Abstract
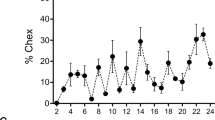
Reproductive process covers lots of procedures, including capacitation, hyperactivation, chemotaxis and the acrosome reaction. Each plays an important role in the success of fertilization. Although multiple studies have reported the toxic effects of fluoride on the male reproduction, the effect of fluoride on sperm chemotaxis is little known. This study is to examine the effect of fluoride on the sperm chemotaxis and then to reveal the underling mechanisms of fluoride toxicity in sperm chemotaxis. 260 healthy Kunming male mice (8 weeks old) were randomly divided into four groups and exposed to 50, 100, 150 mg NaF/L in the drinking water for 8 weeks. At the end of the exposure, sperm chemotaxis was examined using a microchannel-based device. Ca2+ concentration, adenylate cyclase (AC) content and mRNA expression of mACIII, mACVIII, Golf alpha, CatSper1, CatSper2 were measured to elucidate the possible molecular mechanisms. The results showed that the percentage of chemotactic sperm was decreased by NaF in a dose-dependent manner. In the 100 and 150 mg/L groups, Ca2+ concentration and AC content were notably lower than the control group. Compared with the control group, mRNA expression of CatSper1 in the 100 and 150 mg/L treatment groups was decreased significantly, and other genes showed no statistical difference. These data suggested that excessive fluoride did adversely affect sperm chemotaxis. The alteration of Ca2+ concentration, AC content and CatSper1 mRNA expression level may play a key role in the mechanism underlying the affection.




Similar content being viewed by others
References
Alvarez L, Dai L, Friedrich BM, Kashikar ND, Gregor I, Pascal R, Kaupp UB (2012) The rate of change in Ca2+ concentration controls sperm chemotaxis. J Cell Biol 196(5):653–663
Bataineh HN, Nusier MK (2006) Impact of 12-week ingestion of sodium fluoride on aggression, sexual behavior, and fertility in adult male rats. Fluoride 39(4):293–301
Blengini CS, Teves ME, Uñates DR, Guidobaldi HA, Gatica LV, Giojalas LC (2011) Human sperm pattern of movement during chemotactic re-orientation towards a progesterone source. J Asian Androl 13(5):769–773
Branscomb A, Seger J, White RL (2000) Evolution of odorant receptors expressed in mammalian testes. Genetics 156(2):785–997
Chlubek D (2003) Fluoride and oxidative stress. Fluoride 36(4):217–228
Dvoráková-Hortová K, Sandera M, Jursová M, Vasinová J, Peknicová J (2008) The influence of fluorides on mouse sperm capacitation. Anim Reprod Sci 108(1–2):157–170
Eisenbach M (1999) Sperm chemotaxis. Rev Reprod 4(1):56–66
Eisenbach M, Giojalas LC (2006) Sperm guidance in mammals: an unpaved road to the egg. Nat Rev Mol Cell Biol 7(4):276–285
Fawell J, Bailey K, Chilton J, Dahi E, Fewtrell L, Magara Y (2006) Fluoride in drinking-water. IWA Publishing and World Health Publishing, London, p 100
Freni SC (1994) Exposure to high fluoride concentrations in drinking water is associated with decreased birth rates. J Toxicol Environ Health 42(1):109–121
Friedrich BM, Jülicher F (2007) Chemotaxis of sperm cells. PNAS 104(33):13256–13261
Ghosh D, Das Sarkar S, Maiti R, Jana D, Das UB (2002) Testicular toxicity in sodium fluoride treated rats: association with oxidative stress. Reprod Toxicol 16(4):385–390
Guo XY, Sun GF, Sun YC (2003) Oxidative stress from fluoride-induced hepatotoxicity in rats. Fluoride 36(1):25–29
Huang C, Niu RY, Wang JD (2007) Toxic effects of sodium fluoride on reproductive function in male mice. Fluoride 40(3):162–168
Huang C, Yang HB, Niu RY, Sun ZL, Wang JD (2008) Effects of sodium fluoride on androgen receptor expression in male mice. Fluoride 41(1):10–17
Izquierdo-Vega JA, Sánchez-Gutiérrez M, Del Razo LM (2008) Decreased in vitro fertility in male rats exposed to fluoride-induced oxidative stress damage and mitochondrial transmembrane potential loss. Toxicol Appl Pharmacol 230(3):352–357
Kaupp UB, Hildebrand E, Weyand I (2006) Sperm chemotaxis in marine invertebrates: molecules and mechanisms. J Cell Physiol 208(3):487–494
Kirichok Y, Lishko PV (2011) Rediscovering sperm ion channels with the patch-clamp technique. Mol Hum Reprod 17(8):478–499
Ko YJ, Maeng JH, Lee BC, Lee S, Hwang SY, Ahn Y (2012) Separation of progressive motile sperm from mouse semen using on-chip chemotaxis. Anal Sci 28(1):27–32
Koyama S, Amarie D, Soini HA, Novotny MV, Jacobson SC (2006) Chemotaxis assays of mouse sperm on microfluidic devices. Anal Chem 78(10):3354–3359
Krechniak J, Inkielewicz I (2005) Correlations between fluoride concentration and free radical parameters in soft tissues of rats. Fluoride 38(4):293–296
Lawson PB, Yu MH (2003) Fluoride inhibition of superoxide dismutase (SOD) from the earthworm Eisenia fetida. Fluoride 36(3):143–151
Lishko PV, Kirichok Y, Ren D, Navarro B, Chung JJ, Clapham DE (2012) The control of male fertility by spermatozoan ion channels. Annu Rev Physiol 74:453–475
Lou DD, Guan ZZ, Liu YJ, Liu YF, Zhang KL, Pan JG, Pei JJ (2013) The influence of chronic fluorosis on mitochondrial dynamics morphology and distribution in cortical neurons of the rat brain. Arch Toxicol 87(3):449–457
Mittal M, Flora SJ (2007) Vitamin E supplementation protects oxidative stress during arsenic and fluoride antagonism in male mice. Drug Chem Toxicol 30(3):263–281
Nabavi SF, Nabavi SM, Latifi AM, Mirzaei M, Habtemariam S, Moghaddam AH (2012) Mitigating role of quercetin against sodium fluoride-induced oxidative stress in the rat brain. Pharm Biol 50(11):1380–1383
Narayana MV, Chinoy NJ (1994) Effect of fluoride on rat testicular steroidogenesis. Fluoride 27(1):7–12
Oliveira RG, Tomasi L, Rovasio RA, Giojalas LC (1999) Increased velocity and induction of chemotactic response in mouse spermatozoa by follicular and oviductal fluids. J Reprod Fertil 115(1):23–27
Ortiz-Pérez D, Rodríguez-Martínez M, Martínez F, Borja-Aburto VH, Castelo J, Grimaldo JI, de la Cruz E, Carrizales L, Díaz-Barriga F (2003) Fluoride-induced disruption of reproductive hormones in men. Environ Res 93(1):20–30
Ostermeier GC, Dix DJ, Miller D, Khatri P, Krawetz SA (2002) Spermatozoal RNA profiles of normal fertile men. Lancet 360(9335):772–777
Parmentier M, Libert F, Schurmans S, Schiffmann S, Lefort A, Eggerickx D, Ledent C, Mollereau C, Gérard C, Perret J et al (1992) Expression of members of the putative olfactory receptor gene family in mammalian germ cells. Nature 355(6359):453–455
Pushpalatha T, Srinivas M, Sreenivasula Reddy P (2005) Exposure to high fluoride concentration in drinking water will affect spermatogenesis and steroidogenesis in male albino rats. Biometals 18(3):207–212
Shanthakumari D, Srinivasalu S, Subramanian S (2004) Effect of fluoride intoxication on lipidperoxidation and antioxidant status in experimental rats. Toxicology 204(2–3):219–228
Spehr M, Schwane K, Riffell JA, Barbour J, Zimmer RK, Neuhaus EM, Hatt H (2004) Particulate adenylate cyclase plays a key role in human sperm olfactory receptor-mediated chemotaxis. J Biol Chem 279(38):40194–40203
Spittle B (2008) Fluoride and fertility [editorial]. Fluoride 41(2):98–100
Strünker T, Goodwin N, Brenker C, Kashikar ND, Weyand I, Seifert R, Kaupp UB (2011) The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature 471(7338):382–386
Sun F, Giojalas LC, Rovasio RA, Tur-Kaspa I, Sanchez R, Eisenbach M (2003) Lack of species-specificity in mammalian sperm chemotaxis. Dev Biol 255(2):423–427
Sun F, Bahat A, Gakamsky A, Girsh E, Katz N, Giojalas LC, Tur-Kaspa I, Eisenbach M (2005) Human sperm chemotaxis: both the oocyte and its surrounding cumulus cells secrete sperm chemoattractants. Hum Reprod 20(3):761–767
Sun Z, Niu R, Su K, Wang B, Wang J, Zhang J, Wang J (2010) Effects of sodium fluoride on hyperactivation and Ca2+ signaling pathway in sperm from mice: an in vivo study. Arch Toxicol 84(5):353–361
Sun Z, Niu R, Wang B, Jiao Z, Wang J, Zhang J, Wang S, Wang J (2011) Fluoride-induced apoptosis and gene expression profiling in mice sperm in vivo. Arch Toxicol 85(11):1441–1452
Susheela AK, Kumar A (1991) A study of the effect of high concentrations of fluoride on the reproductive organs of male rabbits, using light and scanning electron microscopy. J Reprod Fertil 92(2):353–360
Teves ME, Guidobaldi HA, Uñates DR, Sanchez R, Miska W, Publicover SJ, Morales Garcia AA, Giojalas LC (2009) Molecular mechanism for human sperm chemotaxis mediated by progesterone. PLoS One 4(12):e8211
Varol E, Varol S (2012) Effect of fluoride toxicity on cardiovascular systems: role of oxidative stress. Arch Toxicol 86(10):1627
Wolfner MF (2011) Precious essences: female secretions promote sperm storage in Drosophila. PLoS Biol 9(11):e1001191
Xie L, Ma R, Han C, Su K, Zhang Q, Qiu T, Wang L, Huang G, Qiao J, Wang J, Cheng J (2010) Integration of sperm motility and chemotaxis screening with a microchannel-based device. Clin Chem 56(8):1270–1278
Zhang JH, Liang C, Ma JJ, Niu RY, Wang JD (2006) Effects of sodium fluoride and sulfur dioxide on sperm motility and serum testosterone in male rats. Fluoride 39(2):126–131
Zimmer RK, Riffrey JA (2011) Sperm chemotaxis, fluid shear, and the evolution of sexual reproduction. PNAS 108(32):13200–13205
Acknowledgments
This research was supported by China National Natural Science Foundation (31072180, 31201965 and 31172376).
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhaojing Lu, Shaolin Wang and Zilong Sun have contributed equally to this work and should be considered co-first authors.
Rights and permissions
About this article
Cite this article
Lu, Z., Wang, S., Sun, Z. et al. In vivo influence of sodium fluoride on sperm chemotaxis in male mice. Arch Toxicol 88, 533–539 (2014). https://doi.org/10.1007/s00204-013-1099-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-013-1099-0