Abstract
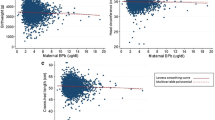
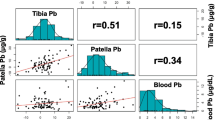
Lead (Pb) can be released from the maternal skeleton during pregnancy and lactation and transferred to the infant. Most support for this hypothesis comes from blood Pb (PbB) studies involving limited sampling during pregnancy, the maximum usually being five samplings, including at delivery. We provide longitudinal data for PbB concentrations and Pb isotopic ratios for three cohorts of pregnant females (n = 31), two of which are based on monthly sampling and the other on quarterly sampling. We also provide data for samples collected post-partum. The data are compared with changes observed in a matched, by country and age, non-pregnant control cohort (n = 5). The monthly data illustrate the variability between subjects, which is also apparent when the data are compared on a trimester basis. Mixed model analyses showed that, in the third trimester, the mean PbB level was significantly lower for women (n = 10) who took a calcium (Ca) supplement (PbB 1.6 µg/dL) than those whose Ca intake was low (low-Ca cohort; n = 15; PbB 2.5 µg/dL) because low Ca means more mobilisation is required for homoeostasis so that more Pb was mobilised from the skeleton. For women who took the supplement, post-partum PbB levels were significantly higher than those in the other periods (2.7 vs 1.4–1.6 µg/dL). For women in the low-Ca cohort, PbB levels were higher at post-partum than in pre-pregnancy and in the first and second trimesters (3.1 vs 1.8 µg/dL), while the levels in the third trimester were higher than those in the first and second trimesters. Importantly, the increase in PbB during gestation was delayed until the third trimester in the Ca-supplemented cohort compared with the low-Ca cohort. Regression analysis showed that the changes over trimester were very similar for PbB and the 206Pb/204Pb ratio providing convincing evidence for extra mobilisation of Pb from the maternal skeleton during pregnancy and lactation. Isotopic ratios in the cord blood samples were similar to those in the maternal blood samples taken prior to parturition with an R 2 0.94 for the migrant subjects and R 2 0.74 for Australian subjects for 206Pb/204Pb ratios, supporting the concept of placental transfer of mobilised skeletal stores of Pb.













Similar content being viewed by others
References
Barry PS (1975) A comparison of concentrations of lead in human tissues. Br J Ind Med 32(2):119–139
Ettinger AS, Lamadrid-Figueroa H, Tellez-Rojo MM et al (2009) Effect of calcium supplementation on blood lead levels in pregnancy: a randomized placebo-controlled trial. Environ Health Perspect 117(1):26–31. doi:10.1289/ehp.11868
Farias P, Borja-Aburto VH, Rios C, Hertz-Picciotto I, Rojas-Lopez M, Chavez-Ayala R (1996) Blood lead levels in pregnant women of high and low socioeconomic status in Mexico City. Environ Health Perspect 104(10):1070–1074
Franklin CA, Inskip MJ, Baccanale CL et al (1997) Use of sequentially administered stable lead isotopes to investigate changes in blood lead during pregnancy in a nonhuman primate (Macaca fascicularis). Fundam Appl Toxicol 39(2):109–119
Goyer RA (1990) Transplacental transport of lead. Environ Health Perspect 89:101–105
Gulson BL, Jameson CW, Mahaffey KR, Mizon KJ, Korsch MJ, Vimpani G (1997a) Pregnancy increases mobilization of lead from maternal skeleton. J Lab Clin Med 130(1):51–62
Gulson BL, Mahaffey KR, Vidal M et al (1997b) Dietary lead intakes for mother/child pairs and relevance to pharmacokinetic models. Environ Health Perspect 105(12):1334–1342
Gulson BL, Jameson CW, Mahaffey KR et al (1998a) Relationships of lead in breast milk to lead in blood, urine, and diet of the infant and mother. Environ Health Perspect 106(10):667–674
Gulson BL, Mahaffey KR, Jameson CW et al (1998b) Mobilization of lead from the skeleton during the postnatal period is larger than during pregnancy. J Lab Clin Med 131(4):324–329
Gulson BL, Mahaffey KR, Jameson CW et al (1999) Impact of diet on lead in blood and urine in female adults and relevance to mobilization of lead from bone stores. Environ Health Perspect 107(4):257–263
Gulson BL, Mizon KJ, Palmer JM et al (2001) Longitudinal study of daily intake and excretion of lead in newly born infants. Environ Res 85(3):232–245. doi:10.1006/enrs.2000.4223
Gulson B, Mizon K, Smith H et al (2002) Skeletal lead release during bone resorption: effect of bisphosphonate treatment in a pilot study. Environ Health Perspect 110(10):1017–1023
Gulson BL, Mizon KJ, Korsch MJ, Palmer JM, Donnelly JB (2003) Mobilization of lead from human bone tissue during pregnancy and lactation—a summary of long-term research. Sci Total Environ 303(1–2):79–104
Gulson BL, Mizon KJ, Palmer JM, Korsch MJ, Taylor AJ, Mahaffey KR (2004) Blood lead changes during pregnancy and postpartum with calcium supplementation. Environ Health Perspect 112(15):1499–1507
Gulson B, Mizon K, Korsch M, Taylor A (2006) Changes in the lead isotopic composition of blood, diet and air in Australia over a decade: globalization and implications for future isotopic studies. Environ Res 100(1):130–138. doi:10.1016/j.envres.2005.03.006
Hernandez-Avila M, Gonzalez-Cossio T, Palazuelos E, Romieu I, Aro A, Fishbein E et al (1996) Dietary and environmental determinants of blood and bone lead levels in lactating postpartum women living in Mexico city. Environ Health Perspect 104:1076–1082
Hernandez-Avila M, Sanin LH, Romieu I, Palazuelos E, Tapia-Conyer R, Olaiz G et al (1997) Higher milk intake during pregnancy is associated with lower maternal and umbilical cord lead levels in postpartum women. Environ Res 74:116–121
Hernandez-Avila M, Gonzalez-Cossio T, Hernandez-Avila JE et al (2003) Dietary calcium supplements to lower blood lead levels in lactating women: a randomized placebo-controlled trial. Epidemiology 14(2):206–212. doi:10.1097/01.EDE.0000038520.66094.34
Hertz-Picciotto I, Schramm M, Watt-Morse M, Chantala K, Anderson J, Osterloh J (2000) Patterns and determinants of blood lead during pregnancy. Am J Epidemiol 152(9):829–837
Hytten F (1985) Blood volume changes in normal pregnancy. Clinic Haematol 14:601–612
Lagerkvist BJ, Ekesrydh S, Englyst V, Nordberg GF, Soderberg HA, Wiklund DE (1996) Increased blood lead and decreased calcium levels during pregnancy: a prospective study of Swedish women living near a smelter. Am J Public Health 86(9):1247–1252
Liu KS, Hao JH, Shi J, Dai CF, Guo XR (2013) Blood lead levels during pregnancy and its influencing factors in Nanjing, China. Chin Med Sci J 28(2):95–101
Manton WI (1977) Sources of lead in blood. Identification by stable isotopes. Arch Environ Health 32:149–159
Manton WI (1985) Total contribution of airborne lead to blood lead. Br J Ind Med 42:168–172
Manton WI (1992) Postpartum changes to maternal blood lead concentrations. Br J Ind Med 49(9):671–672
Manton WI, Angle CR, Stanek KL, Reese YR, Kuehnemann TJ (2000) Acquisition and retention of lead by young children. Environ Res 82(1):60–80. doi:10.1006/enrs.1999.4003
Manton WI, Angle CR, Stanek KL, Kuntzelman D, Reese YR, Kuehnemann TJ (2003) Release of lead from bone in pregnancy and lactation. Environ Res 92(2):139–151
Moura M, Goncalves Valente J (2002) Blood lead levels during pregnancy in women living in Rio de Janeiro, Brazil. Sci Total Environ 299(1–3):123–129
Parsons PJ, Geraghty C, Verostek MF (2001) An assessment of contemporary atomic spectroscopic techniques for the determination of lead in blood and urine matrices’. Spectrochim Acta [B] 56:1593–1604
Rabito FA, Kocak M, Wethermann DW, Tylavsky FA, Palmer CD, Parson PJ (2014) Changes in low levels of lead over the course of pregnancy and the association with birth outcomes. Reprod Toxicol 50:138–144
Rothenberg SJ, Karchmer S, Schnaas L, Perroni E, Zea F, Fernandez Alba J (1994) Changes in serial blood lead levels during pregnancy. Environ Health Perspect 102(10):876–880
Rothenberg SJ, Manalo M, Jiang J et al (1999) Maternal blood lead level during pregnancy in South Central Los Angeles. Arch Environ Health 54(3):151–157. doi:10.1080/00039899909602253
Schell LM, Czerwinski S, Stark AD, Parsons PJ, Gomez M, Samelson R (2000) Variation in blood lead and hematocrit levels during pregnancy in a socioeconomically disadvantaged population. Arch Environ Health 55(2):134–140. doi:10.1080/00039890009603400
Schell LM, Denham M, Stark AD et al (2003) Maternal blood lead concentration, diet during pregnancy, and anthropometry predict neonatal blood lead in a socioeconomically disadvantaged population. Environ Health Perspect 111(2):195–200
Schumacher M, Hernandez M, Domingo JL, Fernandez-Ballart JD, Llobet JM, Corbella J (1996) A longitudinal study of lead mobilization during pregnancy: concentrations in maternal and umbilical cord blood. Trace Elements Electrol 13:177–181
Silbergeld EK (1991) Lead in bone: implications for toxicology during pregnancy and lactation. Environ Health Perspect 91:63–70
Sowers M, Jannausch M, Scholl T, Li W, Kemp FW, Bogden JD (2002) Blood lead concentrations and pregnancy outcomes. Arch Environ Health 57(5):489–495. doi:10.1080/00039890209601442
Tellez-Rojo MM, Hernandez-Avila M, Gonzalez-Cossio T, Romieu I, Aro A, Palazuelos E et al (2002) Impact of breastfeeding on the mobilization of lead from bone. Am J Epidemiol 155:420–428
Tellez-Rojo MM, Hernandez-Avila M, Lamadrid-Figueroa H et al (2004) Impact of bone lead and bone resorption on plasma and whole blood lead levels during pregnancy. Am J Epidemiol 160(7):668–678. doi:10.1093/aje/kwh271
United States Environmental Protection Agency (2013) Office of Research and Development National Center for Environmental Assessment—RTP Division. Integrated Science Assessment for Lead. EPA/600/R-10/075F. Research Triangle Park, NC
US Centers for Disease Control and Prevention (2013) Fourth national exposure report, updated tables. September 2013. http://www.cdc.gov/biomonitoring/Lead_BiomonitoringSummary.html
West WL, Knight EM, Edwards CH et al (1994) Maternal low level lead and pregnancy outcomes. J Nutr 124(6 Suppl):981S–986S
Acknowledgments
We wish to thank: Massimo Chiaradia, Jaqueline Palmer, Matthew James, Nicole Pattison, and Laura Gomez for technical assistance; the participants in this study; Bill Jameson (National Institute of Environmental Health Sciences, NIEHS) and Beth Ragan (NIEHS) and Paul Mushak for their support and encouragement over many years. This research was largely funded from NIEHS through NO1-ES0252. We also thank the reviewers for their constructive and incisive comments and suggestions.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
Approval has been given by Ethics Committees of St. Vincent's Hospital of Sydney, the University of Adelaide in South Australia, Macquarie University and the United States National Institutes of Health.
Informed consent
All persons have given their informed consent prior to their inclusion in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is dedicated to the memory of three wonderful people, Dr. Mahaffey, who was our first US National Institute for Environmental Health Sciences project officer for several years, Professor Tony McMichael, who led the first stage of the contract, and Mary Salter, who was our phlebotomist for 13 years.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gulson, B., Mizon, K., Korsch, M. et al. Revisiting mobilisation of skeletal lead during pregnancy based on monthly sampling and cord/maternal blood lead relationships confirm placental transfer of lead. Arch Toxicol 90, 805–816 (2016). https://doi.org/10.1007/s00204-015-1515-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-015-1515-8