Abstract
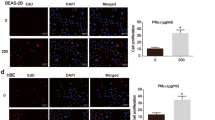
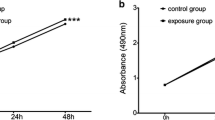
Exposure to particulate matter (PM) has been related to the onset of adverse health effects including lung cancer, but the underlying molecular mechanisms are still under investigation. Epithelial-to-mesenchymal transition (EMT) is regarded as a crucial step in cancer progression. In a previous study, we reported EMT-related responses in the human bronchial epithelial cell line HBEC3-KT, exposed to Milan airborne winter PM2.5. We also found a strong modulation of SERPINB2, encoding for the PAI-2 protein and previously suggested to play an important role in cancer. Here we investigate the role of SERPINB2/PAI-2 in the regulation of EMT-related effects induced by PM exposure in HBEC3-KT. PM exposure (up to 10 µg/cm2) increased SERPINB2 expression, reduced cell migration and induced morphological alterations in HBEC3-KT. Changes in actin structure and cadherin-1 relocalization were observed in PM-exposed samples. Knockdown of SERPINB2 by siRNA down-regulated the CDH1 gene expression, as well as PAI-2 and cadherin-1 protein expression. SERPINB2 knockdown also increased cell migration rate, and counteracted the PM-induced reduction of cell migration and alteration of cell morphology. SERPINB2 was found to be greatly down-regulated in a HBEC2-KT transformed cell line, supporting the importance of this gene in the regulation of EMT. In conclusion, here we show that PAI-2 regulates CDH1 gene/cadherin-1 protein expression in bronchial HBEC3-KT cells, and this mechanism might be involved in the regulation of cell migration. SERPINB2 down-regulation should be considered part of EMT, and the over-expression of SERPINB2 in PM-exposed samples might be interpreted as an initial protective mechanism.








Similar content being viewed by others
References
Bersaas A, Arnoldussen YJ, Sjøberg M et al (2016) Epithelial-mesenchymal transition and FOXA genes during tobacco smoke carcinogen induced transformation of human bronchial epithelial cells. Toxicol In Vitro 35:55–65. https://doi.org/10.1016/j.tiv.2016.04.012
Borm B, Requardt RP, Herzog V, Kirfel G (2005) Membrane ruffles in cell migration: indicators of inefficient lamellipodia adhesion and compartments of actin filament reorganization. Exp Cell Res 302:83–95. https://doi.org/10.1016/j.yexcr.2004.08.034
Brandhagen BN, Tieszen CR, Ulmer TM et al (2013) Cytostasis and morphological changes induced by mifepristone in human metastatic cancer cells involve cytoskeletal filamentous actin reorganization and impairment of cell adhesion dynamics. BMC Cancer 13:35. https://doi.org/10.1186/1471-2407-13-35
Cassee FR, Héroux M-E, Gerlofs-Nijland ME, Kelly FJ (2013) Particulate matter beyond mass: recent health evidence on the role of fractions, chemical constituents and sources of emission. Inhal Toxicol 25:802–812. https://doi.org/10.3109/08958378.2013.850127
Chirino YI, García-Cuellar CM, García-García C et al (2017) Airborne particulate matter in vitro exposure induces cytoskeleton remodeling through activation of the ROCK-MYPT1-MLC pathway in A549 epithelial lung cells. Toxicol Lett 272:29–37. https://doi.org/10.1016/j.toxlet.2017.03.002
Croucher DR, Saunders DN, Lobov S, Ranson M (2008) Revisiting the biological roles of PAI2 (SERPINB2) in cancer. Nat Rev Cancer 8:535–545. https://doi.org/10.1038/nrc2400
Figueiredo J, Simões-Correia J, Söderberg O et al (2011) ADP-ribosylation factor 6 mediates E-cadherin recovery by chemical chaperones. PLoS One 6:e23188. https://doi.org/10.1371/journal.pone.0023188
Izaguirre MF, Casco VH (2016) E-cadherin roles in animal biology: A perspective on thyroid hormone-influence. Cell Commun Signal 14:27. https://doi.org/10.1186/s12964-016-0150-1
LaGier AJ, Manzo ND, Dye JA (2013) Diesel exhaust particles induce aberrant alveolar epithelial directed cell movement by disruption of polarity mechanisms. J Toxicol Environ Health A 76:71–85. https://doi.org/10.1080/15287394.2013.738169
Lewtas J (2007) Air pollution combustion emissions: characterization of causative agents and mechanisms associated with cancer, reproductive, and cardiovascular effects. Mutat Res 636:95–133
Liu Y, Yin T, Feng Y et al (2015) Mammalian models of chemically induced primary malignancies exploitable for imaging-based preclinical theragnostic research. Quant Imaging Med Surg 5:708–729
Longhin E, Holme JA, Gutzkow KB et al (2013) Cell cycle alterations induced by urban PM2.5 in bronchial epithelial cells: characterization of the process and possible mechanisms involved. Part Fibre Toxicol 10:63
Longhin E, Capasso L, Battaglia C et al (2016a) Integrative transcriptomic and protein analysis of human bronchial BEAS-2B exposed to seasonal urban particulate matter. Environ Pollut 209:87–98
Longhin E, Gualtieri M, Capasso L et al (2016b) Physico-chemical properties and biological effects of diesel and biomass particles. Environ Pollut 215:1–10. https://doi.org/10.1016/j.envpol.2016.05.015
Luanpitpong S, Chen M, Knuckles T et al (2014) Appalachian mountaintop mining particulate matter induces neoplastic transformation of human bronchial epithelial cells and promotes tumor formation. Environ Sci Technol 48:12912–12919
Mantecca P, Gualtieri M, Longhin E et al (2012) Adverse biological effects of Milan urban PM looking for suitable molecular markers of exposure. Chem Ind Chem Eng Q 18:635–641. https://doi.org/10.2298/CICEQ120206114M
Melo S, Figueiredo J, Fernandes M et al (2017) Predicting the Functional Impact of CDH1 Missense Mutations in Hereditary Diffuse Gastric Cancer. Int J Mol Sci 18:2687. https://doi.org/10.3390/ijms18122687
Ramirez RD, Sheridan S, Girard L et al (2004) Immortalization of human bronchial epithelial cells in the absence of viral oncoproteins. Cancer Res 64:9027–9034
Ramnefjell M, Aamelfot C, Helgeland L, Akslen LA (2017) Low expression of SerpinB2 is associated with reduced survival in lung adenocarcinomas. Oncotarget 8:90706–90718. https://doi.org/10.18632/oncotarget.21456
Schaeffer D, Somarelli JA, Hanna G et al (2014) Cellular migration and invasion uncoupled: increased migration is not an inexorable consequence of epithelial-to-mesenchymal transition. Mol Cell Biol 34:3486–3499. https://doi.org/10.1128/MCB.00694-14
Schroder WA, Major LD, Le TT et al (2014) Tumor cell-expressed SerpinB2 is present on microparticles and inhibits metastasis. Cancer Med 3:500–513
Tseng C-Y, Chang J-F, Wang J-S et al (2015) Protective effects of N-acetyl cysteine against diesel exhaust particles-induced intracellular ROS generates pro-inflammatory cytokines to mediate the vascular permeability of capillary-like endothelial tubes. PLoS One 10:e0131911. https://doi.org/10.1371/journal.pone.0131911
Valavanidis A, Vlachogianni T, Fiotakis K, Loridas S (2013) Pulmonary oxidative stress, inflammation and cancer: respirable particulate matter, fibrous dusts and ozone as major causes of lung carcinogenesis through reactive oxygen species mechanisms. Int J Environ Res Public Health 10:3886–3907. https://doi.org/10.3390/ijerph10093886
Wei H, Liang F, Cheng W et al (2017) The mechanisms for lung cancer risk of PM2.5: induction of epithelial-mesenchymal transition and cancer stem cell properties in human non-small cell lung cancer cells. Environ Toxicol 32:2341–2351. https://doi.org/10.1002/tox.22437
Wu C, Zhu W, Qian J et al (2013) WT1 promotes invasion of NSCLC via suppression of CDH1. J Thorac Oncol 8:1163–1169. https://doi.org/10.1097/JTO.0b013e31829f6a5f
Xiao D, He J (2010a) Epithelial mesenchymal transition and lung cancer. J Thorac Dis 2:154–159. https://doi.org/10.3978/j.issn.2072-1439.2010.02.03.7
Xiao D, He J (2010b) Epithelial mesenchymal transition and lung cancer. J Thorac Dis 2:154–159
Yang D, Ma M, Zhou W et al (2017) Inhibition of miR-32 activity promoted EMT induced by PM2.5 exposure through the modulation of the Smad1-mediated signaling pathways in lung cancer cells. Chemosphere 184:289–298. https://doi.org/10.1016/j.chemosphere.2017.05.152
Yue H, Yun Y, Gao R, Li G, Sang N (2015) Winter polycyclic aromatic hydrocarbon-bound particulate matter from Peri-urban North China promotes lung cancer cell Metastasis. Environ Sci Technol 49(24):14484–14493. https://doi.org/10.1021/es506280c
Zhang S, Zou L, Yang T et al (2015) The sGC activator inhibits the proliferation and migration, promotes the apoptosis of human pulmonary arterial smooth muscle cells via the up regulation of plasminogen activator inhibitor-2. Exp Cell Res 332:278–287. https://doi.org/10.1016/j.yexcr.2015.02.006
Acknowledgements
The authors want to thank Øivind Skare for the support with the statistical analyses of the migration data.
Funding
This study has been supported by the grants of the Italian Ministry of Foreign Affairs and International Cooperation (MAECI) to PM (Proj. ID PGR00786).
Author information
Authors and Affiliations
Contributions
EL performed the experiments and wrote the manuscript. EL and SM discussed and interpreted the findings. EL, SM and PM designed the project. SM, PM and MC supervised the project and revised the manuscript. AB established the transformed cell line T2KT-CSC-H. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Longhin, E., Camatini, M., Bersaas, A. et al. The role of SerpinB2 in human bronchial epithelial cells responses to particulate matter exposure. Arch Toxicol 92, 2923–2933 (2018). https://doi.org/10.1007/s00204-018-2259-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2259-z