Abstract
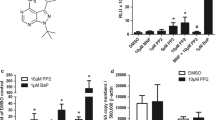
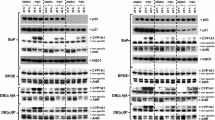
Aromatic amines (AAs) are an important class of chemicals which account for 12 % of known carcinogens. The biological effects of AAs depend mainly on their biotransformation into reactive metabolites or into N-acetylated metabolites which are generally considered as less toxic. Although the activation of the aryl hydrocarbon receptor (AhR) pathway by certain carcinogenic AAs has been reported, the effects of their N-acetylated metabolites on the AhR have not been addressed. Here, we investigated whether carcinogenic AAs and their N-acetylated metabolites may activate/modulate the AhR pathway in the absence and/or the presence of a bona fide AhR ligand (benzo[a]pyrene/B(a)P]. In agreement with previous studies, we found that certain AAs activated the AhR in human liver and lung cells as assessed by an increase in cytochrome P450 1A1 (CYP1A1) expression and activity. Altogether, we report for the first time that these properties can be modulated by the N-acetylation status of the AA. Whereas 2-naphthylamine significantly activated the AhR and induced CYP1A1 expression, its N-acetylated metabolite was less efficient. In contrast, the N-acetylated metabolite of 2-aminofluorene was able to significantly activate AhR, whereas the parent AA, 2-aminofluorene, did not. In the presence of B(a)P, activation of AhR or antagonist effects were observed depending on the AA or its N-acetylated metabolite. Activation and/or modulation of the AhR pathway by AAs and their N-acetylated metabolites may represent a novel mechanism contributing to the toxicological effects of AAs. More broadly, our data suggest biological interactions between AAs and other classes of xenobiotics through the AhR pathway.




Similar content being viewed by others
References
Abel J, Haarmann-Stemmann T (2010) An introduction to the molecular basics of aryl hydrocarbon receptor biology. Biol Chem 391:1235–1248. doi:10.1515/BC.2010.128
Barouki R, Coumoul X, Fernandez-Salguero PM (2007) The aryl hydrocarbon receptor, more than a xenobiotic-interacting protein. FEBS Lett 581:3608–3615. doi:10.1016/j.febslet.2007.03.046
Borza A, Plöttner S, Wolf A et al (2008) Synergism of aromatic amines and benzo[a]pyrene in induction of Ah receptor-dependent genes. Arch Toxicol 82:973–980. doi:10.1007/s00204-008-0381-z
Choi E-Y, Lee H, Dingle RWC et al (2012) Development of novel CH223191-based antagonists of the aryl hydrocarbon receptor. Mol Pharmacol 81:3–11. doi:10.1124/mol.111.073643
Cikryt P, Kaiser T, Göttlicher M (1990) Binding of aromatic amines to the rat hepatic Ah receptor in vitro and in vivo and to the 8S and 4S estrogen receptor of rat uterus and rat liver. Environ Health Perspect 88:213–216
Coumoul X, Diry M, Robillot C, Barouki R (2001) Differential regulation of cytochrome P450 1A1 and 1B1 by a combination of dioxin and pesticides in the breast tumor cell line MCF-7. Cancer Res 61:3942–3948
Dumont J, Jossé R, Lambert C et al (2010) Preferential induction of the AhR gene battery in HepaRG cells after a single or repeated exposure to heterocyclic aromatic amines. Toxicol Appl Pharmacol 249:91–100. doi:10.1016/j.taap.2010.08.027
Ferraz ERA, de Oliveira GAR, Oliveira DP (2012) The impact of aromatic amines on the environment: risks and damages. Front Biosci (Elite Ed) 4:914–923
Freeman HS (2013) Aromatic amines: use in azo dye chemistry. Front Biosci J Virtual Libr 18:145–164
Fukunaga BN, Probst MR, Reisz-Porszasz S, Hankinson O (1995) Identification of functional domains of the aryl hydrocarbon receptor. J Biol Chem 270:29270–29278
Gouédard C, Koum-Besson N, Barouki R, Morel Y (2003) Opposite regulation of the human paraoxonase-1 gene PON-1 by fenofibrate and statins. Mol Pharmacol 63:945–956
Gouédard C, Barouki R, Morel Y (2004) Dietary polyphenols increase paraoxonase 1 gene expression by an aryl hydrocarbon receptor-dependent mechanism. Mol Cell Biol 24:5209–5222. doi:10.1128/MCB.24.12.5209-5222.2004
Hein DW, McQueen CA, Grant DM et al (2000) Pharmacogenetics of the arylamine N-acetyltransferases: a symposium in honor of Wendell W. Weber. Drug Metab Dispos Biol Fate Chem 28:1425–1432
Kim D, Guengerich FP (2005) Cytochrome P450 activation of arylamines and heterocyclic amines. Annu Rev Pharmacol Toxicol 45:27–49. doi:10.1146/annurev.pharmtox.45.120403.100010
Martins M, Rodrigues-Lima F, Dairou J et al (2009) An acetyltransferase conferring tolerance to toxic aromatic amine chemicals: molecular and functional studies. J Biol Chem 284:18726–18733. doi:10.1074/jbc.M109.015230
National Toxicology Program (2011) NTP 12th report on carcinogens. In: Report on carcinogens: carcinogen profiles, vol 12. US Department of Health and Human Services, Public Health Service, National Toxicology Program, pp iii–499
Neumann H-G (2010) Aromatic amines: mechanisms of carcinogenesis and implications for risk assessment. Front Biosci J Virtual Libr 15:1119–1130
Rodrigues-Lima F, Dairou J, Dupret J-M (2008) Effect of environmental substances on the activity of arylamine N-acetyltransferases. Curr Drug Metab 9:505–509
Talaska G (2003) Aromatic amines and human urinary bladder cancer: exposure sources and epidemiology. J Environ Sci Health Part C Environ Carcinog Ecotoxicol Rev 21:29–43. doi:10.1081/GNC-120021372
Turesky RJ, Le Marchand L (2011) Metabolism and biomarkers of heterocyclic aromatic amines in molecular epidemiology studies: lessons learned from aromatic amines. Chem Res Toxicol 24:1169–1214. doi:10.1021/tx200135s
Xue W, Warshawsky D (2005) Metabolic activation of polycyclic and heterocyclic aromatic hydrocarbons and DNA damage: a review. Toxicol Appl Pharmacol 206:73–93. doi:10.1016/j.taap.2004.11.006
Acknowledgments
This work was supported by the Université Paris Descartes, Université Paris Diderot, INSERM (Institut National de la Santé et de la Recherche Médicale), the CNRS (Centre National de la Recherche Scientifique), the ANSES (Agence nationale de sécurité sanitaire de l’alimentation, de l’environnement et du travail, Funding), the Fondation pour la Recherche Médicale (Post doctoral fellowship, Linh-Chi Bui), the Ligue contre le Cancer (Postdoctoral fellowship, Linh-Chi Bui), the Assistance Publique-Hôpitaux de Paris (Erwan Guyot) and the Région Ile de France (PhD fellowships: Ludmila Juricek, Stéphane Pierre). We thank Dr. Lawrence Aggerbeck and Martin Cairns for his critical reading of this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ludmila Juricek and Linh-Chi Bui have contributed equally to this work.
Xavier Coumoul and Rodrigues-Lima share senior authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.
204_2014_1367_MOESM1_ESM.tiff
Supplementary Fig. 1: Measurement of the EROD activity of CYP1A1-XRE-GLuc HepG2 cells following treatment with AAs and BaP. Cells were treated for 8 h with 25 µM of the compounds. EROD activity in cultured cells was measured by the fluorescence of resorufin generated by the conversion of ethoxyresorufin by CYP using a fluorescence plate reader (Coumoul X et al. 2001). Values are the mean ± SEM (n = 4). * p < 0.05; ** p < 0.01 and *** p < 0.001 as compared to corresponding control group. (TIFF 1238 kb)
Rights and permissions
About this article
Cite this article
Juricek, L., Bui, LC., Busi, F. et al. Activation of the aryl hydrocarbon receptor by carcinogenic aromatic amines and modulatory effects of their N-acetylated metabolites. Arch Toxicol 89, 2403–2412 (2015). https://doi.org/10.1007/s00204-014-1367-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-014-1367-7