Abstract
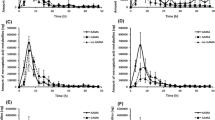
We investigated the human metabolism of AA to the mercapturic acids N-acetyl-S-(2-carbamoylethyl)-l-cysteine (AAMA) and N-(R/S)-acetyl-S-(2-carbamoyl-2-hydroxyethyl)-l-cysteine (GAMA) which are derived from AA itself and from its oxidative genotoxic metabolite glycidamide (GA), respectively. A healthy male volunteer received a single dose of about 1 mg deuterium-labelled acrylamide (d3-AA), representing 13 μg/kg body weight, in drinking water. Urine samples before dosing and within 46 h after the dose were analysed for d3-AAMA and d3-GAMA by LC-ESI-MS/MS. A first phase of increase in urinary concentration was found to last 18 h with a broad plateau between 8 and 18 h for AAMA, and 22 h for GAMA. Elimination half-lives of both AAMA and GAMA were estimated to be approximately 3.5 h for the first phase and more than 10 h up to few days for the second phase. Total recovery in urine after 24 h was about 51% as the sum of AAMA and GAMA and hereby well in accordance with former studies in rats. After 2 days AAMA, accounting for altogether 52% of the total AA dose, was the major metabolite of AA in humans. GAMA, accounting for 5%, appeared as a minor metabolite of AA. In humans we found a urinary ratio of 0.1 for GAMA/AAMA compared to previously reported values of 0.2 for rats and 0.5 for mice. Therefore, the metabolic fate of AA in humans was more similar to that in rats than in mice as already demonstrated in terms of the haemoglobin adducts. Consequently a genotoxic potency of AA mediated by GA could be supposed to be comparable in rats and humans.




Similar content being viewed by others
References
Bergmark E (1997) Hemoglobin adducts of acrylamide and acrylonitrile in laboratory workers, smokers and nonsmokers. Chem Res Toxicol 10(1):78–84
Bergmark E, Calleman CJ, He F, Costa LG (1993) Determination of hemoglobin adducts in humans occupationally exposed to acrylamide. Toxicol Appl Pharmacol 120(1):45–54
Boettcher MI, Angerer J (2005) Determination of the major mercapturic acids of acrylamide and glycidamide in human urine by LC-ESI-MS/MS. J Chrom B 824(1–2):283–294
Boettcher MI, Schettgen T, Kutting B, Pischetsrieder M, Angerer J (2005) Mercapturic acids of acrylamide and glycidamide as biomarkers of the internal exposure to acrylamide in the general population. Mutat Res 580(1–2):167–176
DFG (Deutsche Forschungsgemeinschaft) (2002) Maximale Arbeitsplatzkonzentrationen und biologische Arbeitsstoff Toleranzwerte 2002. Mitteilung XXXVIII der Senatskommission zur Prüfung gesundheitsschädlicher Arbeitsstoffe. Wiley-VCH, Weinheim
Dybing E, Farmer PB, Andersen M, Fennell TR, Lalljie SP, Muller DJ, Olin S, Petersen BJ, Schlatter J, Scholz G et al (2005) Human exposure and internal dose assessments of acrylamide in food. Food Chem Toxicol 43(3):365–410
EPA US. (Office of Toxic Substances; U.S. Environmental Protection Agency, ed) (1994) Chemical summary for acrylamide, Washington DC
Erdreich LS, Friedman MA (2004) Epidemiologic evidence for assessing the carcinogenicity of acrylamide. Regul Toxicol Pharmacol 39(2):150–157
EU European Commission (2001) Opinion on the results of the Risk Assessment of : acrylamide. European Commission, Scientific Committee on Food. Brussels
Fennell TR, Sumner SC, Snyder RW, Burgess J, Spicer R, Bridson WE, Friedman MA (2005) Metabolism and hemoglobin adduct formation of acrylamide in humans. Toxicol Sci 85(1):447–459
Granath F, Törnqvist M (2003) Who knows whether acrylamide in food is hazardous to humans? J Natl Cancer Inst 95(12):842–843
Hagmar L, Wirfalt E, Paulsson B, Törnqvist M (2005) Differences in hemoglobin adduct levels of acrylamide in the general population with respect to dietary intake, smoking habits and gender. Mutat Res 580(1–2):157–165
IARC (International Agency for Research on Cancer) (1994) Acrylamide. IARC-Summaries & Evaluations, vol 60, p 389
Larsen K.(1972) Creatinine assay by a reaction—kinetic principle. Clin Chim Acta 41:209–217
Li CM, Hu CW, Wu KY (2005) Quantification of urinary N-acetyl-S- (propionamide)cysteine using an on-line clean-up system coupled with liquid chromatography/tandem mass spectrometry. J Mass Spectrom
Miller MJ, Carter DE, Sipes IG (1982) Pharmacokinetics of acrylamide in Fisher-344 rats. Toxicol Appl Pharmacol 63(1):36–44
Mottram DS, Wedzicha BL, Dodson AT (2002) Acrylamide is formed in the Maillard reaction. Nature 419(6906):448–449
Mucci LA, Dickman PW, Steineck G, Adami HO, Augustsson K (2003) Dietary acrylamide and cancer of the large bowel, kidney, and bladder: absence of an association in a population-based study in Sweden. Br J Cancer 88(1):84–89
Schettgen T, Broding HC, Angerer J, Drexler H (2002) Hemoglobin adducts of ethylene oxide, propylene oxide, acrylonitrile and acrylamide-biomarkers in occupational and environmental medicine. Toxicol Lett 134(1–3):65–70
Schettgen T, Weiss T, Drexler H, Angerer J (2003) A first approach to estimate the internal exposure to acrylamide in smoking and non-smoking adults from Germany. Int J Hyg Environ Health 206(1):9–14
Schettgen T, Rossbach B, Kutting B, Letzel S, Drexler H, Angerer J (2004) Determination of haemoglobin adducts of acrylamide and glycidamide in smoking and non-smoking persons of the general population. Int J Hyg Environ Health 207(6):531–539
Shaw I, Thomson B (2003) Acrylamide food risk. Lancet 361(9355):434
Smith CJ, Perfetti TA, Rumple MA, Rodgman A, Doolittle DJ (2000) “IARC group 2A Carcinogens” reported in cigarette mainstream smoke. Food Chem Toxicol 38(4):371–383
Sumner SC, MacNeela JP, Fennell TR (1992) Characterization and quantitation of urinary metabolites of [1,2,3-13C]acrylamide in rats and mice using 13C nuclear magnetic resonance spectroscopy. Chem Res Toxicol 5(1):81–89
Sumner SC, Williams CC, Snyder RW, Krol WL, Asgharian B, Fennell TR (2003) Acrylamide: a comparison of metabolism and hemoglobin adducts in rodents following dermal, intraperitoneal, oral, or inhalation exposure. Toxicol Sci 75(2):260–270
Tareke E, Rydberg P, Karlsson P, Eriksson S, Törnqvist M (2002) Analysis of acrylamide, a carcinogen formed in heated foodstuffs. J Agric Food Chem 50(17):4998–5006
WHO (World Health Organisation) (2002) Health implications of acrylamide in food—report of a joint FAO/WHO consultation, Geneva, Switzerland
Acknowledgements
We especially thank the DFG (German Research Foundation) for their financial support of the project (AN 107/17-1 and 17-2).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boettcher, M.I., Bolt, H.M., Drexler, H. et al. Excretion of mercapturic acids of acrylamide and glycidamide in human urine after single oral administration of deuterium-labelled acrylamide. Arch Toxicol 80, 55–61 (2006). https://doi.org/10.1007/s00204-005-0011-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-005-0011-y