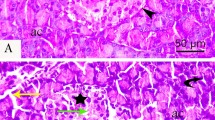
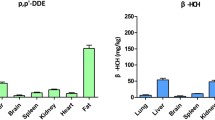
Abstract
Hexachlobenzene (HCB), one of the most persistent environmental pollutants, induces porphyria cutanea tarda (PCT). The aim of this work was to analyze the effect of HCB on some aspects of glucose metabolism, particularly those related to its neosynthesis in vivo. For this purpose, a time-course study on gluconeogenic enzymes, pyruvate carboxylase (PC), phosphoenolpyruvate carboxykinase (PEPCK), glucose-6-phosphatase (G-6-Pase) and on pyruvate kinase (PK), a glycolytic enzyme, was carried out. Plasma glucose and insulin levels, hepatic glycogen, tryptophan contents, and the pancreatic insulin secretion pattern stimulated by glucose were investigated. Oxidative stress and heme pathway parameters were also evaluated. HCB treatment decreased PC, PEPCK, and G-6-Pase activities. The effect was observed at an early time point and grew as the treatment progressed. Loss of 60, 56, and 37%, respectively, was noted at the end of the treatment when a considerable amount of porphyrins had accumulated in the liver as a result of drastic blockage of uroporphyrinogen decarboxylase (URO-D) (95% inhibition). The plasma glucose level was reduced (one-third loss), while storage of hepatic glucose was stimulated in a time-dependent way by HCB treatment. A decay in the normal plasma insulin level was observed as fungicide intoxication progressed (twice to four times lower). However, normal insulin secretion of perifused pancreatic Langerhans islets stimulated by glucose during the 3rd and 6th weeks of treatment did not prove to be significantly affected. HCB promoted a time-dependent increase in urinary chemiluminiscence (fourfold) and hepatic malondialdehide (MDA) content (fivefold), while the liver tryptophan level was only raised at the longest intoxication times. These results would suggest that HCB treatment does not cause a primary alteration in the mechanism of pancreatic insulin secretion and that the changes induced by the fungicide on insulin levels would be an adaptative response of the organism to stimulate gluconeogenesis. They showed for the first time that HCB causes impairment of the gluconeogenic pathway. Therefore, the reduced levels of glucose would thus be the consequence of decreased gluconeogenesis, enhanced glucose storage, and unaffected glycolysis. The impairment of gluconeogenesis (especially for PEPCK) and the related variation in glucose levels caused by HCB treatment could be a consequence of the oxidative stress produced by the fungicide. Tryptophan adds its effect to this decrease in the higher phases of HCB intoxication, where its levels overcome the control values possibly owing to the drastic decline of URO-D. This derangement of carbohydrates leads porphyric hepatocytes to have lower levels of free glucose. These results contribute to our understanding of the protective and modulatory effect that diets rich in carbohydrates have in hepatic porphyria disease.






Similar content being viewed by others
Abbreviations
- ALA-S :
-
δ-aminolevulinate synthase
- G-6-Pase :
-
glucose-6-phosphatase
- HCB :
-
hexachlorobenzene
- MDA :
-
malondialdehyde
- PEPCK :
-
phosphoenolpyruvate carboxykinase
- PCT :
-
porphyria cutanea tarda
- PC :
-
pyruvate carboxylase
- PK :
-
pyruvate kinase
- TCDD :
-
2,3,7,8-tetrachlorodibenzo-p-dioxin
- URO-D :
-
uroporphyrinogen decarboxylase
References
Abdel-Wahab YHA, O’Harte FPM, Barnett CR, Flatt PR (1996) Glycation of insulin in the islet of Langerhans of normal and diabetic animals. Diabetes 45:1489–1496
Baginski ES, Foa PP, Zak B (1974) Glucose-6-phosphatase. In: Bermeyer HU (ed) Method of enzymatic analysis, 2nd edn, vol 1, Verlag Chemie/Academic Press, Weinheim, New York, pp 876–880
Basabe JC, Karabatas LM, Arata M, Pivetta OH, Cresto JC (1986) Secretion and effect of somatostatin in early stages of the diabetic syndrome in C57BL/KsJ-mdb mice. Diabetologia. 29:485–488
Berndt J, Messner B, Still, J (1978) Optical assay of pyruvate carboxylase in crude liver homogenate. Anal Biochem 86:154–158
Billi de Catabbi S, Sterin-Speziale N, Fernandez MC, Minutolo C, Aldonatti C, San Martín de Viale L (1997) Time course hexachlorobenzene-induced alterations of lipid metabolism and their relation to porphyria. Int J Biochem Cell Biol 29:335–344
Billi de Catabbi SC, Aldonatti C, San Martín de Viale LC (2000) Heme metabolism after discontinued hexachlorobenzene administration in rats: possible irreversible changes and biomarker for hexachlorobenzene persistence. Comp Biochem Physiol Part C 127:165–175
Böger A, Koss G, Koransky W, Naummann R, Frenzel H (1979) Rat liver alteration after chronic treatment with hexachlorobenzene. Virchows Arch 382:127–137
Burr IM, Balant L, Stauffacher W, Renold AE, Grodsky G (1969) Dynamic aspects of proinsulin release from perifused rat pancreas. Lancet 2:882–883
Cheng KC, Cahill DS, Kasai H, Nishimura S, Loeb LA (1992) 8-hydroxy-guanine, an abundant form of oxidative DNA damage caused G→ T and A → C substitutions. J Biol Chem 267:166–172
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugar and related compound. Anal Chem 28:350–356
Elder GH (1998) Porphyria cutanea tarda. Semin Liver Dis 18:67–75
Ennamany R, Marzetto S, Saboreaud, D, Creppy EE (1995) Lipid peroxidation induced by bolesatine, a toxin of Boletus Satanas: implications in m5dC variation in Vero cells related to inhibition of cell growth. Cell. Biol. Toxicol. 11:347–354
Fan F, Rozman KK (1994) Relationship between acute toxicity of 2,3,7,8,-tetrachlorodibenzo-p-dioxin (TCDD) and disturbance of intermediary metabolism in the Long-Evans rat. Arch Toxicol 69:73–78
Fan F, Rozman KK (1995) Short- and long-term biochemical effects of 2,3,7,8-tetrachlorodibenzo-p- dioxin in female Long-Evans rats. Toxicol Lett 75:209–216
Ferioli A, Harvey C, De Matteis F (1984) Drug induced accumulation of uroporphyrin in chicken hepatocytes cultures. Biochem J 224:769–777.
Gorski JR, Rozman K (1987) Dose-response and time course of hypothyroxinemia and hypoinsulinemia and characterization of insulin hypersensitivity in 2,3,7,8-tetrachlorobenzo-p-dioxin (TCDD) treated rats. Toxicology 44:297–307
Granner DK, Andreone, TL (1985) Insulin modulation of gene expression. Diabet Metab Rev 1:139–170
Guyton KZ, Lui Y, Gorospe M, Xu Q, Holbrook NJ (1996) Activation of mitogen- activated protein kinase by H2O2. Role in cell survival following oxidant injury. J Biol Chem 271:4138–4142
Hanson RW (1997) Regulation of phosphoenolpyruvate carboxykinase (GTP) gene expression .Annu Rev Biochem 66:581–611
Herbert V, Lau K, Gottlieb CW (1965) Coated charcoal immunoassay of insulin. J Clin Endocrinol Metab 25:1375–1384
Hers HG, Hue L (1983) Gluconeogenesis and related aspects of glycolysis. Annu Rev Biochem 52:617–653
Horváth ME, Faux SP, Smith AG, Blazovics A, van der Looij, Fehér J, Cheeseman KH (2001) Vitamin E protects against iron-hexachlorobenzene induced porphyria and formation of 8-hydroxydeoxyguanosine in liver of C57BL/10 Sc5n mice. Toxicol Lett 122:97–102
Imamura K, Tanaka T (1982) Pyruvate kinase isozymes from rat. In: Wood WA (ed) Methods in enzymology, vol 90: Carbohydrate metabolism, part E. Academic Press, New York, pp 150–165
Ivanov DE, Chernev K, Adjarov D, Krustev L, Georgiev G (1976) The effect of different diets on porphyrin metabolism and microsomal liver enzyme induction in experimental hexachlorobenzene porphyria. In: Porphyrins in human diseases. 1st Int. Porphyrin Meet. Freiburg/Br 1975. Karger, Basle, pp 438–444
Kappas A, Sassa S, Galbraith RA, Nordmann Y (1995) The porphyrias. In: Scriver CR, Beaudet AL, Sly WS, Valle D, Stanbury JB, Wibgaarden JB, Fredickson DS (eds) Metabolic and molecular basis of inherited disease. McGraw-Hill, New York, pp 2103–2159
Koss G, Koransky W (1975). Studies on the toxicology of hexachlorobenzene. I. Pharmacokinetics. Arch Toxicol 34:203–212
Koss G, Seubert S, Seubert A, Koransky W, Ippen H (1978). Studies on the toxicology of hexachlorobenzene. III. Observation in a long term experiment. Arch Toxicol 40:285–294
Kószó F, Horvath L, Simon N, Siklosi C, Kiss, M (1982) The role of possible membrane damage in porphyria cutanea tarda: a spin label study of rat liver cell membranes. Biochem Pharmacol 31:11–17
Kuiper-Goodman T, Grant DL, Moodie CA, Korsrud GO, Munro IC (1977) Subacute toxicity of hexachlorobenzene in the rat. Toxicol Appl Pharmacol 40:529–549
Lacy PE, Kostianovsky M (1967) Method for the isolation of intact islets of Langerhans from the rat pancreas. Diabetes 16:35–39
Lim CK, Rideout JM, Wright DJ (1983) Separation of porphyrins isomers by high-performance liquid chromatography. Biochem J 211:435–438
Lowry OH, Rosebrough NJ, Randall RJ (1951) Protein measurement with the folin phenol reagement. J Biol Chem 193:265–275
Mollenhauer HH, Jhonson JH, Younger RL, Clark DE (1975) Ultraestructural changes in the liver fed rat hexachlorobenzene. Am J Vet Res 37:1777–1781
Moran GR, Fitzpatrick PF (1999) A continuous fluorescence assay for trypthophan hydrolase. Anal Biochem 266:148–152
Morel Y, Barouki R (1998) Down-regulation of cytochrome P450 1A1 gene promoter by oxidative stress. Critical contribution of nuclear factor 1. J Biol Chem 273:26969–26976
Petrescu I, Bojan O, Saied M, Barzu O, Schmidt F, Kühnle HF (1979) Determination of phosphoenolpyruvate carboxykinase activity with deoxyguanosine 5’-diphosphate as nucleotide substrate. Anal Biochem 96:279–281
Pilkis SJ, Fox E, Wolfe L, Rothbarth L, Colosia A, Stewart HB, El-Maghrabi MR (1986) Hormonal modulation of key hepatic regulatory enzymes in the gluconeogenic/glycolytic pathway. Ann NY Acad Sci 478:1-19
Ríos de Molina MC, Iglesias S, Lauría L, San Martín de Viale LC (1996) Precancerous pathology evoked by hexachlorobenzene treatment. Acta Physiol Pharmacol Latinoam 46:71–87
Ríos de Molina MC, Suarez Lissi MA, Armesto A, Lafourcade C, Lissi E (1998) Rat urinary chemoluminiscence. Biolumin Chemilumin 13:63–68
Rose JA, Hellman ES, Tschudy DP (1961) Effect on diet on induction of experimental porphyria. Metabolism 10:514–521
Rozman K, Mueller W, Coulson, F, Korte F (1977) Long-term feeding study of hexachlorobenzene in Rhesus monkeys. Chemosphere 3:81–84
San Martín de Viale LC, Tomio JM, Ferramola AM, Sancovich AH, Tigier HA (1975) Experimental porphyria induced in rats by hexachlorobenzene. Studies on enzyme associated with heme pathway. Effect of 17-β-estradiol. In: Doss M (ed) First Int Porphyrin Meet: Porphyrins in human diseases. Karger, Basle, pp 453–458
San Martín de Viale LC, Ríos de Molina MC, Wainstok de Calmanovici R, Tomio, JM (1977) Porphyrins and porphyrinogen carboxylyase in hexachlorobenzene induced porphyria. Biochem J 168:393–400
Smith AG, De Matteis F (1980) Drugs and hepatic porphyrias. Clin Haematol 9:399 −425
Smith SA, Pogson CI (1977) Tryptophan and the control of plasma glucose concentrations in the rat. Biochem J 168:495–506
Stahl BH, Beer DG, Weber LWD, Rozman K (1993) Reduction of hepatic phosphoenolpyruvate carboxykinase (PEPCK) activity by 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) is due to decreased mRNA levels. Toxicology 79:81–95
Tschudy DP, Welland F, Collins A, Hunter G (1964) The effect of carbohydrate feeding of δ-aminolevulinic acid synthetase. Metabolism 13:396–406
Vaena de Avalos S, Martini CN, San Martin de Viale LC (1998) Effect of hexachlorobenzene on steroids synthesis in adrenal cortex of rats. Proceedings of the XXXIV Annual Meeting of the Argentinian Society of Biochemistry and Molecular Biology (Mendoza) M 175:34
Wainstok de Calmanovici R, Rios de Molina MC, Tomio JM, San Martín de Viale, LC (1984) Mechanism of hexachlorobenzene induced porphyria in rats. effect of phenobarbitone pretreatment. Biochem J 218: 753–763
Wainstok de Calmanovici R, Cochón A, Aldonatti C, Cabral JR, San Martín de Viale LC (1991) Sex comparasion of heme pathway in rays bearing hepatic tumors. Tumori 77:379–384
Weber LWD, Lebofsky M, Greim H, Rozman K (1991) Key enzymes of gluconeogenesis are dose-dependently reduced in 2,3,7,8-tetrachlodibenzo-p-dioxin (TCDD)-treated rats. Arch Toxicol 65:119–123
Weber LWD, Stahl BU, Rozman K (1992) Are serotonergic mechanisms involved in the acute toxicity of chlorinated dibenzo-p-dioxins (CDDs). Chemosphere 25:161–164
Wilson J, Kueveruwa SS (1994) Toxicological profile for hexachlorobenzene. Update. US Department of Health and Human Services. Public Health Service Agency for Toxic Substances Disease Registry
Wolf WA, Kuhn DM (1983) Simultaneous determination of 5-H TRP, its amino acid precursors and acid metabolite in discrete brain regions by HPLC with fluorescence detection. J Chromatog 275:1-9
Wolff SP, Dean RT (1986) Fragmentation of proteins by free radicals and its effect on their susceptibility to enzyme hydrolysis. Biochem J 234:299–403
Acknowledgements
This work was supported by grants from the University of Buenos Aires and CONICET. L.C. San Martín de Viale and J.C. Basabe are Scientific Research Career members of the CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas). Procedures involving animals (care and use) were conducted according to international guidelines (Guide for Care and Use of Laboratory Animals, National Research Council, USA, 1996, and the Council of the European Communities Directive, 86/609/ECC).
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00204-003-0512-5
Rights and permissions
About this article
Cite this article
Mazzetti, M.B., Taira, M.C., Lelli, S.M. et al. Hexachlorobenzene impairs glucose metabolism in a rat model of porphyria cutanea tarda: a mechanistic approach. Arch Toxicol 78, 25–33 (2004). https://doi.org/10.1007/s00204-003-0470-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-003-0470-y