Abstract
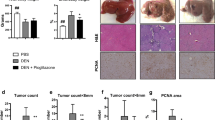
Six-week-old rasH2 mice were injected intraperitoneally with N-diethylnitrosamine (DEN) after partial hepatectomy and administrated 0 or 6,000 ppm troglitazone (TRG) for 10 weeks. Relative liver weight of females increased significantly in the DEN + TRG group compared to the DEN-alone group. The numbers of γ-glutamyltranspeptidase- and proliferating cell nuclear antigen (PCNA)-positive cells tended to increase in both the sexes in the DEN + TRG group; however, these changes were not significantly different from those in the DEN-alone group. Levels of gene expressions for vascular endothelial growth factor (VEGF) and VEGFB (related to angiogenesis), tropomyosin 1 (Tpm1) and transforming growth factor-β (related to ras/MAPK cascade activation), and PCNA (related to cell proliferation) in females were significantly higher in the DEN + TRG than in the untreated control group but not in the DEN-alone group. Only Tpm1 gene had significantly higher expression in the DEN + TRG group than in the DEN-alone group. These results suggest that rasH2 mice are not susceptible to TRG in a two-stage hepatocarcinogenesis model.





Similar content being viewed by others
References
Cater KC, Gandolfi AJ, Sipes IG (1985) Characterization of dimethylnitrosamine-induced focal nodular lesions in the livers of new born mice. Toxicol Pathol 13:3–9
Dai J, Rabie AB (2007) VEGF: an essential mediator of both angiogenesis and endochondral ossification. J Dent Res 86(10):937–50
D’Arcy PF, Harron DWG (eds) (1998) Proceedings of the fourth iternational conference on harmonization. Queen’s University of Belfast, N Ireland
Elangbam CS, Brodie TA, Brown HR, Nold JB, Raczniak TJ, Tyler RD, Lightfoot RM, Wall HG (2002) Vascular effects of Gl262570X (PPAR-gamma agonist) in the brown adipose tissue of Han Wistar rats: a review of 1-month, 13-week, 27-week and 2-year oral toxicity studies. Toxicol Pathol 30:420–426
Ghazzi MN, Perez JE, Antonucci TK, Driscoll JH, Faja Huang SM, BW Whitcomb RW (1997) Cardiac and glycemic benefits of troglitazone treatment in NIDDM. The troglitazone study group. Diabetes 46:433–439
Hanigan MH, Kemp CJ, Ginsler JJ, Drinkwater NR (1988) Rapid growth of preneoplastic lesions in hepatocarcinogen-sensitive C3H/HeJ male mice relative to C57BL/6 J male mice. Carcinogenesis 9:855–890
Hardisty JF, Elwell MR, Ernst H, Greaves P, Kolenda-Roberts H, Malarkey DE, Mann PC, Tellier PA (2007) Histopathology of hemangiosarcomas in mice and hamsters and liposarcomas/gibrosarcomas in rats associated with PPAR agonists. Toxicol Pathol 35(7):928–41
Herman JR, Dethloff LA, McGuire EJ, Parker RF, Walsh KM, Gough AW, Masuda H, de la Iglesia FA (2002) Rodent carcinogenicity with the thiazolidinedione antidiabetic agent troglitazone. Toxicol Sci 68:226–236
Ito N, Tsuda H, Tatematsu M, Inoue T, Tagawa Y, Aoki T, Uwagawa S, Kagawa M, Ogiso T, Masui T, Imaida K, Fukushima S, Asamoto M (1988) Enhancing effect of various hepatocarcinogens on induction of preneoplastic glutathione S-transferase placental form positive foci in rats an approach for a new medium-term bioassay system. Carcinogenesis 9:387–394
Jeri El-Hage (2004) “Preclinical and clinical safety assessments for PPAR agonists” DIA Presentations, Washington, DC. http://www.fda.gov/cder/present/DIA2004/default.htm
Jin M, Takahashi M, Moto M, Muguruma M, Ito K, Watanabe K, Kenmochi Y, Kono T, Hasumi K, Mitsumori K (2007) Carcinogenic susceptibility of rasH2 mice to troglitazone. Arch Toxicol 81:883–894
Keller H, Mahfoudi A, Dreyer C, Hihi AK, Medin J, Ozato K, Wahli W (1993) Peroxisome proliferators-activated receptors and lipid metabolism. Ann NY Acad Sci 684:157–173
Lee GH, Nomura K, Kitagawa T (1989) Comparative study of diethulnitrosamine-initiated two-stage hepatocarcinogenesis in C3H, B57BL and BALB mice promoted by various hepatopromoters. Carcinogenesis 12:2227–2230
Maruyama C, Tomisawa M, Wakana S, Ymazaki H, Kijima H, Suemizu H, Ohnishi Y, Urano Y, Hioki K, Usui T, Nakayama M, Tsuchida T, Mitumori K, Nomura T, Tamaoki N, Ueyama Y (2001) Overexpression of human H-ras transgene is responsible for tumors induced by chemical carcinogens in mice. Oncol Rep 8:233–237
Nesfield SR, Clarke CJ, Hoivik DJ, Miller RT, Allen JS, Selinger K, Santostefano MJ (2005) Evaluation of the carcinogenic potential of clofibrate in the rasH2 mouse. Int J Toxicol 24:301–311
Okamura M, Suida K, Muto T, Kashida Y, Machida N, Watanabe T, Mitsumori K (2004) Analysis of gene expression profiles of forestomach tumors in rasH2 mice initiated with N-ethyl-N-nitrosourea. Arch Toxicol 78:688–696
Okamura M, Unami A, Matsumoto M, Oishi Y, Kashida Y, Mitsumori K (2006) Gene expression analysis of urethane-induced lung tumors in rasH2 mice. Toxicology 217:129–138
Peraino C, Richards WL, Stevens FJ (1983) Multistage hepatocarcinogenesis. Environ Health Perspect 50:1–53
Porta GD, Dragani TA, Manenti G (1987) Two-stage liver carcinogenesis in the mouse. Toxicol Pathol 15:299–233
Rutenburg AM, Kim H, Fischbein JW, Hanker JS, Wasserkrug HL, Seligman AM (1969) Histochemical and ultranstructural demonstration of γ-glutamyl transpeptidase activity. Cytochem 17:517–526
Roskoski R Jr (2007) Vascular endothelial growth factor (VEGF) signaling in tumor progression. Crit Rev Oncol Hematol 62(3):179–213
Sakai H, Tsukamoto T, Yamamoto M, Shirai N, Iidaka T, Yanai T, Masegi T, Tatematsu M (2001) Differential effects of partial hepatectomy and carbon tetrachloride administration on induction of liver cell foci in a model for detection of initiation activity. Jpn J Cancer Res 92:1018–1025
Saltiel AR, Horikoshi H (1995) Thiazolidinediones are novel insulin-sensitizing agents. Curr Opin Endocrinol Diabetes 2:341–347
Sato K, Kitahara A, Satoh K, Ishikawa T, Tatematsu M, Ito N (1984) The placental form of glutathione S-transferase as a new marker protein for preneoplasia in rat chemical hepatocarcinogenesis. Gann 75:199–202
Shah V, Braverman R, Prasad GL (1998) Suppression of neoplastic transformation and regulation of cytoskeleton by tropomyosins. Somat Cell Mol Genet 24:573–580
Storer RD, Freench JE, Haseman J, Hajian G, Legrand EK, Long GG, Mixson LA, Ochoa R, Sagartz JE, Soper KA (2001) p53± Hemizygous knockout mose: Overview of available data. Toxicol Pathol 29:30–50
Tamaoki N (2001) The rasH2 transgnic mouse nature of the model and mechanistic studies on tumorigenesis. Toxicol Pathol 29:81–89
Tatematsu M, Mera Y, Ito N, Satoh K, Sato K (1985) Relative merits of immunohistochemical demonstration S-transferase and histochemical demonstration of γ-glutamyl transferase as markers of altered foci during liver carcinogenesis in rats. Carcinogenesis 6:1621–1626
Toyosawa K, Okimoto K, Kobayashi I, Kijima K, Kikawa E, Kohchi M, Koujitani T, Tanaka K, Matsuoka N (2001) Di (2-ethylhexyl)phthalate induces hepatocellular adenoma in transgenic mice carrying a human prototype c-Ha-ras gene in a 26-week carcinogenicity study. Toxicol Pathol 29:458–466
Usui T, Mutai M, Hisada S, Takoaka M, Soper KA, McCullough B, Alden C (2001) CB6F1-rasH2 mouse: overview of available data. Toxicol Pathol 209:90–108
Yamamoto S, Mitsumori K, Kodama Y, Matsunuma N, Manabe S, Okamiya H, Suzuki H, Fukuda T, Sakamaki Y, Sunaga M, Nomura G, Hioki K, Wakana S, Nomura T, Hayashi Y (1996) Rapid induction of more malignant tumors by various genotoxic carcinogens in transgenic mice harboring a human prototype c-Ha-ras gene than in control non-transgenic mice. Carcinogenesis 17:2455–2461
Yoshida LS, Miyazawa T, Hatayama I, Sato K, Fujimoto K, Kaneda T (1993) Phosphatidylcholine peroxidation and liver cancer in mice fed a choline-deficient diet with ethionine. Free Radic Biol Med 14:191–199
Westra WH, Offerhaus GJ, Goodman SN, Slebos RJ, Polak M, Baas IO, Rodenhuis S, Hruban RH (1993) Overexpression of the p53 tumor suppressor gene product in primary lung adenocarcinomas is associated with cigarette smoking. Am J Surg Pathol 17:213–220
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, M., Saekusa, Y., Dewa, Y. et al. Hepatocarcinogenic susceptibility of rasH2 mice to troglitazone in a two-stage hepatocarcinogenesis model. Arch Toxicol 83, 173–181 (2009). https://doi.org/10.1007/s00204-008-0335-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-008-0335-5