Abstract
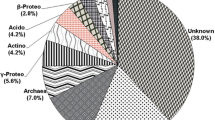
Pichavaram mangrove forest was established as a wetland of International Importance by Article 2.1 in April 2022 by the Ministry of Environment, Forest and Climate Change, India. Even though it is a conserved site, xenobiotic agrochemical leaching on the forest land during monsoon is inevitable. These threaten the microbial diversity in the environment. Xenobiotic degradation is achieved using bacterial consortia already acclimatised to this environment. This study aims to identify the indigenous microbial consortia able to degrade xenobiotic compounds such as fluorobenzoate, furfural, and steroids. Pichavaram mangrove metagenomic dataset was obtained by shotgun sequencing of soil DNA and processed using the automated tool SqueezeMeta. Further, the DIAMOND database provided the taxonomical classification of the microbes in each contig. With reference to the KEGG database, the selected xenobiotic degradation pathways were confirmed in the dataset. Of 1,253,029 total contigs, 1332, 72 and 1262 were involved in fluorobenzoate, furfural and steroid degradation, respectively. This study identified that microbial consortia comprising Marinobacter, Methyloceanibacter and Vibrio natriegens/Gramella sp. can degrade fluorobenzoate. While Afipia, Nitrosopumilus sp., and Phototrophicus methaneseepsis favour the degradation of furfural compound. The steroid degradation pathway possessed a plethora of bacteria belonging to the phylum Proteobacteria.




Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available in NCBI SRA database at https://www.ncbi.nlm.nih.gov/sra, reference number PRJNA1043081.
References
Aravind P et al (2019) Removal of BPA from thermal cash receipts via electro oxidation cum biodegradation: evaluating its degradation mechanism and in silico toxicity analysis. J Water Process Eng 31:100849. https://doi.org/10.1016/J.JWPE.2019.100849
Arora PK et al (2022) Current bioinformatics tools for biodegradation of xenobiotic compounds. Front Environ Sci. https://doi.org/10.3389/fenvs.2022.980284
Bergstrand LH et al (2016) Delineation of steroid-degrading microorganisms through comparative genomic analysis. Mbio. https://doi.org/10.1128/mbio.00166-16
Buchfink B, Xie C, Huson DH (2014) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12(1):59–60. https://doi.org/10.1038/nmeth.3176
Carvalho MF et al (2005) Isolation and properties of a pure bacterial strain capable of fluorobenzene degradation as sole carbon and energy source. Environ Microbiol 7(2):294–298. https://doi.org/10.1111/j.1462-2920.2004.00714.x
Carvalho MF et al (2006) Degradation of fluorobenzene by Rhizobiales strain F11 via ortho cleavage of 4-fluorocatechol and catechol. Appl Environ Microbiol 72(11):7413–7417. https://doi.org/10.1128/AEM.01162-06
Crowe AM et al (2017) ‘Catabolism of the last two steroid rings in Mycobacterium tuberculosis and other bacteria. Mbio. https://doi.org/10.1128/mBio.00321-17
Datta S et al (2020) Endophytic bacteria in xenobiotic degradation. Microbial endophytes: prospects for sustainable agriculture. Elsevier, pp 125–156. https://doi.org/10.1016/B978-0-12-818734-0.00006-1
Farías A et al (2022) Furfural biodegradation in consortium through Bacillus licheniformis, Microbacterium sp. and Brevundimonas sp. J Sustain Dev Energy Water Environ Syst. https://doi.org/10.13044/j.sdewes.d9.0392.
Holert J et al (2018) Metagenomes reveal global distribution of bacterial steroid catabolism in natural, engineered, and host environments. Mbio. https://doi.org/10.1128/mBio.02345-17
Kathiresan K (2000) A review of studies on Pichavaram mangrove, southeast India. Hydrobiologia 430:185–205
Kumari L, Shamsher KS (2015) Cholesterol oxidase: role in biotransformation of cholesterol. J Appl Biol Biotechnol. https://doi.org/10.7324/jabb.2015.3609
Maltseva OV et al (1994) Dienelactone hydrolase from Rhodococcus erythropolis I CP: purification and properties. Arch Microbiol 162(5):368–374
Mishra S et al (2021) Recent advanced technologies for the characterization of xenobiotic-degrading microorganisms and microbial communities. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2021.632059
Muñoz-García A et al (2019a) Influence of salinity on the degradation of xenobiotic compounds in rhizospheric mangrove soil. Environ Pollut 249:750–757. https://doi.org/10.1016/J.ENVPOL.2019.03.056
Murphy CD (2010) Biodegradation and biotransformation of organofluorine compounds. Biotechnol Lett 32(3):351–359. https://doi.org/10.1007/s10529-009-0174-3
Navgire GS et al (2022) Analysis and Interpretation of metagenomics data: an approach. Biol Proced Online. https://doi.org/10.1186/s12575-022-00179-7
Nikolova CN et al (2021) ‘Response and oil degradation activities of a northeast Atlantic bacterial community to biogenic and synthetic surfactants. Microbiome: https://doi.org/10.1186/s40168-021-01143-5
Russell JN et al (2021) Metagenomic and metatranscriptomic analysis reveals enrichment for xenobiotic-degrading bacterial specialists and xenobiotic-degrading genes in a Canadian Prairie two-cell biobed system. Environ Microbiol Rep 13(5):720–727. https://doi.org/10.1111/1758-2229.12990
Salghuna NN, Pillutla RCP (2017) Mapping mangrove species using hyperspectral data: a case study of Pichavaram mangrove ecosystem, Tamil Nadu. Earth Syst Environ. https://doi.org/10.1007/s41748-017-0024-8
Shah V et al (2013) Taxonomic profiling and metagenome analysis of a microbial community from a habitat contaminated with industrial discharges. Microb Ecol 66(3):533–550. https://doi.org/10.1007/s00248-013-0253-9
Tamames J, Puente-Sánchez F (2019) SqueezeMeta, a highly portable, fully automatic metagenomic analysis pipeline. Front Microbiol. https://doi.org/10.3389/fmicb.2018.03349
Teas HJ (1983) Biology and ecology of mangroves. Springer, Netherlands
Thatoi H et al (2013) Biodiversity and biotechnological potential of microorganisms from mangrove ecosystems: a review. Ann Microbiol 63(1):1–19. https://doi.org/10.1007/s13213-012-0442-7
Tomoyuki Morita KOAMKTTHFM (2005) Female sex hormone degrading bacteria and use thereof
Ullah R, Zafar MS, Shahani N (2017) ‘Potential fluoride toxicity from oral medicaments: a review. Iran J Basic Med Sci 20(8):841–848. https://doi.org/10.22038/ijbms.2017.9104
Vartoukian SR, Palmer RM, Wade WG (2010) Strategies for culture of “unculturable” bacteria. FEMS Microbiol Lett. https://doi.org/10.1111/j.1574-6968.2010.02000.x
Wang X et al (2023) Systematic characterization of sediment microbial community structure and function associated with anaerobic microbial degradation of PBDEs in coastal wetland. Mar Pollut Bull 188:114622. https://doi.org/10.1016/J.MARPOLBUL.2023.114622
Wierckx N et al (2011) Microbial degradation of furanic compounds: biochemistry, genetics, and impact. Appl Microbiol Biotechnol 92(6):1095–1105. https://doi.org/10.1007/s00253-011-3632-5
Acknowledgements
The authors would like to thank SRM Insitute of Science and Technology for supporting the research. We are grateful for the continuous support and encouragement of our Department of Biotechnology SRMIST colleagues. Also, we thank the SRM-HPCC for supporting software and platforms to analyse the metagenomic data.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
LMS & PK— conceptualization, data curation, formal analysis, investigation, methodology, supervision, software, validation, visualization, writing original draft, review and editing. IV & BB — data curation and formal analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest or any financial competing interests associated with submitting this manuscript.
Consent for publication
The authors declare that this manuscript is not under consideration for publication elsewhere.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kannan, P., Verma, I., Banerjee, B. et al. Unveiling bacterial consortium for xenobiotic biodegradation from Pichavaram mangrove forest soil: a metagenomic approach. Arch Microbiol 206, 27 (2024). https://doi.org/10.1007/s00203-023-03765-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03765-9