Abstract
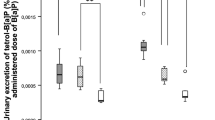
This study was aimed at investigating the correlation between biomarkers of exposure to polycyclic aromatic hydrocarbons and, more specifically, at examining the role of urinary 1-hydroxypyrene (1-OHP) as a reliable measure of internal dose linked to the electrophilic tissue burden (ETB), assessed as covalent binding of the ultimate carcinogen benzo(a)pyrene diolepoxide (BaPDE) with cellular proteins in target organs. The protocol included experimental verification of a previously proposed algorithm for adjustment of reference values for urinary 1-OHP with exposure to different mixtures of polycyclic aromatic hydrocarbons in a rat model. Hence, the relationships between ETB in liver, lung, and heart as well as the BaPDE–haemoglobin adducts level on the one hand, and urinary/faecal 1-OHP or urinary/faecal 3-hydroxybenzo(a)pyrene (3-OHBaP) on the other hand have been examined. Male Sprague-Dawley rats received intraperitoneally, once daily for 10 consecutive days, binary mixtures of benzo(a)pyrene (BaP) and pyrene (P) in three different exposure scenarios corresponding to BaP/P ratios of 0.2, 1 and 5, with three doses of BaP (2, 6 and 20 mg/kg) for each scenario. The ETB levels were measured as the ultimate analyte benzo(a)pyrene tetrol (BaPTeT) obtained after mild acid hydrolysis of BaPDE adducts with proteins. It was experimentally confirmed that: (1) urinary 1-OHP is a reliable biomarker linked to the ETB in tissues that are targets for carcinogenicity, such as lung, for the BaP/P ratios of 0.2 and 1 (linear regression p=0.0099 and 0.0293, respectively); (2) urinary 3-OHBaP is correlated with the BaPDE–haemoglobin adducts for all three exposure scenarios (p=0.0011 for BaP/P=0.2, p<0.0001 for BaP/P=1 and p=0.0099 for BaP/P=5). The experimental relationship between ETB and urinary 1-OHP was used to interpolate biological limit values for the urinary metabolite assuming three arbitrary critical levels of ETB. These were compared with the values calculated from the algorithm using the BaP/P ratio 1 mixture as a reference. The ratios of calculated to observed values varied from 1.0 to 1.6 for the BaP/P 0.2 mixture, and from 1.9 to 3.0 for the BaP/P 5 mixture. The results obtained in the present study indicate that the algorithm mentioned above applies well for two of the three exposure scenarios corresponding to realistic occupational BaP/P ratios of 0.2 and 1. This suggests that, using ETB as an endpoint, the proposed algorithm will reasonably predict the critical value of urinary 1-OHP for mixtures having different BaP/P ratios. Stronger linear relationships between ETB in all chosen tissues and 1-OHP or 3-OHBaP excretion were obtained with urinary metabolites than with their faecal analogues. Thus urinary 1-OHP and 3-OHBaP are more reliable biomarkers in biological monitoring strategies.




Similar content being viewed by others
References
Angerer J, Mannschreck C, Gundel J (1997) Biological monitoring and biochemical effect monitoring of exposure to polycyclic aromatic hydrocarbons [Review]. Int Arch Occup Environ Health 70:365–377
Bechtold WE, Sun JD, Wolff RK, Griffith WC, Kilmer JW, Bond JA (1991) Globin adducts of benzo(a)pyrene: markers of inhalation exposure as measured in F344/N rats. J Appl Toxicol 11:115–118
Bieniek G (1998) Aromatic and polycyclic hydrocarbons in air and their urinary metabolites in coke plant workers. Am J Ind Med 34:445–454
Bouchard M, Viau C (1995) Benzo(a)pyrenediolepoxide-hemoglobin adducts and 3-hydroxybenzo(a)pyrene urinary excretion profiles in rats subchronically exposed to benzo(a)pyrene. Arch Toxicol 69:540–546
Bouchard M, Viau C (1996) Urinary excretion of benzo[a]pyrene metabolites following intravenous, oral, and cutaneous benzo[a]pyrene administration. Can J Physiol Pharmacol 75:185–192
Bouchard M, Viau C (1999) Urinary 1-hydroxypyrene as a biomarker of exposure to polycyclic aromatic hydrocarbons: biological monitoring strategies and methodology for determining biological exposure indices for various work environments. Biomarkers 4:159–187
Bouchard M, Dodd C, Viau C (1994) Improved procedure for the high-performance liquid chromatographic determination of monohydroxylated PAH metabolites in urine. J Anal Toxicol 18:261–264
Bouchard M, Krishnan K, Viau C (1998) Urinary excretion kinetics of 1-hydroxypyrene following intravenous administration of binary and ternary mixtures of polycyclic aromatic hydrocarbons in the rat. Arch Toxicol 72:475–482
Bouchard M, Thuot R, Carrier G, Viau C (2002) Urinary excretion kinetics of 1-hydroxypyrene in rats subchronically exposed to pyrene or polycyclic aromatic hydrocarbon mixtures. J Toxicol Environ Health A 65:1195–1209
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chipman JK, Hirom PC, Frost GS, Millburn P (1981) The biliary excretion and enterohepatic circulation of benzo(a)pyrene and its metabolites in the rat. Biochem Pharmacol 30:937–944
Chipman JK, Millburn P, Brooks TM (1983) Mutagenicity and in vivo disposition of biliary metabolites of benzo(a)pyrene. Toxicol Lett 17:233–240
dell’Omo M, Muzi G, Marchionna G, Latini L, Carrieri P, Paolemili P, Abbritti G (1998) Preventive measures reduce exposure to polycyclic aromatic hydrocarbons at a graphite electrode plant. Occup Environ Med 55:401–406
Gerde, P, Muggenburg BA, Thornton-Manning JR, Lewis JL, Pyon KH, Dahl AR (1997) Benzo(a)pyrene at an environmentally relevant dose is slowly absorbed by, and extensively metabolized in, tracheal epithelium. Carcinogenesis 18:1825–1832
Gündel J, Schaller K-H, Angerer J (2000) Occupational exposure to polycyclic aromatic hydrocarbons in a fireproof stone producing plants: biological monitoring of 1-hydroxypyrene, 1-, 2-, 3- and 4-hydroxyphenanthrene, 3-hydroxybenz(a)anthracene and 3-hydroxybenzo(a)pyrene. Int Arch Occup Environ Health 73:270–274
IARC (International Agency for Research on Cancer) (1983) IARC Monographs on the evaluation of the carcinogenic risk of chemicals to humans, vol 32. Polynuclear aromatic compounds. Part 1. Chemical, environmental and experimental data. IARC Press, WHO, Lyon
IARC (International Agency for Research on Cancer) (1987) IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans. Supplement 7. Overall evaluations of carcinogenicity: an updating of the IARC monographs vols 1 to 42. IARC Press, Lyon
Jacob J, Seidel A (2002) Biomonitoring of polycyclic aromatic hydrocarbons in human urine. J Chromatogr B Analyt Technol Biomed Life Sci 778:31–47
Jongeneelen FJ (1992) Biological exposure limit for occuptional exposure to coal tar pitch volatiles at cokeovens. Int Arch Occup Environ Health 63:511–516
Jongeneelen FJ (1993) Biological exposure limit for occupational exposure to polycyclic aromatic hydrocarbons in the coke industry. Arch Toxicol 67:55–58
Jongeneelen FJ, Leijdekkers CM, Bos RP, Theuws JLG, Henderson PT (1985) Excretion of 3-hydroxy-benzo(a)pyrene and mutagenicity in rat urine after exposure to benzo(a)pyrene. J Appl Toxicol 5:277–282
Jongeneelen FJ, Bos RP, Anzion RBM, Theuws JLG, Henderson PT (1986) Biological monitoring of polycyclic aromatic hydrocarbons: metabolites in urine. Scand J Work Environ Health 12:137–143
Jongeneelen FJ, Anzion RBM, Henderson PT (1987) Determination of hydroxylated metabolites of polycyclic aromatic hydrocarbons in urine. J Chromatogr 413:227–232
Krewski D, Thorslund T, Withey J (1989) Carcinogenic risk assessment of complex mixtures. Toxicol Ind Health 5:851–867
Kuljukka T, Vaaranrinta R, Mutanen P, Veidebaum T, Sorsa M, Kalliokoski P, Peltonen K (1997) Assessment of occupational exposure to PAHs in an Estonian coke oven plant: correlation of total external exposure to internal dose measured as 1-hydroxypyrene concentration. Biomarkers 2:87–94
Lee BM, Santella RM (1988) Quantitation of protein adducts as a marker of genotoxic exposure: immunologic detection of benzo(a)pyrene–globin adducts in mice. Carcinogenesis 9:1773–1777
Levin JO (1995) First international workshop on hydroxypyrene as a biomarker for PAH exposure in man—summary and conclusions. Sci Total Environ 163:165–168
Lewtas J, Mumford J, Everson RB, Hulka B, Wilcosky T, Kozumbo W, Thompson C, George M, Dobias L, Sram R, Li X, Gallagher J (1993) Comparison of DNA adducts from exposure to complex mixtures in various human tissues and experimental systems. Environ Health Perspect 99:89–97
Mitchell CE (1982) Distribution and retention of benzo(a)pyrene in rats after inhalation. Toxicol Lett 11:35–42
Ny ET, Heederik D, Kromhout H, Jongeneelen F (1993) The relationship between polycyclic aromatic hydrocarbons in air and in urine of workers in a Soderberg potroom. Am Ind Hyg Assoc J 54:277–284
Ovrebo S, Haugen A, Fjeldstad PE, Hemminki K, Szyfter K (1994) Biological monitoring of exposure to polycyclic aromatic hydrocarbon in an electrode paste plant. J Occup Med 36:303–310
Peltonen K, Dipple A (1995) Polycyclic aromatic hydrocarbons: chemistry of DNA adduct formation. J Occup Environ Med 37:52–58
Petry T, Schmid P, Schlatter C (1996) Airborne exposure to polycyclic aromatic hydrocarbons (PAHs) and urinary excretion of 1-hydroxypyrene of carbon anode plant workers. Ann Occup Hyg 40:345–357
Poirier MC, Beland FA (1992) DNA adduct measurements and tumor incidence during chronic carcinogen exposure in animal models—implications for DNA adduct-based human cancer risk assessment. Chem Res Toxicol 5:749–755
Popp W, Vahrenholz C, Schell C, Grimmer G, Dettbarn G, Kraus R, Brauksiepe A, Schmeling B, Gutzeit T, von Bülow J, Norpoth K (1997) DNA single strand breakage, DNA adducts, and sister chromatid exchange in lymphocytes and phenanthrene and pyrene metabolites in urine of coke oven workers. Occup Environ Med 54:76–183
Sathirakul K, Suzuki H, Yamada T, Hanano M, Sugiyama Y (1994) Multiple transport systems for organic anions across the bile canalicular membrane. J Pharmacol Exp Ther 268:65–73
Shugart L, Kao J (1985) Examination of adduct formation in vivo in the mouse between benzo(a)pyrene and DNA of skin and hemoglobin of red blood cells. Environ Health Perspect 62:223–226
Simon P, Lafontaine M, Delsaut P, Morele Y, Nicot T (2000) Trace determination of urinary 3-hydroxybenzo[a]pyrene by automated column-switching high-performance liquid chromatography. J Chromatogr B 748:337–348
Tyndyk ML, Zabezhinski MA, Bykov VJ, Dikun PP, Dymochka LA, Nepomnyaschaya OB, Yatsuk OS, Yermilov VB, Likhachev AJ (1994) Individual values of excretion of benzo[a]pyrene metabolites and susceptibility to its carcinogenic effect in rats. Cancer Lett 78:163–170
Tzekova A, Leroux S, Viau C (2003) Electrophilic tissue burden in male Sprague-Dawley rats following repeated exposure to binary mixtures of polycyclic aromatic hydrocarbons. Arch Toxicol (in press) DOI: 10.1007/s00204-003-0518-z
van Welie RTH, van Dijck RGJM, Vermeulen NPE, Vansittert NJ (1992) Mercapturic acids, protein adducts, and DNA adducts as biomarkers of electrophilic chemicals. Crit Rev Toxicol 22:271–306
Viau C, Mercier M, Blondin O (1993) Measurement of hemoglobin and albumin adducts of benzo(a)pyrenediolepoxide and their rate of elimination in the female Sprague-Dawley rat. Arch Toxicol 67:468–472
Viau C, Bouchard M, Carrier G, Brunet R, Krishnan K (1999) The toxicokinetics of pyrene and its metabolites in rats. Toxicol Lett 108:201–208
Acknowledgements
This study was supported by the Canadian Institutes for Health Research. All experiments were conducted in accordance with the guidelines and principles of the Canadian Council on Animal Care.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tzekova, A., Thuot, R. & Viau, C. Correlation between biomarkers of polycyclic aromatic hydrocarbon exposure and electrophilic tissue burden in a rat model. Arch Toxicol 78, 351–361 (2004). https://doi.org/10.1007/s00204-003-0539-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-003-0539-7