated using a linear mixed-effects model. B[a]P vastly penetrated the skin within 8 h. The major metabolites were 3-hydroxybenzo[a]pyrene (3-OHB[a]P) and 7,8,9,10-tetrahydroxy-7,8,9,10-tetrahydrobenzo[a]pyrene (B[a]P-tetrol). This latter predominantly derives from the most carcinogenic metabolite of B[a]P, benzo[a]pyrene-7,8-diol-9,10-epoxide (BPDE), as well as benzo[a]pyrene-9,10-diol-7,8-epoxide (reverse-BPDE). Benzo[a]pyrene-trans-7,8-dihydrodiol (B[a]P-7,8-diol) was a minor metabolite, and benzo[a]pyrene-trans-4,5-dihydrodiol (B[a]P-4,5-diol) was never quantified. Unmetabolized B[a]P bioavailability was limited following dermal exposure since less than 3% of the applied dose could be measured in the culture medium. B[a]P was continuously absorbed and metabolized by human skin over 48 h. B[a]P-tetrol production became saturated as the applied dose increased, while no effect was measured on the other metabolic pathways. Age had a slight positive effect on B[a]P absorption and metabolism. This work supports the relevance of B[a]P-tetrol to assess occupational exposure and carcinogenic risk after cutaneous absorption of B[a]P.




Similar content being viewed by others
References
Aklillu E, Øvrebø S, Botnen IV, Otter C, Ingelman-Sundberg M (2005) Characterization of common CYP1B1 variants with different capacity for Benzo[<em>a</em>]pyrene-7,8-dihydrodiol epoxide formation from Benzo[<em>a</em>]pyrene. Cancer Res 65(12):5105–5111 https://doi.org/10.1158/0008-5472.can-05-0113
Andersson JT, Achten C (2015) Time to Say goodbye to the 16 EPA PAHs? Toward an up-to-date use of PACs for environmental purposes. Polycyclic Aromat Compd 35(2–4):330–354. https://doi.org/10.1080/10406638.2014.991042
Barbeau D, Lutier S, Choisnard L, Marques M, Persoons R, Maitre A (2017) Urinary trans-anti-7,8,9,10-tetrahydroxy-7,8,9,10-tetrahydrobenzo(a)pyrene as the most relevant biomarker for assessing carcinogenic polycyclic aromatic hydrocarbons exposure. Environ Int 112:147–155. https://doi.org/10.1016/j.envint.2017.12.012
Barbeau D, Maitre A, Marques M (2011) Highly sensitive routine method for urinary 3-hydroxybenzo[a]pyrene quantitation using liquid chromatography-fluorescence detection and automated off-line solid phase extraction. Analyst 136(6):1183–1191. https://doi.org/10.1039/C0AN00428F
Bickers DR, Kappas A (1978) Human skin aryl hydrocarbon hydroxylase. Induction by coal tar. J Clin Invest 62(5):1061–1068. https://doi.org/10.1172/JCI109211
Boffetta P, Jourenkova N, Gustavsson P (1997) Cancer risk from occupational and environmental exposure to polycyclic aromatic hydrocarbons. Cancer Causes Control 8(3):444–472. https://doi.org/10.1023/a:1018465507029
Bouchard M, Viau C (1996) Urinary excretion kinetics of Pyrene and Benzo(a)pyrene metabolites following intravenous administration of the parent compounds or the metabolites. Toxicol Appl Pharmacol 139(2):301–309. https://doi.org/10.1006/taap.1996.0169
Brinkmann J, Stolpmann K, Trappe S et al (2013) Metabolically competent human skin models: activation and genotoxicity of benzo[a]pyrene. Toxicol Sci 131(2):351–359. https://doi.org/10.1093/toxsci/kfs316
Bronaugh RL, Maibach HI (2005) Percutaneous absorption: drugs, cosmetics, mechanisms, methods. CRC Press, Boca Raton
Bronaugh RL, Stewart RF (1985) Methods for in vitro percutaneous absorption studies IV: the flow-through diffusion cell. J Pharm Sci 74(1):64–67
Bronaugh RL, Stewart RF (1986) Methods for in vitro percutaneous absorption studies VI: preparation of the Barrier layer. J Pharm Sci 75(5):487–491. https://doi.org/10.1002/jps.2600750513
Cavet M, Leonard M (2013) Les expositions aux produits chimiques cancérogènes en 2010. Dares Anal 54:9
Cheung C, Smith CK, Hoog J-O, Hotchkiss SAM (1999) Expression and localization of human alcohol and aldehyde dehydrogenase enzymes in skin. Biochem Biophys Res Commun 261(1):100–107. https://doi.org/10.1006/bbrc.1999.0943
Chu I, Dick D, Bronaugh R, Tryphonas L (1996) Skin reservoir formation and bioavailability of dermally administered chemicals in hairless guinea pigs. Food Chem Toxicol 34(3):267–276. https://doi.org/10.1016/0278-6915(95)00112-3
Farahmand S, Maibach HI (2009) Transdermal drug pharmacokinetics in man: interindividual variability and partial prediction. Int J Pharma 367(1–2):1–15. https://doi.org/10.1016/j.ijpharm.2008.11.020
Fernando S, Shaw L, Shaw D et al (2016) Evaluation of firefighter exposure to wood smoke during training exercises at burn houses. Environ Sci Technol 50(3):1536–1543. https://doi.org/10.1021/acs.est.5b04752
Flaten GE, Palac Z, Engesland A, Filipović-Grčić J, Vanić Ž, Škalko-Basnet N (2015) In vitro skin models as a tool in optimization of drug formulation. Eur J Pharm Sci 75:10–24. https://doi.org/10.1016/j.ejps.2015.02.018
Fustinoni S, Campo L, Cirla PE et al (2010) Dermal exposure to polycyclic aromatic hydrocarbons in asphalt workers. Occup Environ Med 67(7):456–463. https://doi.org/10.1136/oem.2009.050344
Förster K, Preuss R, Roßbach B, Brüning T, Angerer J, Simon P (2008) 3-Hydroxybenzo[a]pyrene in the urine of workers with occupational exposure to polycyclic aromatic hydrocarbons in different industries. Occup Environ Med 65(4):224–229. https://doi.org/10.1136/oem.2006.030809
Genies C, Maître A, Lefèbvre E, Jullien A, Chopard-Lallier M, Douki T (2013) The extreme variety of genotoxic response to Benzo[a]pyrene in three different human cell lines from three different organs. PloS one 8(11):e78356. https://doi.org/10.1371/journal.pone.0078356
Hecht SS, Carmella SG, Villalta PW, Hochalter JB (2010) Analysis of phenanthrene and Benzo[a]pyrene tetraol enantiomers in human urine: relevance to the bay region diol epoxide hypothesis of Benzo[a]pyrene carcinogenesis and to biomarker studies. Chem Res Toxicol 23(5):900–908. https://doi.org/10.1021/tx9004538
Hewitt NJ, Edwards RJ, Fritsche E et al (2013) Use of human in vitro skin models for accurate and ethical risk assessment: metabolic considerations. Toxicol Sci 133(2):209–217. https://doi.org/10.1093/toxsci/kft080
Hopf NB, Spring P, Hirt-Burri N et al (2018) Polycyclic aromatic hydrocarbons (PAHs) skin permeation rates change with simultaneous exposures to solar ultraviolet radiation (UV-S). Toxicol Lett 287:122–130. https://doi.org/10.1016/j.toxlet.2018.01.024
IARC (2010) Some non-heterocyclic polycyclic aromatic hydrocarbons and some related exposures. IARC monographs on the evaluation of carcinogenic risks to humans/World Health Organization. Int Agency Res Cancer 92:1–853
Ibrahim SA, Li SK (2009) Effects of solvent deposited enhancers on transdermal permeation and their relationship with Emax. J Controlled Release 136(2):117–124. https://doi.org/10.1016/j.jconrel.2009.01.023
Jacques C, Perdu E, Duplan H et al (2010) Disposition and biotransformation of 14C-Benzo(a)pyrene in a pig ear skin model: ex vivo and in vitro approaches. Toxicol Lett 199(1):22–33. https://doi.org/10.1016/j.toxlet.2010.08.001
Jongeneelen FJ (2001) Benchmark guideline for urinary 1-hydroxypyrene as biomarker of occupational exposure to polycyclic aromatic hydrocarbons. Ann Occup Hyg 45(1):3–13. https://doi.org/10.1093/annhyg/45.1.3
Kammer R, Tinnerberg H, Eriksson K (2011) Evaluation of a tape-stripping technique for measuring dermal exposure to pyrene and benzo(a)pyrene. J Environ Monit 13(8):2165–2171. https://doi.org/10.1039/C1EM10245A
Kao J, Patterson FK, Hall J (1985) Skin penetration and metabolism of topically applied chemicals in six mammalian species, including man: an in vitro study with benzo[a]pyrene and testosterone. Toxicol Appl Pharmacol 81(3 Pt 1):502–516
Kontir DM, Glance CA, Colby HD, Miles PR (1986) Effects of organic solvent vehicles on benzo[a]pyrene metabolism in rabbit lung microsomes. Biochem Pharmacol 35(15):2569–2575. https://doi.org/10.1016/0006-2952(86)90055-9
Lehman PA, Raney SG, Franz TJ (2011) Percutaneous absorption in man: in vitro-in vivo correlation. Skin Pharmacol Physiol 24(4):224–230
Liddle C, Goodwin BJ, George J, Tapner M, Farrell GC (1998) Separate and interactive regulation of cytochrome P450 3A4 by triiodothyronine, dexamethasone, and growth hormone in cultured hepatocytes1. J Clin Endocrinol Metabol 83(7):2411–2416. https://doi.org/10.1210/jcem.83.7.4877
Lutier S, Maître A, Bonneterre V et al (2016) Urinary elimination kinetics of 3-hydroxybenzo(a)pyrene and 1-hydroxypyrene of workers in a prebake aluminum electrode production plant: Evaluation of diuresis correction methods for routine biological monitoring. Environ Res 147:469–479. https://doi.org/10.1016/j.envres.2016.02.035
Mcclean M, Rinehart R, Herrick R (2006) Dermal exposure and urinary 1-hydroxypyrene among asphalt roofing workers. Epidemiology 17(6):S134–S135
Moody RP, Nadeau B, Chu I (1995) In vivo and in vitro dermal absorption of benzo[a]pyrene in rat, guinea pig, human and tissue-cultured skin. J Dermatol Sci 9(1):48–58
Moody RP, Yip A, Chu I, Glycol E (2009) Effect of cold storage on in vitro human skin absorption of six 14C-radiolabeled environmental contaminants: Benzo[a]Pyrene. methyl parathion, naphthalene, nonyl phenol, and toluene. J Toxicol Environ Health Part A 72(8):505–517. https://doi.org/10.1080/15287390802328713
Moorthy B, Chu C, Carlin DJ (2015) Polycyclic aromatic hydrocarbons: from metabolism to lung cancer. Toxicol Sci 145(1):5–15. https://doi.org/10.1093/toxsci/kfv040
Moreau M, Bouchard M (2014) Comparison of the kinetics of various biomarkers of benzo[a]pyrene exposure following different routes of entry in rats. J Appl Toxicol 35(7):781–790. https://doi.org/10.1002/jat.3070
Ng KM, Chu I, Bronaugh RL, Franklin CA, Somers DA (1992) Percutaneous absorption and metabolism of pyrene, benzo[a]pyrene, and di(2-ethylhexyl) phthalate: comparison of in vitro and in vivo results in the hairless guinea pig. Toxicol Appl Pharmacol 115(2):216–223
Ngo MA, Maibach HI (2010) Dermatotoxicology: Historical perspective and advances. Toxicol Appl Pharmacol 243(2):225–238. https://doi.org/10.1016/j.taap.2009.12.008
Payan JP, Lafontaine M, Simon P et al (2009) 3-Hydroxybenzo(a)pyrene as a biomarker of dermal exposure to benzo(a)pyrene. Arch Toxicol 83(9):873–883. https://doi.org/10.1007/s00204-009-0440-0
Penning TM (2014) Human Aldo-Keto reductases and the metabolic activation of polycyclic aromatic hydrocarbons. Chem Res Toxicol 27(11):1901–1917. https://doi.org/10.1021/tx500298n
Pinheiro JC, Bates DM (2000) Mixed-effects models in S and S-PLUS. Springer, New York, pp 528
Plakunov I, Smolarek TA, Fischer DL, Wiley JC Jr, Baird WM (1987) Separation by ion-pair high-performance liquid chromatography of the glucuronide, sulfate and glutathione conjugates formed from benzo[a]pyrene in cell cultures from rodents, fish and humans. Carcinogenesis 8(1):59–66
van de Sandt JJM, van Burgsteden JA, Cage S et al (2004) In vitro predictions of skin absorption of caffeine, testosterone, and benzoic acid: a multi-centre comparison study. Regul Toxicol Pharmacol 39(3):271–281. https://doi.org/10.1016/j.yrtph.2004.02.004
Sartorelli P, Aprea C, Cenni A et al (1998) Prediction of percutaneous absorption from physicochemical data: a model based on data of in vitro experiments. Ann Occup Hyg 42(4):267–276
Schwarz D, Kisselev P, Cascorbi I, Schunck WH, Roots I (2001) Differential metabolism of benzo[a]pyrene and benzo[a]pyrene-7,8-dihydrodiol by human CYP1A1 variants. Carcinogenesis 22(3):453–459
Shimada T (2006) Xenobiotic-metabolizing enzymes involved in activation and detoxification of carcinogenic polycyclic aromatic hydrocarbons. Drug Metabol Pharmacokinet 21(4):257–276
Sobus JR, McClean MD, Herrick RF et al (2009) Comparing urinary biomarkers of airborne and dermal exposure to polycyclic aromatic compounds in asphalt-exposed workers. Ann Occup Hyg 53(6):561–571. https://doi.org/10.1093/annhyg/mep042
Storm JE, Collier SW, Stewart RF, Bronaugh RL (1990) Metabolism of xenobiotics during percutaneous penetration: role of absorption rate and cutaneous enzyme activity. Fundam Appl Toxicol 15(1):132–141
VanRooij JG, Bodelier-Bade MM, Jongeneelen FJ (1993a) Estimation of individual dermal and respiratory uptake of polycyclic aromatic hydrocarbons in 12 coke oven workers. Br J Ind Med 50(7):623–632
VanRooij JGM, De Roos JHC, Bodelier-Bade MM, Jongeneelen FJ (1993b) Absorption of polycyclic aromatic hydrocarbons through human skin: differences between anatomical sites and individuals. J Toxicol Environ Health 38(4):355–368. https://doi.org/10.1080/15287399309531724
VanRooij JGM, Bodelier-Bade M, De Looff M, Dijkmans AJA, Jongeneelen APGF (1992) Dermal exposure to polycyclic aromatic hydrocarbons among primary aluminium workers. La Medicina del lavoro 83(5):519–529
Xue W, Warshawsky D (2005) Metabolic activation of polycyclic and heterocyclic aromatic hydrocarbons and DNA damage: a review. Toxicol Appl Pharmacol 206(1):73–93. https://doi.org/10.1016/j.taap.2004.11.006
Zanger UM, Schwab M (2013) Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther 138(1):103–141. https://doi.org/10.1016/j.pharmthera.2012.12.007
Zhong Y, Carmella SG, Hochalter JB, Balbo S, Hecht SS (2011) Analysis of r-7,t-8,9,c-10-Tetrahydroxy-7,8,9,10-tetrahydrobenzo[a]pyrene in Human Urine: a biomarker for directly assessing carcinogenic polycyclic aromatic hydrocarbon exposure plus metabolic activation. Chem Res Toxicol 24(1):73–80. https://doi.org/10.1021/tx100287n
Acknowledgements
This work was funded by the French National Institute of Health and Medical Research (INSERM) (Grant Number ENV201412) and Plan Cancer. The authors wish to thank the team “Service de Chirurgie Plastique et Maxillo-faciale CHU Grenoble Alpes” for their help in skin sample collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Experiments were conducted in accordance with the article L1245-2 of the French Public Health Code on the use of surgical wastes for research purposes. Collection, storage, and use of human skin samples were made anonymously, declared to the French authorities, and validated in the CODECOH DC-2008-444 document.
Informed consent
Informed consent was obtained from all skin donors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bourgart, E., Barbeau, D., Marques, M. et al. A realistic human skin model to study benzo[a]pyrene cutaneous absorption in order to determine the most relevant biomarker for carcinogenic exposure. Arch Toxicol 93, 81–93 (2019). https://doi.org/10.1007/s00204-018-2329-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2329-2