Abstract
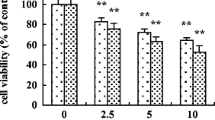
The cytotoxic effects of hydroxylated fullerenes, also termed fullerenols or fullerols [C60(OH) n ], which are known nanomaterials and water-soluble fullerene derivatives, were studied in freshly isolated rat hepatocytes. The exposure of hepatocytes to C60(OH)24 caused not only concentration (0–0.25 mM)- and time (0–3 h)-dependent cell death accompanied by the formation of cell blebs, loss of cellular ATP, reduced glutathione (GSH), and protein thiol levels, but also the accumulation of glutathione disulfide and malondialdehyde, indicating lipid peroxidation. Of the other analogues examined, the cytotoxic effects of C60(OH)12 and fullerene C60 at a concentration of 0.125 mM were less than those of C60(OH)24. The loss of mitochondrial membrane potential and generation of oxygen radical species in hepatocytes incubated with C60(OH)24 were greater than those with C60(OH)12 and fullerene C60. In the oxygen consumption of mitochondria isolated from rat liver, the ratios of state-3/state-4 respiration were more markedly decreased by C60(OH)24 and C60(OH)12 compared with C60. In addition, C60(OH)24 and C60(OH)12 resulted in the induction of the mitochondrial permeability transition (MPT), and the effects of C60(OH)12 were less than those of C60(OH)24. Taken collectively, these results indicate that (a) mitochondria are target organelles for fullerenols, which elicit cytotoxicity through mitochondrial failure related to the induction of the MPT, mitochondrial depolarization, and inhibition of ATP synthesis in the early stage and subsequently oxidation of GSH and protein thiols, and lipid peroxidation through oxidative stress at a later stage; and (b) the toxic effects of fullerenols may depend on the number of hydroxyl groups participating in fullerene in rat hepatocytes.







Similar content being viewed by others
Abbreviations
- DCHF-DA:
-
2′,7′-Dichlorodihydrofluorescein diacetate
- DMSO:
-
Dimethyl sulfoxide
- GSH:
-
Glutathione
- GSSG:
-
Glutathione disulfide
- HEPES:
-
N-(2-hydroxyethyl)-piperazine-N-(2-ethanesulfonic acid)
- MDA:
-
Malondialdehyde
- MPT:
-
Mitochondrial permeability transition
- ROS:
-
Reactive oxygen species
- ΔΨ:
-
Membrane potential
References
Albano E, Rundgren M, Hervison PJ, Nelson SD, Moldéus P (1985) Mechanisms of N-acetyl-p-benzoquinone imine cytotoxicity. Mol Pharmacol 28:306–311
Arora AS, Jones BJ, Patel TC, Bornk SF, Gores JG (1997) Ceramide induces hepatocytes cell death through disruption of mitochondrial function in the rat. Hepatology 25:958–963
Bellomo G, Mirabelli F, Richelmi P, Malorni W, Iosi F, Orrenius S (1990) The cytoskeleton as a target in quinone toxicity. Free Radic Res Commun 8:391–399
Bogdanović G, Kojić V, Dordević A, Canadanović-Brunet J, Vojinović-Miloradov M, Baltić VV (2004) Modulating activity of fullerols C60(OH)22 on doxorubicin-induced cytotoxicity. Toxicol In Vitro 18:629–637
Borm PJA, Robbins D, Haubold S, Kuhlbusch T, Fissan H, Donaldson K, Schins R, Stone V, Kreyling W, Lademann J, Krutmann J, Warheit D, Oberdorster W (2006) The potential risks of nanomaterials: a review carried out for ECETOC. Part Fibre Toxicol 3:11. doi:10.1186/1743-8977-3-11
Cain K, Skilleter DS (1987) Preparation and use of mitochondria in toxicological research. In: Snell K, Mullock B (eds) Biochemical toxicology: a practical approach. IRL Press, Oxford, pp 217–254
Chen YW, Hwang KC, Yen CC, Lai YL (2004) Fullerene derivatives protect against oxidative stress in RAW 264.7 cells and ischemia-reperfused lungs. Am J Physiol Integr Comp Physiol 287:R21–R26
Chen C, Xing G, Wang J, Zhao F, Chai Z, Fang X (2005) Multihydroxylated [Gd@C82(OH)22] n nanoparticles: antineoplastic activity of high efficiency and low toxicity. Nano Lett 5:2050–2057
Dugan LL, Gabrielsen JK, Yu SP, Lin TS, Choi DW (1996) Buckminsterfullerenol free radical scavengers reduce excitotoxic and apoptotic death of cultured cortical neurons. Neurobiol Dis 3:129–135
Fawthrop DJ, Boobis AR, Davies DS (1991) Mechanisms of cell death. Arch Toxicol 65:437–444
Foley S, Crowley C, Smaihi M, Bonfils C, Erlanger BF, Seta P, Larroque C (2002) Cellular localisation of a water-soluble fullerene derivative. Biochem Biophys Res Commun 294:116–119
Gelderman MP, Simkova O, Clogston JD, Partri AK, Siddiqui SF, Vostal AC, Simka J (2008) Adverse effects of fullerenes on endothelial cells: fullerenol C60(OH)24 induced tissue factor and ICAM-I membrane expression and apoptosis in vitro. Int J Nanomedicine 3:59–68
Griffiths EJ, Halestrap AP (1995) Mitochondrial non-specific pores remain closed during cardiac ischemia, but open upon reperfusion. Biochem J 307:93–98
Hoet PHM, Brüske-Hohlfeld I, Salata O (2004) Nanoparticles-known and unknown health risks. J Nanobiotechnol 2:1–15
Injac R, Perse M, Obermajer N, Djordjevic-Milic V, Prijatelj M, Djordjevic A, Cerar A, Strukelj B (2008) Potential hepatoprotective effects of fullerenol C60(OH)24 in doxorubicin-induced hepatotoxicity in rats with mammary carcinomas. Biomaterials 29:3451–3460
Injac R, Radic N, Govedarica B, Perse M, Cerar A, Djordjevic A, Strukelj B (2009) Acute doxorubicin pulmotoxicity in rat with malignant neoplasm is effectively treated with fullerenol C60(OH)24 through inhibition of oxidative stress. Pharmacol Rep 61:335–342
Isakovic A, Markovic Z, Todorovic-Markovic B, Nikolic N, Vranjes-Djuric S, Mirkovic M, Dramicanin M, Harhaji L, Raicevic N, Nikolic Z, Trakovic V (2006) Distinct cytotoxic mechanisms of pristine versus hydroxylated fullerene. Toxicol Sci 91:173–183
Jin H, Chen WQ, Tang XW, Chiang LY, Yang CY, Schloss JV, Wu JY (2000) Polyhydroxylated C60, fullerenols, as glutamate receptor antagonists and neuroprotective agents. J Neurosci Res 62:600–607
Johnson-Lyles DN, Peifley K, Lockett S, Neun BW, Hansen M, Clogston J, Stern ST, McNeil SE (2010) Fullerenol cytotoxicity in kidney cells is associated with cytoskeleton disruption, autophagic vacuole accumulation, and mitochondrial dysfunction. Toxicol Appl Pharmacol 248:249–258
Jones DP (1981) Determination of pyridine dinucleotides in cell extracts by high-performance liquid chromatography. J Chromatogr 225:446–449
Kamat JP, Devasagayam TP, Priyadarsini KI, Mohan H (2000) Reactive oxygen species mediated membrane damage induced by fullerene derivatives and its possible biological implications. Toxicology 155:55–61
Kehrer JP, Jones DP, Lemasters JJ, Farber H, Jaeschke H (1990) Mechanisms of hypoxic cell injury. Summary of the symposium presented at the 1990 annual meeting of the society of toxicology. Toxicol Appl Pharmacol 106:165–178
Kettenhofen NJ, Wood MJ (2010) Formation, reactivity, and detection of protein sulfenic acids. Chem Res Toxicol 23:1633–1646
Kim J-S, Jin Y, Lemasters JJ (2006) Reactive oxygen species, but not Ca2+, trigger pH and mitochondrial permeability transition-dependent killing of adult rat myocytes after ischemia/reperfusion. Am J Phyisol Heart Circ Physiol 290:H2024–H2034
Kon K, Kim J-S, Uchiyama A, Jaeschke H, Lemasters JJ (2010) Lysosomal iron mobilization and induction of the mitochondrial permeability transition in acetaminophen-induced toxicity to mouse hepatocytes. Toxicol Sci 117:101–108
Kroemer G (1999) Mitochondrial control of apoptosis: an overview. Biochem Soc Symp 66:1–15
Kroto HW, Heath JR, O’Brien SC, Curl RF, Smalley RE (1985) C60: buckministerfullerene. Nature 318:162–163
Lemasters JJ, Nieminen AL, Chacon E, Imberti R, Gores G, Reece JM, Herman B (1993) Use of fluorescent probes to monitor mitochondrial membrane potential in isolated mitochondria, cell suspensions, and cultured cells. In: Lash LH, Jones DP (eds) Mitochondrial dysfunction. Academic Press, San Diego, pp 404–415
Lemasters JJ, Nieminen AL, Qian T, Trost LC, Herman B (1997) The mitochondrial permeability transition in toxic, hypoxic and reperfusion injury. Mol Cell Biochem 174:159–165
Levi N, Hantgan RR, Lively MO, Carroll DL, Prasad GL (2006) C60-fullerenes: detection of intracellular photoluminescence and lack of cytotoxic effects. J Nanobiotechnol 4:14
Li N, Sioutas C, Cho A, Schmitz D, Misra C, Sempf J, Wang M, Terry Oberley T, Froines J, Nel A (2003) Ultrafine particulate pollutants induce oxidative stress and mitochondrial damage. Environ Health Percept 111:455–460
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mehta R, Chan K, Lee O, Tafazoli S, O’Brien PJ (2008) Drug-associated mitochondrial toxicity. In: Dykens JA, Will Y (eds) Drug-induced mitochondrial dysfunction. Wiley, Hoboken, pp 71–126
Moldéus P, Hogberg J, Orrenius S (1978) Isolation and use of liver cells. Method Enzymol 52:60–71
Möller W, Brown DM, Kreyling WG, Stone V (2005) Ultrafine particles cause cytoskeletal dysfunctions in macrophages: role of intracellular calcium. Part Fibre Toxicol 2:7. doi:10.1186/1743-8977-2-7
Mrdanović J, Solajić C, Bogdanović V, Stankov K, Bogdanović G, Djordjevic A (2009) Effects of fullerenol C60(OH)24 on the frequency of micronuclei and chromosome aberrations in CHO-K1 cells. Mutat Res 680:25–30
Murugan MA, Gangadharan B, Mathur P (2002) Antioxidative effect of fullerenol on goat epididymal spermatozoa. Asian J Androl 4:149–152
Nakagawa Y, Moldéus P (1999) Role of mitochondrial membrane permeability transition in p-hydroxybenzoate ester-induced cytotoxicity in rat hepatocytes. Biochem Pharmacol 58:811–816
Nakagawa Y, Suzuki T, Kamimura H, Nagai F (2005) N-Nitrosofenfluramine induces cytotoxicity via mitochondrial dysfunction and oxidative stress in isolated rat hepatocytes. Arch Toxicol 79:312–320
Nakagawa Y, Suzuki T, Tayama S, Ishii H, Ogata A (2009) Cytotoxic effects of 3,4-methylenedioxy-N-alkylamphetamines, MDMA and its analogues, on isolated rat hepatocytes. Arch Toxicol 83:69–80
Nicotera P, Bellomo G, Orrenius S (1992) Calcium-mediated mechanisms in chemically induced cell death. Annu Rev Pharmacol Toxicol 32:449–470
Niwa Y, Iwai N (2007) Nanomaterials induce oxidized low-density lipoprotein cellular uptake in macrophages and platelet aggregation. Circ J 71:437–444
Petronilli V, Constantini P, Scorrano L, Colonna R, Passamonti S, Bermardi P (1994) The voltage sensor of the mitochondrial permeability transition pore is turned by the oxidation-reduction state of vicinal thiols. Increase of the gating potential by oxidation and its reversal by reducing agents. J Biol Chem 269:16638–16642
Pourahmad J, Khan S, O’Brien PJ (2001) Lysosomal oxidative stress cytotoxicity induced by nitrofurantoin redox cycling in hepatocytes. Adv Exp Med Biol 500:261–265
Qian T, Nieminen A-L, Herman B, Lemasters JJ (1997) Mitochondrial permeability transition in pH-dependent reperfusion injury to rat hepatocytes. Am J Physiol 273:C1783–C1792
Reed DJ (1990) Glutathione: toxicological implications. Annu Rev Pharmacol Toxicol 30:603–631
Reed DJ, Babson JR, Beatty PW, Brodie AE, Ellis WW, Potter DW (1980) High-performance liquid chromatography analysis of nanomole levels of glutathione, glutathione disulfide and related thiols and disulfides. Anal Biochem 106:55–62
Roberts JE, Wielgus AR, Boyes WK, Andley U, Chignell CF (2008) Phototoxicity and cytotoxicity of fullerols in human lens epithelial cells. Toxicol Appl Pharmacol 228:49–58
Sadauskas E, Wallin H, Stoltenberg M, Vogel U, Doering P, Larsen A, Danscher G (2007) Kupffer cells are central in the removal of nanoparticles from the organism. Part Fibre Toxicol 4:10. doi:10.1186/1743-8911-4-10
Saitoh Y, Xiao L, Mizuno H, Kato S, Aoshima H, Taira H, Kokubo K, Miwa N (2010) Novel polyhydroxylated fullerene suppresses intracellular oxidative stress together with repression of intracellular lipid accumulation during the differentiation of OP9 preadipocytes into adipocytes. Free Radic Res 44:1072–1081
Sandy MS, Moldéus P, Ross D, Smith M (1986) Role of redox cycling and lipid peroxidation in bipyridyl herbicide cytotoxicity. Studies with a compromised isolated hepatocyte model system. Biochem Pharmacol 35:3095–3101
Sayes CM, Fortner JD, Guo W, Lyon D, Boyd AM, Ausman KD, Tao YJ, Sitharaman B, Wilson LJ, Hughes JB, West JL, Colvin VL (2004) The differential cytotoxicity of water-soluble fullerenes. Nano Lett 4:1881–1887
Shen H-M, Shi C-Y, Shen Y, Ong C-N (1996) Detection of elevated reactive oxygen species level in cultured rat hepatocytes treated with aflatoxin B1. Free Radic Biol Med 21:139–146
Trost LC, Lemasters JJ (1996) The mitochondrial permeability transition: a new pathophysiological mechanism for Rey’s syndrome and toxic lover injury. J Pharmacol Exp Ther 278:1000–1005
Tsai MC, Chen YH, Chiang LY (1997) Polyhydroxylated C60, fullerenol, a novel free-radical trapper, prevented hydrogen peroxide- and cumene hydroperoxide-elicted changes in rat hippocampus in vitro. J Pharma Pharmacol 49:438–445
Uchiyama A, Kim JS, Kon K, Jaeschke H, Ikejima K, Watanabe S, Lemasters JJ (2008) Translocation of iron from lysosomes into mitochondria is a key event during oxidative stress-induced hepatocellular injury. Hepatology 48:1644–1654
Ueng T-H, Kang JJ, Wang HW, Cheng YW, Chiang LY (1997) Suppression of microsomal cytochrome P450-dependent monooxygenases and mitochondrial oxidative phosphorylation by fullerenol, a polyhydroxylated fullerene C60. Toxicol Lett 93:29–37
Vileno B, Marcoux PR, Lekka M, Sienkiewicz A, Fehér T, Forró L (2006) Spectroscopic and photophysical properties of a highly derivatized C60 fullerol. Adv Funct Mater 16:120–128
Wallace KB, Eells JT, Madeira VM, Cortopassi G, Jones DP (1997) Mitochondria-mediated cell injury. Symposium overview. Fundam Appl Toxicol 38:23–37
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27:612–616
Yamawaki H, Iwai N (2006) Cytotoxicity of water-soluble fullerene in vascular endothelial cells. Am J Physiol Cell Physiol 290:C1495–C1502
Zhang JG, Tirmenstein MA, Nicholls-Grzemski FA, Fariss NW (2001) Mitochondrial electron transport inhibitors cause lipid peroxidation-dependent and -independent cell death: protective role of antioxidants. Arch Biochem Biophys 393:87–96
Zhao B, He YY, Chignell CF, Yin JJ, Andley U, Roberts JE (2009) Difference in phototoxicity of cyclodextrin complexed fullerene [(γ-CyD)2/C60] and its aggregated derivatives toward human lens epithelial cells. Chem Res Toxicol 22:660–667
Conflict of interest
The authors declare that they have no conflict of interest in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakagawa, Y., Suzuki, T., Ishii, H. et al. Cytotoxic effects of hydroxylated fullerenes on isolated rat hepatocytes via mitochondrial dysfunction. Arch Toxicol 85, 1429–1440 (2011). https://doi.org/10.1007/s00204-011-0688-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-011-0688-z