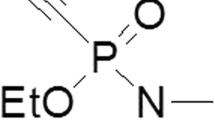
Abstract
Despite six decades of extensive research in medical countermeasures against nerve agent poisoning, a broad spectrum acetylcholinesterase (AChE) reactivator is not yet available. One current approach is directed toward synthesizing oximes with high affinity and reactivatability toward butyrylcholinesterase (BChE) in plasma to generate an effective pseudocatalytic scavenger. An interim solution could be the administration of external AChE or BChE from blood products to augment pseudocatalytic scavenging with slower but clinically approved oximes to decrease nerve agent concentrations in the body. We here semiquantitatively investigate the ability of obidoxime and HI-6 to decrease the inhibitory activity of VX with human AChE and BChE from whole blood, erythrocyte membranes, erythrocytes, plasma, clinically available fresh frozen plasma and packed red blood cells. The main findings are that whole blood showed a VX concentration-dependent decrease in inhibitory activity with HI-6 being more potent than obidoxime. Using erythrocytes and erythrocyte membranes again, HI-6 was more potent compared to obidoxime. With freshly prepared plasma, obidoxime and HI-6 showed comparable results for the decrease in VX. The use of the clinically available blood products revealed that packed red blood cells showed similar kinetics as fresh erythrocytes. Fresh frozen plasma resulted in a slower and incomplete decrease in inhibitory plasma compared to freshly prepared plasma. In conclusion, the administration of blood products in combination with available oximes augments pseudocatalytic scavenging and might be useful to decrease the body load of persistent, highly toxic nerve agents.





Similar content being viewed by others
References
Aldridge WN, Reiner E (1972) Enzyme inhibitors as substrates: interactions of esterases with esters of organophosphorus and carbamic acids. North-Holland, Amsterdam
Augustinsson KB (1955) The normal variation of human blood cholinesterase activity. Acta Physiol Scand 35:40–52. doi:10.1111/j.1748-1716.1955.tb01262.x
Aurbek N, Thiermann H, Eyer F et al (2009) Suitability of human butyrylcholinesterase as therapeutic marker and pseudo catalytic scavenger in organophosphate poisoning: a kinetic analysis. Toxicology 259:133–139. doi:10.1016/j.tox.2009.02.014
Clement JG, Bailey DG, Madill HD et al (1995) The acetylcholinesterase oxime reactivator HI-6 in man: pharmacokinetics and tolerability in combination with atropine. Biopharm Drug Dispos 16:415–425
Dolgin E (2013) Syrian gas attack reinforces need for better anti-sarin drugs. Nat Med 19:1194–1195. doi:10.1038/nm1013-1194
Eisenkraft A, Gilburd D, Kassirer M, Kreiss Y (2014) What can we learn on medical preparedness from the use of chemical agents against civilians in Syria? Am J Emerg Med 32:186. doi:10.1016/j.ajem.2013.11.005
Elsinghorst PW, Worek F, Thiermann H, Wille T (2013) Drug development for the management of organophosphorus poisoning. Expert Opin Drug Discov 8:1467–1477. doi:10.1517/17460441.2013.847920
Herkenhoff S, Szinicz L, Rastogi VK et al (2004) Effect of organophosphorus hydrolysing enzymes on obidoxime-induced reactivation of organophosphate-inhibited human acetylcholinesterase. Arch Toxicol 78:338–343. doi:10.1007/s00204-004-0547-2
Klose R, Gutensohn G (1976) Behandlung einer Alkylphosphatintoxikation mit gereinigter Serumcholinesterase. Prakt Anästh 11:1–7
Kovarik Z, Katalinić M, Sinko G et al (2010) Pseudo-catalytic scavenging: searching for a suitable reactivator of phosphorylated butyrylcholinesterase. Chem Biol Interact 187:167–171. doi:10.1016/j.cbi.2010.02.023
Kwong TC (2002) Organophosphate pesticides: biochemistry and clinical toxicology. Ther Drug Monit 24:144–149
Lee EC (2003) Clinical manifestations of sarin nerve gas exposure. JAMA 290:659–662. doi:10.1001/jama.290.5.659
Maxwell DM, Saxena A, Gordon RK, Doctor BP (1999) Improvements in scavenger protection against organophosphorus agents by modification of cholinesterases. Chem Biol Interact 119–120:419–428
Mumford H, Price ME, Lenz DE, Cerasoli DM (2011) Post-exposure therapy with human butyrylcholinesterase following percutaneous VX challenge in guinea pigs. Clin Toxicol 49:287–297. doi:10.3109/15563650.2011.568944
Mumford H, Docx CJ, Price ME et al (2013) Human plasma-derived BuChE as a stoichiometric bioscavenger for treatment of nerve agent poisoning. Chem Biol Interact 203:160–166. doi:10.1016/j.cbi.2012.08.018
Nachon F, Brazzolotto X, Trovaslet M, Masson P (2013) Progress in the development of enzyme-based nerve agent bioscavengers. Chem Biol Interact 206:536–544. doi:10.1016/j.cbi.2013.06.012
Pavlic M, Haidekker A, Grubwieser P, Rabl W (2002) Fatal intoxication with omethoate. Int J Leg Med 116:238–241. doi:10.1007/s00414-002-0299-6
Radić Z, Dale T, Kovarik Z et al (2013) Catalytic detoxification of nerve agent and pesticide organophosphates by butyrylcholinesterase assisted with non-pyridinium oximes. Biochem J 450:231–242. doi:10.1042/BJ20121612
Reiter G, Mikler J, Hill I et al (2011) Simultaneous quantification of VX and its toxic metabolite in blood and plasma samples and its application for in vivo and in vitro toxicological studies. J Chromatogr B Anal Technol Biomed Life Sci 879:2704–2713. doi:10.1016/j.jchromb.2011.07.031
Sidell FR, Groff WA (1970) Toxogonin: blood levels and side effects after intramuscular administration in man. J Pharm Sci 59:793–797
Sit RK, Fokin VV, Amitai G et al (2014) Imidazole aldoximes effective in assisting butyrylcholinesterase catalysis of organophosphate detoxification. J Med Chem 57:1378–1389. doi:10.1021/jm401650z
Taylor P, Radić Z (1994) The cholinesterases: from genes to proteins. Annu Rev Pharmacol Toxicol 34:281–320. doi:10.1146/annurev.pa.34.040194.001433
von der Wellen J, Bierwisch A, Worek F et al (2016) Kinetics of pesticide degradation by human fresh frozen plasma (FFP) in vitro. Toxicol Lett 244:124–128. doi:10.1016/j.toxlet.2015.07.014
Wille T, Thiermann H, Worek F (2014) In vitro kinetics of nerve agent degradation by fresh frozen plasma (FFP). Arch Toxicol 88:301–307. doi:10.1007/s00204-013-1130-5
Worek F, Thiermann H (2013) The value of novel oximes for treatment of poisoning by organophosphorus compounds. Pharmacol Ther 139:249–259. doi:10.1016/j.pharmthera.2013.04.009
Worek F, Mast U, Kiderlen D et al (1999) Improved determination of acetylcholinesterase activity in human whole blood. Clin Chim Acta 288:73–90
Worek F, Thiermann H, Szinicz L, Eyer P (2004) Kinetic analysis of interactions between human acetylcholinesterase, structurally different organophosphorus compounds and oximes. Biochem Pharmacol 68:2237–2248. doi:10.1016/j.bcp.2004.07.038
Worek F, Seeger T, Reiter G et al (2014a) Post-exposure treatment of VX poisoned guinea pigs with the engineered phosphotriesterase mutant C23: a proof-of-concept study. Toxicol Lett 231:45–54. doi:10.1016/j.toxlet.2014.09.003
Worek F, Seeger T, Zengerle M et al (2014b) Effectiveness of a substituted β-cyclodextrin to prevent cyclosarin toxicity in vivo. Toxicol Lett 226:222–227. doi:10.1016/j.toxlet.2014.02.010
Worek F, Thiermann H, Wille T (2016) Catalytic bioscavengers in nerve agent poisoning: a promising approach? Toxicol Lett 244:143–148. doi:10.1016/j.toxlet.2015.07.012
Acknowledgments
The study was funded by the German Ministry of Defence. However, the design, performance, data interpretation and manuscript writing were under the complete control of the authors and have never been influenced. The authors are grateful to G. Duman for her engaged technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wille, T., von der Wellen, J., Thiermann, H. et al. Pseudocatalytic scavenging of the nerve agent VX with human blood components and the oximes obidoxime and HI-6. Arch Toxicol 91, 1309–1318 (2017). https://doi.org/10.1007/s00204-016-1776-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1776-x