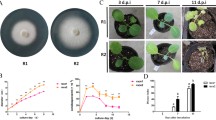
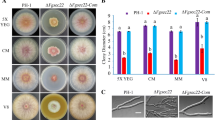
Abstract
Fusarium oxysporum f. sp. lycopersici is a soil-borne phytopathogenic species which causes vascular wilt disease in the Solanum lycopersicum (tomato). Due to the continuous competition for zinc usage by Fusarium and its host during infection makes zinc-binding proteins a hotspot for focused investigation. Zinc-binding effector proteins are pivotal during the infection process, working in conjunction with other essential proteins crucial for its biological activities. This work aims at identifying and analysing zinc-binding proteins and zinc-binding proteins effector candidates of Fusarium. We have identified three hundred forty-six putative zinc-binding proteins; among these proteins, we got two hundred and thirty zinc-binding proteins effector candidates. The functional annotation, subcellular localization, and Gene Ontology analysis of these putative zinc-binding proteins revealed their probable role in wide range of cellular and biological processes such as metabolism, gene expression, gene expression regulation, protein biosynthesis, protein folding, cell signalling, DNA repair, and RNA processing. Sixteen proteins were found to be putatively secretory in nature. Eleven of these were putative zinc-binding protein effector candidates may be involved in pathogen–host interaction during infection. The information obtained here may enhance our understanding to design, screen, and apply the zinc-metal ion-based antifungal agents to protect the S. lycopersicum and control the vascular wilt caused by F. oxysporum.






Similar content being viewed by others
Data availability
The data are already provided in the manuscript as supplementary files.
References
Almagro Armenteros JJ, Salvatore M, Emanuelsson O et al (2019) Detecting sequence signals in targeting peptides using deep learning. Life Sci Alliance 2:e201900429. https://doi.org/10.26508/lsa.201900429
Altschul SF, Gish W, Miller W et al (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Andreini C, Bertini I, Cavallaro G et al (2008) Metal ions in biological catalysis: from enzyme databases to general principles. J Biol Inorg Chem 13:1205–1218. https://doi.org/10.1007/s00775-008-0404-5
Ashburner M, Ball CA, Blake JA et al (2000) Gene Ontology: tool for the unification of biology. Nat Genet 25:25–29. https://doi.org/10.1038/75556
Bindea G, Mlecnik B, Hackl H et al (2009) ClueGO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 25:1091–1093. https://doi.org/10.1093/bioinformatics/btp101
Blum T, Briesemeister S, Kohlbacher O (2009) MultiLoc2: integrating phylogeny and Gene Ontology terms improves subcellular protein localization prediction. BMC Bioinform 10:274. https://doi.org/10.1186/1471-2105-10-274
Buchan DWA, Jones DT (2019) The PSIPRED protein analysis workbench: 20 years on. Nucleic Acids Res 47:W402–W407. https://doi.org/10.1093/nar/gkz297
Contestabile R, Paiardini A, Pascarella S et al (2001) <scp>l</scp>-Threonine aldolase, serine hydroxymethyltransferase and fungal alanine racemase. Eur J Biochem 268:6508–6525. https://doi.org/10.1046/j.0014-2956.2001.02606.x
Dangl JL, Jones JDG (2001) Plant pathogens and integrated defence responses to infection. Nature 411:826–833. https://doi.org/10.1038/35081161
Dixit H, Upadhyay V, Kulharia M, Verma SK (2023) The putative metal-binding proteome of the coronaviridae family. Metallomics. https://doi.org/10.1093/mtomcs/mfad001
Eide DJ (2020) Transcription factors and transporters in zinc homeostasis: lessons learned from fungi. Crit Rev Biochem Mol Biol 55:88–110. https://doi.org/10.1080/10409238.2020.1742092
Fasken MB, Corbett AH, Stewart M (2019) Structure-function relationships in the Nab2 polyadenosine-RNA binding Zn finger protein family. Protein Sci 28:513–523. https://doi.org/10.1002/pro.3565
Finn RD, Bateman A, Clements J et al (2014) Pfam: the protein families database. Nucleic Acids Res 42:D222–D230. https://doi.org/10.1093/nar/gkt1223
Gerwien F, Skrahina V, Kasper L et al (2018) Metals in fungal virulence. FEMS Microbiol Rev. https://doi.org/10.1093/femsre/fux050
Guo X, Zhong D, Xie W et al (2019) Functional identification of novel cell death-inducing effector proteins from Magnaporthe oryzae. Rice 12:59. https://doi.org/10.1186/s12284-019-0312-z
Haft DH (2003) The TIGRFAMs database of protein families. Nucleic Acids Res 31:371–373. https://doi.org/10.1093/nar/gkg128
Hall M, Frank E, Holmes G et al (2009) The WEKA data mining software. ACM SIGKDD Explor Newsl 11:10–18. https://doi.org/10.1145/1656274.1656278
Hoglund A, Donnes P, Blum T et al (2006) MultiLoc: prediction of protein subcellular localization using N-terminal targeting sequences, sequence motifs and amino acid composition. Bioinformatics 22:1158–1165. https://doi.org/10.1093/bioinformatics/btl002
Horton P, Park K-J, Obayashi T et al (2007) WoLF PSORT: protein localization predictor. Nucleic Acids Res 35:W585–W587. https://doi.org/10.1093/nar/gkm259
Hu S-P, Li J-J, Dhar N et al (2021) Lysin Motif (LysM) proteins: interlinking manipulation of plant immunity and fungi. Int J Mol Sci 22:3114. https://doi.org/10.3390/ijms22063114
Jashni MK, Dols IHM, Iida Y et al (2015) Synergistic action of a metalloprotease and a serine protease from Fusarium oxysporum f. sp. lycopersici cleaves chitin-binding tomato chitinases, reduces their antifungal activity, and enhances fungal virulence. Mol Plant-Microbe Interact 28:996–1008. https://doi.org/10.1094/MPMI-04-15-0074-R
Jaswal R, Kiran K, Rajarammohan S et al (2020) Effector biology of biotrophic plant fungal pathogens: current advances and future prospects. Microbiol Res 241:126567. https://doi.org/10.1016/j.micres.2020.126567
Jiang RHY, Tripathy S, Govers F, Tyler BM (2008) RXLR effector reservoir in two Phytophthora species is dominated by a single rapidly evolving superfamily with more than 700 members. Proc Natl Acad Sci 105:4874–4879. https://doi.org/10.1073/pnas.0709303105
Kall L, Krogh A, Sonnhammer ELL (2007) Advantages of combined transmembrane topology and signal peptide prediction–the Phobius web server. Nucleic Acids Res 35:W429–W432. https://doi.org/10.1093/nar/gkm256
Kelley LA, Mezulis S, Yates CM et al (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10:845–858. https://doi.org/10.1038/nprot.2015.053
Kochańczyk T, Drozd A, Krężel A (2015) Relationship between the architecture of zinc coordination and zinc binding affinity in proteins—insights into zinc regulation. Metallomics 7:244–257. https://doi.org/10.1039/C4MT00094C
Kozlov G, Bastos-Aristizabal S, Määttänen P et al (2010) Structural basis of cyclophilin B binding by the calnexin/calreticulin P-domain. J Biol Chem 285:35551–35557. https://doi.org/10.1074/jbc.M110.160101
Krogh A, Larsson B, von Heijne G, Sonnhammer ELL (2001) Predicting transmembrane protein topology with a hidden markov model: application to complete genomes11Edited by F. Cohen J Mol Biol 305:567–580. https://doi.org/10.1006/jmbi.2000.4315
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. J Chem Inf Model 51:2778–2786. https://doi.org/10.1021/ci200227u
Letunic I, Bork P (2018) 20 years of the SMART protein domain annotation resource. Nucleic Acids Res 46:D493–D496. https://doi.org/10.1093/nar/gkx922
Letunic I, Bork P (2021) Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res 49:W293–W296. https://doi.org/10.1093/nar/gkab301
Li Y, Han Y, Qu M et al (2020) Apoplastic cell death-inducing proteins of filamentous plant pathogens: roles in plant-pathogen interactions. Front Genet. https://doi.org/10.3389/fgene.2020.00661
Limacher A, Kloer DP, Flückiger S et al (2006) The crystal structure of Aspergillus fumigatus cyclophilin reveals 3D domain swapping of a central element. Structure 14:185–195. https://doi.org/10.1016/j.str.2005.10.015
Lin Y-F, Cheng C-W, Shih C-S et al (2016) MIB: metal ion-binding site prediction and docking server. J Chem Inf Model 56:2287–2291. https://doi.org/10.1021/acs.jcim.6b00407
Liu T-B, Xue C (2011) The Ubiquitin-proteasome system and f-box proteins in pathogenic fungi. Mycobiology 39:243–248. https://doi.org/10.5941/MYCO.2011.39.4.243
López-Berges MS (2020) ZafA-mediated regulation of zinc homeostasis is required for virulence in the plant pathogen Fusarium oxysporum. Mol Plant Pathol 21:244–249. https://doi.org/10.1111/mpp.12891
Lorenzo-Gutiérrez D, Gómez-Gil L, Guarro J et al (2019) Role of the Fusarium oxysporum metallothionein Mt1 in resistance to metal toxicity and virulence. Metallomics 11:1230–1240. https://doi.org/10.1039/c9mt00081j
Lothian A, Hare DJ, Grimm R et al (2013) Metalloproteomics: principles, challenges and applications to neurodegeneration. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2013.00035
Lubec G, Afjehi-Sadat L (2007) Limitations and pitfalls in protein identification by mass spectrometry. Chem Rev 107:3568–3584. https://doi.org/10.1021/cr068213f
Marchler-Bauer A, Derbyshire MK, Gonzales NR et al (2015) CDD: NCBI’s conserved domain database. Nucleic Acids Res 43:D222–D226. https://doi.org/10.1093/nar/gku1221
Maret W (2016) The metals in the biological periodic system of the elements: concepts and conjectures. Int J Mol Sci 17:66. https://doi.org/10.3390/ijms17010066
McCall KA, Huang C, Fierke CA (2000) Function and mechanism of zinc metalloenzymes. J Nutr 130:1437S-1446S. https://doi.org/10.1093/jn/130.5.1437S
Morgan W, Kamoun S (2007) RXLR effectors of plant pathogenic oomycetes. Curr Opin Microbiol 10:332–338. https://doi.org/10.1016/j.mib.2007.04.005
Ökmen B, Kemmerich B, Hilbig D et al (2018) Dual function of a secreted fungalysin metalloprotease in Ustilago maydis. New Phytol 220:249–261. https://doi.org/10.1111/nph.15265
Pennington HG, Jones R, Kwon S et al (2019) The fungal ribonuclease-like effector protein CSEP0064/BEC1054 represses plant immunity and interferes with degradation of host ribosomal RNA. PLoS Pathog 15:e1007620. https://doi.org/10.1371/journal.ppat.1007620
Putignano V, Rosato A, Banci L, Andreini C (2018) MetalPDB in 2018: a database of metal sites in biological macromolecular structures. Nucleic Acids Res 46:D459–D464. https://doi.org/10.1093/nar/gkx989
Quevillon E, Silventoinen V, Pillai S et al (2005) InterProScan: protein domains identifier. Nucleic Acids Res 33:W116–W120. https://doi.org/10.1093/nar/gki442
Rafiei V, Vélëz H, Tzelepis G (2021) The role of glycoside hydrolases in phytopathogenic fungi and oomycetes virulence. Int J Mol Sci 22:9359. https://doi.org/10.3390/ijms22179359
Renna M, Durante M, Gonnella M et al (2018) Quality and nutritional evaluation of Regina Tomato, a Traditional Long-Storage Landrace of Puglia (Southern Italy). Agriculture 8:83. https://doi.org/10.3390/agriculture8060083
Rocafort M, Fudal I, Mesarich CH (2020) Apoplastic effector proteins of plant-associated fungi and oomycetes. Curr Opin Plant Biol 56:9–19. https://doi.org/10.1016/j.pbi.2020.02.004
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Sharma A, Sharma D, Verma SK (2018) In silico study of iron zinc and copper binding proteins of Pseudomonas syringae pv. lapsa: emphasis on secreted metalloproteins. Front Microbiol. https://doi.org/10.3389/fmicb.2018.01838
Shi W, Punta M, Bohon J et al (2011) Characterization of metalloproteins by high-throughput X-ray absorption spectroscopy. Genome Res 21:898–907. https://doi.org/10.1101/gr.115097.110
Singh VK, Singh AK, Kumar A (2017) Disease management of tomato through PGPB: current trends and future perspective. 3 Biotech 7:255. https://doi.org/10.1007/s13205-017-0896-1
Sonah H, Deshmukh RK, Bélanger RR (2016) Computational prediction of effector proteins in fungi: opportunities and challenges. Front Plant Sci. https://doi.org/10.3389/fpls.2016.00126
Sperschneider J, Dodds PN (2022) EffectorP 3.0: prediction of apoplastic and cytoplasmic effectors in fungi and oomycetes. Mol Plant-Microbe Interact 35:146–156. https://doi.org/10.1094/MPMI-08-21-0201-R
Sperschneider J, Dodds PN, Singh KB, Taylor JM (2018) <scp>ApoplastP</scp>: prediction of effectors and plant proteins in the apoplast using machine learning. New Phytol 217:1764–1778. https://doi.org/10.1111/nph.14946
Srinivas C, Nirmala Devi D, Narasimha Murthy K et al (2019) Fusarium oxysporum f. sp. lycopersici causal agent of vascular wilt disease of tomato: Biology to diversity—a review. Saudi J Biol Sci 26:1315–1324. https://doi.org/10.1016/j.sjbs.2019.06.002
Stanton C, Sanders D, Krämer U, Podar D (2022) Zinc in plants: Integrating homeostasis and biofortification. Mol Plant 15:65–85. https://doi.org/10.1016/j.molp.2021.12.008
Tamura K, Stecher G, Peterson D et al (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Tatusov RL (2001) The COG database: new developments in phylogenetic classification of proteins from complete genomes. Nucleic Acids Res 29:22–28. https://doi.org/10.1093/nar/29.1.22
Teufel F, Almagro Armenteros JJ, Johansen AR et al (2022) SignalP 6.0 predicts all five types of signal peptides using protein language models. Nat Biotechnol. https://doi.org/10.1038/s41587-021-01156-3
Tusnady GE, Simon I (2001) The HMMTOP transmembrane topology prediction server. Bioinformatics 17:849–850. https://doi.org/10.1093/bioinformatics/17.9.849
Urban M, Cuzick A, Seager J et al (2022) PHI-base in 2022: a multi-species phenotype database for Pathogen-Host Interactions. Nucleic Acids Res 50:D837–D847. https://doi.org/10.1093/nar/gkab1037
Villa-Rivera MG, Cano-Camacho H, López-Romero E, Zavala-Páramo MG (2021) The role of arabinogalactan type II degradation in plant-microbe interactions. Front Microbiol. https://doi.org/10.3389/fmicb.2021.730543
Yakovlev GI, Karpeisky MYa, Bezborodova SI et al (1980) Guanyl-specific ribonuclease from the fungus Penicillium chrysogenum strain 152 and its complex with guanosine 3’-phosphate studied by nuclear magnetic resonance. Eur J Biochem 109:75–85. https://doi.org/10.1111/j.1432-1033.1980.tb04769.x
Yamashita MM, Wesson L, Eisenman G, Eisenberg D (1990) Where metal ions bind in proteins. Proc Natl Acad Sci 87:5648–5652. https://doi.org/10.1073/pnas.87.15.5648
Yang B, Wang Y, Tian M et al (2021) Fg12 ribonuclease secretion contributes to Fusarium graminearum virulence and induces plant cell death. J Integr Plant Biol 63:365–377. https://doi.org/10.1111/jipb.12997
Ye W, Wang Y, Wang Y (2015) Bioinformatics analysis reveals abundant short alpha-helices as a common structural feature of oomycete RxLR effector proteins. PLoS ONE 10:e0135240. https://doi.org/10.1371/journal.pone.0135240
Yu C-S, Chen Y-C, Lu C-H, Hwang J-K (2006) Prediction of protein subcellular localization. Proteins Struct Funct Bioinform 64:643–651. https://doi.org/10.1002/prot.21018
Yu Q-K, Han L-T, Wu Y-J, Liu T-B (2020) The role of oxidoreductase-like protein Olp1 in sexual reproduction and virulence of Cryptococcus neoformans. Microorganisms 8:1730. https://doi.org/10.3390/microorganisms8111730
Zhang Z-N, Wu Q-Y, Zhang G-Z et al (2015) Systematic analyses reveal uniqueness and origin of the CFEM domain in fungi. Sci Rep 5:13032. https://doi.org/10.1038/srep13032
Acknowledgements
The authors are thankful to the Indian Institute of Technology Roorkee, Uttarakhand and the Central University of Himachal Pradesh for its computational facility.
Funding
ASK is grateful to the University Grants Commission of India for providing the research fellowship (Ref. No. 19/06/2016(I) EU-V). SKV is thankful to the Institution of Eminence, University of Delhi for the Faculty Research Programme Grant (Ref. No./IoE/2021/12/FRP).
Author information
Authors and Affiliations
Contributions
ASK performed all experiments, collected and analysed data, and wrote the manuscript. HD and VU extracted the zinc-binding proteins and its clustering. SKV and ASK conceived the idea of work. SKV edited the manuscript. RP provided guidance and supervision throughout the whole work process.
Corresponding author
Ethics declarations
Conflict of interest
The authors affirm that they have no known financial or personal affiliations that could have seemed to affect the work disclosed in this study.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kushwah, A.S., Dixit, H., Upadhyay, V. et al. Elucidating the zinc-binding proteome of Fusarium oxysporum f. sp. lycopersici with particular emphasis on zinc-binding effector proteins. Arch Microbiol 205, 298 (2023). https://doi.org/10.1007/s00203-023-03638-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03638-1