Abstract
The River Nile is the main source of fresh water in Egypt, where its water is used for irrigation, drinking, fisheries, industrial uses, and recreation. For sustainable utilization of the River Nile and its branches in the Nile Delta region, it is necessary to monitor regular investigation for the biodiversity of protozoan fauna in the Damietta branch and other freshwater canals in Dakahlyia Governorate. Water samples were collected monthly from different water sources, for 1 year, and examined for protozoans, using phase-contrast microscopy and recorded video films, The genus Vannella Bovee 1965 is recorded for the first time in four freshwater localities: Demietta branch of the River Nile, Mansouria Canal, Bouhia Canal, and Bahr El-Saghir Canal. A detailed morphological description with a brief report of their locomotion has been given for four morphologically different Vannella species. The locomotive form of Vannella sp.1 has a long pointed posterior tail and 2 lateral posterior processes. Such a tail was absent in other Vannella species. Vannella sp.2 is unique among other recorded species, where its locomotive form possesses a long posterior rounded tail region and a frontal hyaloplasm provided with a wavy surface that forms several lobes and finger-like processes during locomotion. In addition, the hyaloplasm produces several transverse waves that vary in thickness and density. The floating form of Vannella sp.2 is of a radial type and has comparatively long hyaline pointed and spiral pseudopodia. The process of transformation of locomotive form to floating form in Vannella sp.2 has been followed up using several recorded video films. The locomotive form of Vannella sp.3 is bear-shaped, while that of Vannella sp.4 has variable shapes from semicircular to rectangular and sometimes fan-shaped. During movement in vivo, locomotive cells of all Vannella species, except Vannella sp.1, move in nearly a straight line, but there were variations in their rate of locomotion. Vannella sp.4 recorded the highest rate (6.8 µm/s), followed by Vannella sp.2 (4.5 µm/s), Vannella sp.3 (2.4 µm/s), and finally Vannella sp.1 (1.0 µm/s). Molecular studies and transmission electron microscope examinations are still needed to confirm the precise identity of each Vannella species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The River Nile is the main source of fresh water in Egypt, where its water is used for different purposes, including irrigation, drinking, fisheries, and industrial uses (Ali et al. 2014; Abdel Galil et al. 2020). Damietta Branch of the river Nile provides many Governorates in Egypt (Dakahlyia, Qaluobyia, Damietta, and Gharbyia) with their need for drinking and irrigation waters (Abdo 2004). Most previous studies on the water quality of the River Nile were concentrated on the physicochemical and heavy metal parameters (Abdel-Satar 2005; Abdel-Rahim et al. 2013; Khallaf et al. 2014; Ali et al. 2014; Abdel Galil et al. 2020) and the effect of pollution on the zooplankton densities and distribution (El-Shabrawy et al. 2005; Hegab 2010; Gaber 2013; Khalifa and Bendary 2016; Fishar et al. 2019). For sustainable utilization of the River Nile and its branches in the Nile Delta region, it was found necessary to monitor regular investigation for protozoan fauna in the Damietta branch and other freshwater canals in Dakahlyia Governorate. Added interest is that most of these canals (Mansouria, Bouhia, and Bahr El-Saghir canals) were not previously examined for Protozoa, although they represent the main source of water for three important treatment plants, namely: Aga, El-Senbellaween, and Meniette El-Nasr, respectively. During the study of the biodiversity of aquatic protozoan fauna in these localities, members of lobose amoebae were isolated and identified as Vannella species.
The genus Vannella Bovee 1965 belongs to the family Vannellidae, order Vannellida and subclass Flabellinia of the class Discosea (Smirnov et al. 2011). The genus represents one of the most common genera in marine and freshwater habitats (Page 1988; Ariza et al. 1989; Smirnov and Goodkov 1995; Smirnov and Brown 2000; Smirnov et al. 2007). During locomotion, these amoebae appear flattened, fan-shaped, semicircular, crescent-shaped, or spatulate (Bovee 1965; Mesentsev et al. 2021). The cells move as a whole and never form discrete pseudopodia. The frontal hyaloplasm is flattened and occupies up to half the length of the cell. The breadth is usually greater than the length, making the cell appear fan-shaped (Smirnov and Goodkov 1999). In some cells, the hyaloplasm extends laterally to cover the sides of the cell, while in others, the cell appears spatulate with a long tail. There are no longitudinal wrinkles on the body surface. The floating forms of all freshwater species and most marine species are provided with markedly tapered pseudopodia, some of them are pointed, while others are tightly helical. Vannella with a cyst form has been recorded in some species, such as Vannella persistens, isolated from upland grassland soil in Sourhope, Scotland (Smirnov and Brown 2000) and Vannella pentlandii from course pasture in the Pentland Hills, Scotland (Maciver et al. 2017). At the ultrastructural level, the cell coat of most identified species has pentagonal glycostyles slightly more than 100 nm tall (Smirnov et al. 2007).
Amoebae of the genus Vannella are widely distributed and are among the most common organisms in environmental samples. However, species diversity within this genus requires further attention (Page 1988, 1991). To date, more than 32 valid species of the genus Vannella have been described. The present study provides a full description of four Vannella species with different characters using a phase-contrast microscope and video analysis. Moreover, assessing locomotion characteristics and the speed of free-living amoeba appears necessary to measure their capability of hunting their prey (Claußen and Schmidt 2017). However, further investigations using transmission electron microscopy (TEM) and molecular analysis are still needed to give a precise identity at the species level.
Materials and methods
Water samples were collected monthly from April 2017 to March 2018 using sterilized 1 L polypropylene containers 30 cm below the water surface from inlets (influents) of five different water treatment plants: Mansoura East and Sherbeen (on Damietta branch), Aga (on Mansouria canal), Meniette El-Nasr (on Bahr El-Saghir canal) and El-Senbellaween (on Bouhia canal) (Fig. 1). The collected samples were directly transferred to the Invertebrate Laboratory, Zoology Department, Faculty of Science, Mansoura University. Each water sample was filtered through a zooplankton cellulose acetate membrane net of 0.45 μm mesh diameter using a magnetic funnel and suction pump. The filtrate was diluted in 10 mL filtered freshwater and centrifuged at 2000 rpm. A drop of the precipitate was transferred to a clean slide, covered with a coverslip, examined using a Leitz Laborlux 20 EB phase-contrast research microscope, and photographed with an Omax (18 Megapixels) digital camera. Moreover, video films were prepared for each identified organism, and the locomotion was followed up. To establish the average speed of each Vannella species, a fixed point of the body, for example, the leading region was selected to measure the distance covered over a specific time using individual pictures of a movie sequence. Identification was done according to Smirnov et al. (2007). At least twenty locomotives and five floating cells of each Vannella species were examined and measured in vivo. All measurements and scale bars were calculated using the OMAX TopView 3.7 program.
Results
Four species of the genus Vannella Bovee 1965 were detected.
Systematic position according to Smirnov et al. (2011)
Phylum Amoebozoa Lühe 1913 sensu Cavalier-Smith 1998.
Subphylum Lobosa Carpenter, 1861 sensu Cavalier-Smith 2009.
Class Discosea Cavalier-Smith 2004 sensu Smirnov et al. 2011.
Subclass Flabellinia Smirnov et al. 2005 sensu Smirnov et al. 2011.
Order Vannellida Smirnov et al. 2005.
Family Vannellidae Bovee 1979.
Genus Vannella Bovee 1965.
Vannella sp.1 (Figs. 2a–c, 3a–f, S1)
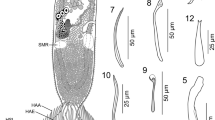
Phase-contrast microscope photographs of Vannella sp.1. a–c Locomotive form. Scale bars = 5 μm. d–f Floating form. Scale bars = 10 μm for (d) and 20 μm for (e, f). lsps long spiral pseudopodium, pps pointed pseudopodium. Other abbreviations as in Fig. 2
The locomotive form of Vannella sp.1 varies from semicircular and crescent-shaped to fan-shaped (Figs. 2a, b, 3a–c). The most abundant form is the fan-shaped and the cytoplasm is differentiated into hyaloplasm and granuloplasm. The cell possesses a pronounced long posterior tail (Figs. 2a, 3a, b). In some specimens, other postero-lateral processes are seen on the posterior straight edge (Figs. 2b, 3c). Measurements of all organs are shown in Table 1. The length of the locomotive form is 18.6(17–20) μm, while its maximum breadth is 20(18.8–20) μm. The length-to-breadth ratio is 0.9:1. The hyaloplasm constitutes about half or two-thirds of the cell size. It extends laterally to cover the body surface and may reach the basal edge of the posterior body region. In most locomotory cells examined, the surface edge of the frontal and lateral hyaloplasm is smooth but in some cells, the surface appeared slightly wavy (Fig. 3b). The nucleus is vesicular (Figs. 2a, 3a) and measures 3.0(2.9–3.2) μm in diameter. It possesses a centrally located nucleolus measuring 1.8 μm in diameter. The contractile vacuole is nearly spherical (Figs. 2a, 3b, c) and measures 2.3(1.5–2.8) μm in diameter. The posterior tail is relatively long and measures 4.6(3.8–5.0) μm in length. During movement on the surface of the glass slide as a substratum, the free locomotive cells show variations in their shape and size (S1). The cells do not move in a straight line but deviate slightly on both sides The average speed of locomotion is 1.0(0.8–1.5) μm/s (S1).
The floating form is of a radial type, with a central body mass measuring 20(20–22) μm in diameter and several tapering hyaline pseudopodia (Figs. 2c, 3d–f). However, the appearance of the floating form varies between specimens and appears to be dependent on its developmental condition. Some floating forms have only one or two pseudopodia, which are rather short initially and later become elongated. The body of well-developed floating forms is almost stellate and has 4–8 tapering, straight hyaline pseudopodia (Figs. 2c, 3d, e), measuring 40.4(28–64) μm in length. Some pseudopodia are coiled or helical in their middle or distal parts (Fig. 3f) and measure 53(40–88) μm in length.
Vannella sp.2 (Figs. 4a–e, 5a–f, 6a–f, S2)
Schematic drawing of Vannella sp.2. a–c Locomotive form. Scale bars = 10 μm. c, d Floating form. Scale bars = 20 μm. fp finger-like process, fps finger-like pseudopodium, lo surface lobe, rt rounded tail region, ws wavy surface; 1,2,3, successive layers of hyaloplasm formed during locomotion. Other abbreviations as in Fig. 2
The locomotive form varies in shape from semicircular to fan-shaped (Figs. 4a–c, 5a–f). Its cytoplasm is differentiated into hyaloplasm and granuloplasm. The length of the locomotive form is 42.5(37.5–47.5) μm, while its maximum breadth is 41.7(40–42.5) μm (Table 1). The length-to-breadth ratio is 1.1:1. The hyaloplasm constitutes about half to two-thirds of the cell size. It extends laterally to cover most of the body surface but never reaches the surface of the basal projected rounded edge of the posterior body region (Figs. 4a, 5a). In most locomotory cells examined, the surface edge of the frontal and lateral hyaloplasm is wavy (Figs. 4c, 5c, d). During locomotion, the frontal and lateral surfaces of the hyaloplasm waver, and many surface lobes and finger-like processes are formed (Figs. 4a–c, 5c–e, S2). Moreover, the hyaloplasm produces several waves and appears to be differentiated into two or three successive regions that vary in thickness and density (Figs. 4a, b, 5e, f). The outer region often appears denser than the other regions. The granuloplasm is packed with many translucent food vacuoles and colored contents which may represent algae. The nucleus is of a vesicular type, spherical in outline, and measures 7.3(7.3–7.5) μm in diameter (Figs. 4a, 5a). It possesses a centrally located nucleolus measuring 4.2(3.8–4.8) μm in diameter. The contractile vacuole is nearly spherical and measures 7.3(6.5–8) μm in diameter (Figs. 4c, 5a). The posterior tail region is relatively long and rounded in outline (Figs. 4a, b, 5c, d) and measures 11.3(10–12.5) μm in length. The present video films (S2) provided a good opportunity to follow up the process of transformation of locomotive form into a floating one. In the first step, the locomotive form moves rapidly by the frontal hyaloplasm in an anterior direction with a locomotion rate of 4.5(3.2–6.1) μm/s. The rate of movement decreases, while a long spiral pseudopodium and two or sometimes three short finger-like pseudopodia are formed at the posterior rounded tail region (Fig. 6a). The long pseudopodium often extends terminally to form a thin spiral region. Meanwhile, the frontal side of the body (hyaloplasm region) performs a distinct wavy surface with lobes and finger-like processes (Figs. 4d, 6b–d). In the next step, several pointed pseudopodia (4–6) are formed beside the long spiral pseudopodium (Fig. 6d). The pointed pseudopodia measure 53.3(40–72) μm in length, while the long spiral pseudopodium measures 90.7(40–128) μm in length. At the final stage, the completely developed floating form is of a radial type, with a central body mass measuring 53.3(36–68) μm in diameter and several tapering and finger-like hyaline pseudopodia measuring 53.3(40–72) μm in length (Figs. 4e, 6e, f). However, the appearance of the floating form varies between specimens and seems to be dependent on its developmental condition.
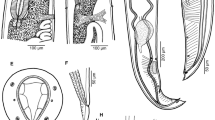
Vannella sp.3 (Figs. 7a, b, 8a–d, S3)
The locomotive form is mostly pear-shaped (Figs. 7a, 8a) and its length is 49.2(45–50) μm, while its maximum breadth is 39.2(35–40) μm. The length-to-breadth ratio is 1.3:1. The hyaloplasm constitutes about half of the cell size. It extends laterally to cover the body surface and never covers the edge of the posterior body region which extends posteriorly as a cone-shaped structure (Figs. 7a, 8c). The surface of the hyaloplasm is completely smooth. The granuloplasm fills most of the posterior body half and is packed with food vacuoles and a granular matrix. The nucleus is vesicular and measures 9.6(8.5–11.0) μm in diameter, while the nucleolus is 6.5(6.1–7.6) μm in diameter (Figs. 7a, 8b). The contractile vacuole is nearly spherical and measures 11.2(10.5–11.5) μm in diameter (Figs. 7a, 8b). The locomotive cell moves in a nearly straight line with the leading hyaloplasm anteriorly (S3). Little changes in shape and size were noticed during locomotion. The average rate of locomotion was 2.4 (2.3–2.8) μm/s. The floating form is of a radial type, with a central body mass measuring 15.3(13.8–17.5) μm in diameter and nearly 4 pointed hyaline pseudopodia measuring 38.3(25–50) in length (Figs. 7b, 8d).
Vannella sp.4 (Fig. 9a–f, S4)
Schematic drawing (a) and phase-contrast microscope photographs (b–f) of locomotive form of Vannella sp.4. Abbreviations as in Fig. 2. Scale bars = 10 μm
The present Vannella species was observed only in locomotive form. The floating form could be present but was not observed. The locomotive form varies in shape from semicircular to rectangular or sometimes fan-shaped (Fig. 9a–f). Its length is 24(17–32) μm, while its maximum breadth is 30.3(24–33.2) μm. The length-to-breadth ratio is 0.8:1. The hyaloplasm constitutes about one-third to two-thirds of the cell size (Fig. 9a–c). It extends laterally to cover the body surface but never reaches the basal edge bounding the posterior body region. The surface of the hyaloplasm is mostly smooth but sometimes wavy in some regions (Fig. 9a, c). The granuloplasm appears denser than the hyaloplasm since it is packed with food vacuoles and a granular matrix (Fig. 9a, e, f). The nucleus is vesicular and measures 5.7(5.4–6.0) μm in diameter, while the nucleolus is centrally located and measures 3.5(3.4–3.9) μm in diameter (Fig. 9a, e, f). In most specimens examined, more than one contractile vacuole (sometimes three) is present (Fig. 9a, c, e). Most of these vacuoles are spherical in outline and vary in size even in the same specimen. They measure 4.8(2.8–8.0) μm in diameter. During locomotion, the leading hyaloplasm moves in a nearly straight line and the locomotive cell shows variations in its shape and size (S4). Moreover, the frontal leading region of the cell increases in breadth and spreads over a larger area of the substratum with noticeable waving of the body surface (Fig. 9f). The average rate of locomotion is 6.8(6.3–7.5) μm/s.
Discussion
In the present study, four free-living species of the genus Vannella Bovee 1965 were collected from different water sources of Dakahlyia Governorate and described in detail using phase-contrast microscopy. As far as our knowledge is concerned, this is the first record of Vannella species in the freshwater sources of Dakahlyia Governorate of the Nile Delta region, Egypt. In Egypt, numerous studies were done on the protozoan diversity of the freshwater sources, particularly the River Nile, its two main branches (Demietta and Rosetta), and their tributaries. El-Serehy (1993) studied the protozoan fauna of Ismailia canal and recorded ciliates, flagellates, and dinoflagellates but had no record of sarcodines. Galal (2013) studied the protozoan diversity in the activated sludge at Benha waste-water treatment plant, in Kaluobeyia Province, and recorded some rhizopods, such as Amoeba proteus, A. striata, Arcella discoids, and Arcella vulgaris, and actinopods as Actinophrys sp. and Actinosphaerium sp. Again, there was no evidence of sarcodines, such as vannellid species. Galal and Nabet (2017) studied the protozoan fauna in 3 aquatic freshwater sources in El-Menoufeyia Province and recorded sarcodines belonging to the genera Amoeba, Arcella, and Nuclearia. Despite extensive studies made by Galal (2018) on the protozoan fauna of surface water from Rosetta and Damietta branches of the River Nile, Vannella species were not detected in these localities. Fishar et al. (2019) studied the community composition of zooplankton in El-Rayah El-Behery, Egypt, and recorded nine protozoans with no names except Vorticella. Galal et al. (2020) investigated the protozoan fauna of Mansoura East treatment plant in El-Mansoura city, Dakahlyia Governorate, Egypt, and recorded some sarcodines but without mentioning their precise genera or species.
The locomotive form of Vannella sp.1 is semicircular or fan-shaped and possesses a relatively long tail with a frontal hyaloplasm occupying about half of the cell and a smooth or wavy surface. The floating form of Vannella sp.1 is of a radial type with several hyaline-pointed pseudopodia and a few coiled spiral ones. In this respect, Vannella sp.1 may resemble Vannella simplex (Wohlfarth-Botterman 1960) Smirnov et al. (2002) in the previous characters but differs mainly in its smaller size and the absence of a cyst form. The average length of Vannella sp.1 is 18–20 μm, while that of V. simplex is 42–52 μm. Although Vannella sp.1 shares Vannella platypodia (Glaeser 1912) Page 1976 in the habitat, size, crescent shape of the frontal hyaloplasm, presence of a pronounced long tail, and absence of the cyst, most pseudopodia of the floating form of V. platypodia are spirally coiled. Moreover, V. platypodia is polymorphic and sometimes has a distinct long rounded tail (WoRMS 2023).
The most important findings that distinguish Vannella sp.2 from other recorded Vannella species of the present study is that the locomotive form has a long posterior rounded tail region and a frontal hyaloplasm with an obvious wavy surface that forms several lobes and finger-like processes during locomotion. Moreover, the hyaloplasm produces several transverse waves and differentiates into two or three successive regions with varying thickness and density. Similar but narrow transverse waves or so-called ripples were reported in Vannella douvresi (Sawyer 1975; Smirnov et al. 2007). These waves were found to flow anteriorly, often one after the other, and quickly disappear on reaching the anterior margin (Smirnov et al. 2007). Lateral waves (ripples) were not seen either in Vannella sp.2 or in V. douvresi, but waves or even short longitudinal ridges were observed on the frontal hyaloplasm of Vannella ebro (see Smirnov 2001). In Vannella croatica, Smirnov et al. (2016) reported that the moving cells form depressions on the ventral surface of the hyaloplasm and waves on its dorsal surface. Moreover, a prominent longitudinal ridge is formed for a short time in some cells. Neither ventral depressions nor longitudinal ridges have been seen in the present Vannella sp.2. However, old V. croatica share Vannella sp.2 in the formation of lobes on the dorsal surface of the hyaloplasm area particularly during locomotion. The floating form of Vannella sp.2 is of a radial type and has comparatively long hyaline pointed and spiral pseudopodia. In this respect, Vannella sp.2 appears to be unique among the currently described Vannella species and indeed among other previously described ones. However, Vannella sp.2 may resemble Vannella danica (Smirnov et al. 2002) Smirnov et al. (2007) in size and some internal structures but differs mainly in the habitat and cyst formation; V, danica is brackish and forms a cyst (Smirnov et al. 2002), while Vannella sp.2 is freshwater and have no cyst form. The appearance of the wavier surface, surface lobes, and finger-like processes in the locomotive form of Vannella sp.2 during locomotion might support the great role they may play in performing the movement action. These structures may enable the organism to proceed with its movement when it faces solid objects in water. In the present study, it was interesting to follow up, through video films, the process of transformation from locomotive to the floating form of Vannella sp.2. It was exciting to see the rapid movement of the locomotive form with a rate of 4.5(3.2–6.1) μm/s. The process included the formation of long spiral and short finger-like pseudopodia at the rounded tail region. With time, the pseudopodia increased in number (4–6) and the hyaloplasm region with its surface lobes disappeared. The completely formed floating form is of a radial type with a central mass and 4–7 hyaline, long-pointed, and spiral pseudopodia.
While the locomotive forms of both Vannella sp.1 and Vannella sp.2 are fan-shaped, the locomotive form of Vannella sp.3 is unique, being mostly pear-shaped and has a longer length if compared with other Vannella species. Moreover, the floating form of Vannella sp.3 has a few numbers of pointed pseudopodia, while those of Vannella sp.1 and Vannella sp.2 are numerous and differentiated into both spiral and pointed ones. Morphologically, no similarity has been found between Vannella sp.3 and the formerly described Vannella species (Smirnov et al. 2007). Therefore, molecular and electron microscope studies are recommended to explore the identity of this amoeba. The locomotive form of Vannella sp.4 has variable shapes from semicircular to rectangular and sometimes fan-shaped. It has a medium size if compared with other Vannella species and is the only species among the currently described Vannella to show more than one contractile vacuole. In addition, the floating form of this species has not been observed.
In the present study, it was interesting to record variations in the movement speed rate among described Vannella species. Vannella sp.4 recorded the highest rate (6.8 µm/s), followed by Vannella sp.2 (4.5 µm/s), Vannella sp.3 (2.4 µm/s), and finally Vannella sp.1 (1.0 µm/s). In this respect, the range of movement speed for Vannella sp.4, Vannella sp.3, and Vannella sp.2 is higher than that recorded for Acanthamoeba and Mayorella species (1.0 µm/s) reported by Claußen and Schmidt (2017). The relatively high speed of most Vannella spp. may help them to hunt down their prey, which includes some bacteria and protists. Moreover, variability in the speed rate among recorded Vannella species may reflect a difference in their behavior and probably their structure.
Previous studies on members of the genus Vannella did not show any indication that Vannella itself is pathogenic, like other free-living amoebae. However, Vannella can facilitate the growth of bacteria (Loret et al. 2008; Schulz et al. 2015), including Legionella (Kuroki et al. 1998) and other organisms (Scheid 2007), some of which are human pathogens. Vannella spp. could represent one of the sources of taste and odor problems in water (Sousa-Ramos et al. 2022). The record of a cyst stage in Vannella persistens supports the possibility of being pathogens since these cysts are protected as is known to be the case for other free-living amoebae (Smirnov and Brown 2000). In addition, Vannella is commonly found in the human water supply (Thomas et al. 2008; Poitelon et al. 2009), in domestic appliances (Rivera et al. 1993), and even in our crops (Chavatte et al. 2016). Molecular analysis and electron microscope studies are still needed to differentiate between recorded Vannella species and determine their precise identity. Preparation of these techniques is going on in our laboratory at Mansoura University and will be carried out as soon as possible.
Conclusion
The present study provides the first record of the genus Vannella in the River Nile Damietta Branch and other canals (Mansouria, Bouhia, and Bahr El-Saghir) which represent the sources of influent water for five treatment plants at Mansoura East, Sherbeen, Aga, El-Senbellaween and Meniette El-Nasr cities. Four Vannella species with different morphological characteristics were described in vivo at the light microscope level. The study revealed variations in the locomotion pattern and average speed during locomotion between different Vannella species. Therefore, studying the locomotion behavior of Vannellid species could be used as a taxonomical criterion differentiating between species of the genus Vannella. Further studies are recommended for other water canals in the Nile Delta region to reveal and identify the protozoan fauna of these communities. Molecular analysis and electron microscope studies are still needed to differentiate between recorded Vannella species and determine their precise identity.
Data availability
All data used in this study are available upon personal request to the authors.
References
Abdel Galil M, Hegazy TA, Hasaballah AF, Al-Madboly NM (2020) Chemical characteristics of the surface water around Ras El-Bar Island, Damietta Governorate. Egypt J Environ Sci 49(1):18–27. https://doi.org/10.21608/JOESE.2020.147763
Abdel-Rahim KAA, Hassanein AM, Younis MS, Heikal AA (2013) Correlation between physicochemical and microbiological characteristics of River Nile water in Sohag governorate, Egypt. J Pure Appl Microbiol 7:63–77
Abdel-Satar AM (2005) Water quality assessment of River Nile from Idfo to Cairo, Egypt. J Aquat Res 31(2):200–223
Abdo MH (2004) Environmental studies on the River Nile at Damietta branch region, Egypt. J Egypt Acad Soc Environ Dev 5(2):85–104
Ali EM, Shabaan-Dessouki SA, Soliman AI, El Shenawy AS (2014) Characterization of chemical water quality in the Nile River, Egypt. Int J Pure Appl Biosci 2(3):35–53
Ariza C, Guevara DAC, Ubeda JM, Cutillas C (1989) Description of four species of the genus Vannella isolated from freshwater. Microbiologia 5(1):25–33
Bovee EC (1965) An emendation of the ameba genus Flabellula and a description of Vannella gen. nov. Trans Amer Microsc Soc 84:217–227. https://doi.org/10.2307/3224288
Bovee EC (1979) Protozoa from acid-bog mosses and forest mosses of the Lake Itasca region (Minnesota, USA). Univ Kans Sci Bull 51:615–629
Cavalier-Smith T (1998) A revised six-kingdom system of life. Biol Rev Camb Philos Soc 73(3):203–266. https://doi.org/10.1017/s0006323198005167
Cavalier-Smith T (2009) Megaphylogeny, cell body plans, adaptive zones: causes and timing of eukaryote basal radiations. J Eukaryot Microbiol 56:26–33. https://doi.org/10.1111/j.1550-7408.2008.00373.x
Chavatte N, Lambrecht E, Van-Damme I, Sabbe K, Houf K (2016) Abundance, diversity, and community composition of free-living protozoa on vegetable sprouts. Food Microbiol 55:55–63. https://doi.org/10.1016/j.fm.2015.11.013
Claußen M, Schmidt S (2017) Biodegradation of phenol and p-cresol by the hyphomycete Scedosporium apiospermum. Res Microbiol 149(6):399–406. https://doi.org/10.1016/S0923-2508(98)80322-7
El-Serehy HA (1993) Species composition and distribution of sand-dwelling ciliates in the Great Bitter Lake of the Suez Canal. J Egypt Ger Soc (zool) 10(D):127–141
El-Shabrawy GM, Elowa ShE, Rizk ST, El-Fetouh OM (2005) Impact of industrial pollution on zooplankton community structure in Rosetta Nile Branch at Kafr El-Zayat area, Egypt. Afri J Biol Sci 1(1):1–14
Fishar MR, Mahmoud NH, El-Feqy FA, Gaber KMG (2019) Community composition of zooplankton in El-Rayah El-Behery, Egypt. EJABF 23(1):135–150. https://doi.org/10.21608/EJABF.2019.26815
Gaber KM (2013) Studies on the distribution and diversity of zooplankton in River Nile and its branches (Rosetta and Damietta branches), Egypt, 2013, M.Sc. Thesis, Fac. Sci., Al-Azhar Univ., Cairo, 198pp
Galal M (2013) Protozoan diversity in the activated sludge at Benha waste-water treatment plant, Kalubeyia Province, Egypt. JADES 2(1):1–8. https://doi.org/10.21608/jades.2020.73182
Galal MA (2018) Field studies on the protozoan distribution in Damietta and Rosetta branches of the River Nile, Egypt. J Egypt Acad Soc Environ Dev 19(1):33–41. https://doi.org/10.21608/JADES.2018.62430
Galal ME, Nabet KN (2017) Ecological evaluation of ciliated protozoa at Bahr-Shebeen canal, El-Menoufeyia province, Egypt. JBAAR 3(1):66–72
Galal M, Khallaf SA, Authman MN, El-Sebaae A (2020) Ecological studies on some planktonic organisms at Mahallet Ziad wastewater treatment plant in El-Gharbia Province, Egypt. JADES 2(1):1–10. https://doi.org/10.21608/jades.2020.73182
Glaeser H (1912) Untersuchungen ueber die Teilung einiger Amoeben. Arch Protistenkd 25:27–152
Hegab MH (2010) Effect of some environmental parameters on distribution of zooplankton and the relationship between them and food & feeding of some fish in Rosetta Branch of River Nile, Egypt, 2010, M.Sc. Thesis, Fac. Sci., Al-Azhar Univ., Cairo, 243pp
Khalifa NS, Bendary RE (2016) Composition and biodiversity of zooplankton and macrobenthic populations in El-Rayah El-Menoufiya, Egypt. Int J App Env Sci 11(2):683–700
Khallaf EA, Galal M, Elsbbagh S, Nabet NM (2014) A study of the physicochemical and biological characteristic of raw water, filtrated and treated water at a water treatment plant in Shebin Elkom, Menoufiya, Egypt. EJABF 18(1):115–125. https://doi.org/10.21608/EJABF.2014.2197
Kuroki T, Sata S, Yamai S et al (1998) Occurrence of free-living amoebae and Legionella in whirlpool baths. Kansenshogaku Zasshi 72(10):1056–1063. https://doi.org/10.11150/kansenshogakuzasshi1970.72.1056
Loret JF, Jousset M, Robert S et al (2008) Amoebae-resisting bacteria in drinking water: risk assessment and management. Water Sci Technol 58:571–577. https://doi.org/10.2166/wst.2008.423
Maciver SK, Valle ADOFD, Koutsogiannis Z (2017) Vannella pentlandii n. sp., (Amoebozoa, Discosea, Vannellida) a small, cyst-forming soil Amoeba. Exp Parasitol 183:109–116. https://doi.org/10.1016/j.exppara.2017.07.011
Mesentsev Y, Bondarenko N, Nassonova E, Smirnov A (2021) Vannella primoblina n. sp.—an unusual species of the genus Vannella (Amoebozoa, Discosea, Vannellida) with pronounced dorsal ridges and folds. Eur J Protistol 77:125757. https://doi.org/10.1016/j.ejop.2020.125757
Page FC (1991) Nackte Rhizopoda. In: Page FC, Siemensma FJ (eds) Nackte Rhizopoda and Heliozoea. Fischer Verlag, Stuttgart, pp 1–170
Page FC (1988) A new key to freshwater and soil gymnamoebae. Freshwater Biological Association, Ambleside
Poitelon JB, Joyeux M, Welte B et al (2009) Identification and phylogeny of eukaryotic 18S rDNA phylotypes detected in chlorinated finished drinking water samples from three Parisian surface water treatment plants. Lett App Microbiol 49:589–595. https://doi.org/10.1111/j.1472-765X.2009.02710.x
Rivera F, Ramírez E, Bonilla P et al (1993) Pathogenic and free-living amoebae isolated from swimming pools and physiotherapy tubs in Mexico. Environ Res 62:43–52. https://doi.org/10.1006/enrs.1993.1087
Sawyer TK (1975) Marine amoebae from surface waters of chincoteague bay, virginia: two new genera and nine new species within the families Mayorellidae, Flabellulidae and Stereomyxidae. Trans Am Microsc Soc 94:71–92
Scheid P (2007) Mechanism of intrusion of a microspordian-like organism into the nucleus of host amoebae (Vannella sp.) isolated from a keratitis patient. Parasitol Res 101:1097–1102. https://doi.org/10.1007/s00436-007-0590-z
Schulz F, Tyml T, Pizzetti I et al (2015) Marine amoebae with cytoplasmic and perinuclear symbionts deeply branching in the Gammaproteobacteria. Sci Rep 5:13381. https://doi.org/10.1038/srep13381
Smirnov AV (2001) Vannella ebro n. sp. (Lobosea, Gymnamoebia), isolated from cyanobacterial mats in Spain. Eur J Protistol 37:147–153. https://doi.org/10.1078/0932-4739-00811
Smirnov AV, Brown S (2000) First isolation of a cyst forming Vannella species, from soil—Vannella persistens n. sp. (Gymnamoebia, Vannellidae). Protistology 1:120–123
Smirnov AV, Goodkov AV (1995) Systematic diversity of gymnamoebae in the bottom sediments of a freshwater lake in Karelia (Lobosea, Gymnamoebia). Zoosyst Rossica 4(2):201–203
Smirnov AV, Goodkov AV (1999) An illustrated list of basic morphotypes of Gymnamoebia (Rhizopoda, Lobosea). Protistology 1:20–29
Smirnov AV, Nassonova ES, Holzmann M, Pawlowski J (2002) Morphological, ecological and molecular studies of Vannella simplex Wohlfarth-Bottermann 1960 (Lobosea, Gymnamoebia), with a new diagnosis of this species. Protistology 153:367–377. https://doi.org/10.1078/14344610260450109
Smirnov AV, Nassonova ES, Berney C, Fahrni J, Bolivar I, Pawlowski J (2005) Molecular phylogeny and classification of the lobose amoebae. Protist 156:129–142. https://doi.org/10.1016/j.protis.2005.06.002
Smirnov A, Nassonova ES, Chao E, Cavalier-Smith T (2007) Phylogeny, evolution and taxonomy of vannellid amoebae. Protist 158:295–324. https://doi.org/10.1016/j.protis.2007.04.004
Smirnov AV, Elena EC, Nassonova S, Cavalier-Smith T (2011) A revised classification of naked lobose amoebae (Amoebozoa: Lobosa). Protistology 162(4):545–570. https://doi.org/10.1016/j.protis.2011.04.004
Smirnov AV, Bondarenko N, Glotova A, Nassonova E (2016) Morphology and phylogeny of Vannella croatica n. sp. (Amoebozoa, Discosea, Vannellida). Eur J Protistol 52:65–72. https://doi.org/10.1016/j.ejop.2015.11.002
Sousa-Ramos D, Reyes-Batlle M, Bellini NK, Rodríguez-Expósito RL, Martín-Real C, Piñero JE, Lorenzo-Morales J (2022) Pathogenic free-living amoebae from water sources in Cape Verde. Parasitol Res 121(8):2399–2404. https://doi.org/10.1007/s00436-022-07563-y
Thomas V, Loret JF, Jousset M, Greub G (2008) Biodiversity of amoebae and amoebae-resisting bacteria in a drinking water treatment plant. Environ Microbiol 10(10):2728–2745. https://doi.org/10.1111/j.1462-2920.2008.01693.x
Wohlfarth-Botterman K (1960) Protistenstudien. X. Licht- und electronenmikroskopische Untersuchungen an der amoeba Hyalodiscus simplex n. sp. Protoplasma 52:58–107
WoRMS (2023) Vannella platypodia Glaser, 1912. https://www.marinespecies.org/aphia.php?p=taxdetails&id=120661. Accessed 17 Dec 2023
Acknowledgements
This paper is part of a PhD thesis to be submitted to the Zoology Department, Faculty of Science, Mansoura University, Egypt.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
AM, MM, ER, and Mel-N made a substantial contribution to the conception and design of the study. All authors contributed to data acquisition. AM collected water samples, examined the protozoa, and photographed them. AM, MM, ER, and Mel-N analyzed and interpreted the data. AM has written the first draft of the manuscript, and all authors revised it critically. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
No experiments were performed on live animals. All applicable institutional, national, and international guidelines were followed. Mansoura University ethics approval no Sci-Z-Ph-2017–2.
Consent to participate
The authors declare that they have participated in this work.
Consent for publication
The authors declare that they know the content of this manuscript and agreed to submit it to “Archives of Microbiology”.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 S1 Video film of locomotive form of Vannella sp.1 (AVI 13931 KB)
Supplementary file2 S2 Video film of locomotive form of Vannella sp.2 (MP4 7387 KB)
Supplementary file3 S3 Video film of locomotive form of Vannella sp.3 (AVI 6645 KB)
Supplementary file4 S4 Video film of locomotive form of Vannella sp.4 (MP4 5360 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Marzouk, A.M., Mashaly, M.I., Reda, E.S. et al. The record of Vannella species (Vannellidae, Discosea, Amoebozoa) from freshwater sources in Dakahlyia Governorate, Egypt. Arch Microbiol 206, 113 (2024). https://doi.org/10.1007/s00203-024-03837-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03837-4