Abstract
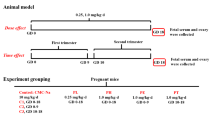
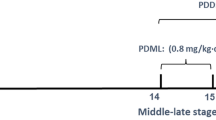
Ketoconazole (KT) is a broad-spectrum antifungal agent whose pharmacological activity is based on the capability to interfere with steroid biosynthesis through an interaction with fungal cytochrome P-450 enzymes and thereby avoiding the formation of fungal walls. As the inhibition of fungal cytochrome P-450 by KT is not specific, the mammalian cytochrome P-450 species, which play an important role in the biosynthesis of steroidogenesis, are also affected. The reproductive and developmental toxicity of KT have been assessed. This antimycotic agent has been reported as embryotoxic and teratogenic when administered in high doses (80 mg/kg) to pregnant rats. The mechanisms by which KT exert teratogenic effects remains to be elucidated. When considering the potential inhibitory effect of KT on mammalian steroid biosynthesis as a possible responsible for the skeletal anomalies induced by this drug, this study aimed at determining whether steroid maternal supplementation may prevent the skeletal anomalies induced by KT. To test this hypothesis, maternal supplementation with prednisone (PRED) (0.1, 0.2 or 0.4 mg/kg) and 80 mg/kg of KT were administered to pregnant Wistar rats (n = 10) during organogenesis period. On gestational day 21, the dams were euthanized and examined for standard parameters of reproductive outcome. In summary, the results showed that PRED supplementation therapy may cause reductions in the incidence of KT-induced cranial and appendicular skeletal anomalies as well as cleft palate in the rat, being these results more consistent with 0.4 mg/kg of this drug. These results suggest an important role for glucocorticoids in KT-induced teratogenesis
Similar content being viewed by others
References
AHFS (1998) American Hospital Formulary Service. Drug Information. McEvoy GK (ed) USA, pp 96–119
Amaral VCS, Nunes GP Jr (2008) Ketoconazole-and fluconazole-induced embryotoxicity and skeletal anomalies in Wistar rats: a comparative study. Braz Arch Biol Technol 51:1153–1161
Briggs GG, Freeman RK, Yaffe SJ (1998) Drugs in pregnancy and lactation: a reference guide to fetal and neonatal risk. Lippincott Williams & Wilkins, USA
Buttar HS, Moffatt JH, Bura C (1989) Pregnancy outcome in ketoconazole-treated rats and mice. Teratology 39:444
Como JA, Dismukes WE (1994) Drug therapy: oral azole drugs as systemic antifungal therapy. N Engl J Med 330:263–272. doi:10.1056/NEJM199401273300407
Cummings AM, Hedge JL, Laskey J (1997) Ketoconazole impairs early pregnancy and decidual cell response via alterations in ovarian function. Fundam Appl Toxicol 40:238–246. doi:10.1006/faat.1997.2392
Engelhardt D, Weber MM, Miksch T, Abedinpour F, Jaspers C (1991) The influence of ketoconazole on human adrenal steroidogenesis—incubation studies with tissue-slices. Clin Endocrinol (Oxf) 35:163–168. doi:10.1111/j.1365-2265.1991.tb03516.x
Friedman JM, Polifka JE (1996) The effect of drugs on the fetus and nursing infant. Johns Hopins University Press, Baltimore
Iranmanesh A, Ovetsky RM, Lizarralde G, Schleupner CJ (1987) Effect of ketoconazole on the 11-hydroxylation step of adrenal steroidogenesis. South Med J 80:725–728. doi:10.1097/00007611-198706000-00015
Kazy Z, Puhó E, Czeizel AE (2005) Population-based case-control study of oral ketoconazole: treatment for birth outcomes. Congenit Anom (Kyoto) 45:5–8. doi:10.1111/j.1741-4520.2005.00053.x
Kimmel CA, Wilson JG (1973) Skeletal deviations in rats: malformations or variations? Teratology 8:309–316. doi:10.1002/tera.1420080311
King CT, Rogers PD, Cleary JD, Chapman SW (1998) Antifungal therapy during pregnancy. Clin Infect Dis 27:1151–1160. doi:10.1086/514977
Loose DS, Kan PB, Hirst MA, Marcus RA, Feldman D (1983) Ketoconazole blocks adrenal steroidogenesis by inhibiting cytochrome P450-dependent enzymes. J Clin Invest 71:1495–1499. doi:10.1172/JCI110903
MacDonald GJ (1978) Factors involved in maintenance of pregnancy in the rat. The temporal need for estrogen, pituitary prolactin and the ovary. Biol Reprod 19:817–823. doi:10.1095/biolreprod19.4.817
Manson JM, Zenick H, Costlow RD (1982) Teratology test methods for laboratory animals. In: Hayves AW (ed) Principles of toxicology. Wallace Hayes, New York, pp 141–184
Moudgal VV, Sobel JD (2003) Antifungal drugs in pregnancy: a review. Expert Opin Drug Saf 2:457–483. doi:10.1517/14740338.2.5.475
Rodriguez RJ, Acosta D Jr (1995) Comparison of ketoconazole and fluconazole induced hepatotoxicity in a primary culture system of rat hepatocytes. Toxicology 96:83–92. doi:10.1016/0300-483X(94)02911-D
Sheehan DJ, Hitchcock CA, Sibley CM (1999) Current and emerging azole antifungal agents. Clin Microbiol Rev 12:40–79
Sobel JD (2000) Use of antifungal drugs in pregnancy: a focus on safety. Drug Saf 23:77–85. doi:10.2165/00002018-200023010-00005
Sonino N (1987) The use of ketoconazole as an inhibitor of steroid production. N Engl J Med 317:812–818
Van Cauteren H, Lampo A, Vandenberghe J et al (1989) Toxicological profile and safety evaluation of antifungal azole derivates. Mycoses 32:60–66
Van Cauteren H, Lampo A, Vandenberghe J et al (1990) Safety aspects of oral antifungal agents. Br J Clin Pract Suppl 71:47–49
Vanden Bossche H (1985) Biochemical targets for antifungal azole derivates: hypothesis on the mode of action. Curr Top Med Mycol 1:313–351
Vanden Bossche H (1992) Inhibitors of P450-dependent steroid biosynthesis: from research to medical treatment. J Steroid Biochem Mol Biol 8:1003–1021. doi:10.1016/0960-0760(92)90328-G
Zarn JA, Bruschweiler BJ, Schlatter JR (2003) Azole fungicides affect mammalian steroidogenesis by inhibiting sterol 14 alpha-demethylase and aromatase. Environ Health Perspect 111:255–261
Acknowledgments
The authors are grateful to Fundação de Apoio à Pesquisa—FUNAPE for supporting this work. The authors also thank Janssen Cilag Pharmaceutica (São Paulo-Brazil) for generously providing ketoconazole for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amaral, V.C.S., Nunes , G.P. Prednisone reduces ketoconazole-induced skeletal defects in rat fetuses. Arch Toxicol 83, 863–871 (2009). https://doi.org/10.1007/s00204-009-0439-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-009-0439-6