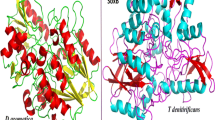
Abstract
Diverse thermophilic microorganisms with the potential to withstand extreme physiological conditions have long been investigated and explored for human commercial benefit. Thermozymes with distinct functional and structural properties isolated from these thermophiles are known to have high thermostability without significant loss of specific enzyme activity. Thermophiles isolated and characterised from the thermophilic ecological niche of India are well documented. There is a plethora of work in the literature emphasising its industrial significance. However, in-depth knowledge of the thermophilic oxidoreductase group of enzymes (Oxizymes) is restricted. Sulfur Oxygenase Reductases or Sulfur Oxygen-Reductases (SORs) are a group of thermophilic oxizymes reported predominantly from thermophilic and mesophilic archaea and bacteria, which catalyse oxygen-dependent disproportionation reactions of elemental sulfur, producing sulfite, thiosulfate, and sulphide. There have been few reports on isolated and characterised SORs from the Indian geothermal niche. The review article will highlight the SORs reported till date with a concise overview of different archaeal and bacterial species producing the enzymes. Based on the literature available till date, characteristics including physico-chemical properties, amino acid sequence homology, conserved motifs and their 3D structure comparison have been discussed. In-silico sequence and structure level preliminary comparative analysis of various SORs has also been discussed. However, a few SORs whose structural information is not reported in the protein data bank have been modelled to enrich our analysis.









Similar content being viewed by others
References
Anderson I, Chertkov O, Chen A, Saunders E, Lapidus A, Nolan M, Lucas S, Hammon N, Deshpande S, Cheng JF, Han C, Tapia R, Goodwin LA, Pitluck S, Liolios K, Pagani I, Ivanova N, Mikhailova N, Pati A, Palaniappan K, Land M, Pan C, Rohde M, Pukall R, Göker M, Detter JC, Woyke T, Bristow J, Eisen JA, Markowitz V, Hugenholtz P, Kyrpides NC, Klenk HP, Mavromatis K (2012) Complete genome sequence of the moderately thermophilic mineral-sulfide-oxidizing firmicute sulfobacillus acidophilus type strain (NAL T). Standards Genom Sci 6(3):293–303. https://doi.org/10.4056/sigs.2736042
Chen ZW, Liu YY, Wu JF, She Q, Jiang CY, Liu SJ (2007) Novel bacterial sulfur oxygenase reductases from bioreactors treating gold-bearing concentrates. Appl Microbiol Biotechnol 74(3):688–698. https://doi.org/10.1007/s00253-006-0691-0
Claire V, Zeikus GJ (2001) Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65(1):1–43
Friedrich CG (1997) Physiology and genetics of sulfur-oxidizing bacteria. Adv Microb Physiol 39:235–289. https://doi.org/10.1016/S0065-2911(08)60018-1
Gygli G, van Berkel W (2015) Oxizymes for biotechnology. Curr Biotechnol 4(2):100–110.https://doi.org/10.2174/2211550104666150423202036
Haruyuki A, Sato T, Kanai T (2011) Application of hyperthermophiles and their enzymes. Curr Opin Biotechnol 22(5):618–626. https://doi.org/10.1016/j.copbio.2011.06.010
he, zhengguo, yaqin li, peijin zhou, and shuang jiang liu, (2000) cloning and heterologous expression of a sulfur oxygenase/reductase gene from the thermoacidophilic archaeon acidianus sp. S5 in Escherichia Coli. FEMS Microbiol Lett 193(2):217–221. https://doi.org/10.1016/S0378-1097(00)00485-7
Husain Q (2017) High yield immobilization and stabilization of oxidoreductases using magnetic nanosupports and their potential applications: an update. Curr Catal 6(3):168–187. https://doi.org/10.2174/2211544706666170704141828
Janosch C, Thyssen C, Vera M, Bonnefoy V, Rohwerder T, Sand W (2009) Sulfur oxygenase reductase in different acidithiobacillus caldus-like strains. Adv Mater Res 71–73:239–242. https://doi.org/10.4028/www.scientific.net/AMR.71-73.239
Janosch C, Remonsellez F, Sand W, Vera M (2015) Sulfur oxygenase reductase (Sor) in the moderately thermoacidophilic leaching bacteria: studies in sulfobacillus thermosulfidooxidans and acidithiobacillus caldus. Microorganisms 3(4):707–724. https://doi.org/10.3390/microorganisms3040707
Khaleque HN, Corbett MK, Ramsay JP, Kaksonen AH, Boxall NJ, Watkin ELJ (2017) Complete genome sequence of acidihalobacter prosperus strain f5, an extremely acidophilic, iron- and sulfur-oxidizing halophile with potential industrial applicability in saline water bioleaching of chalcopyrite. J Biotechnol 262:56–59. https://doi.org/10.1016/j.jbiotec.2017.10.001
Kletzin A (1992) Molecular characterization of the sor gene, which encodes the sulfur oxygenase/reductase of the thermoacidophilic archaeum desulfurolobus ambivalens. J Bacteriol 174(18):5854–5859. https://doi.org/10.1128/jb.174.18.5854-5859.1992
Kletzin, A (1989) Coupled enzymatic production of sulfite, thiosulfate, and hydrogen sulfide from sulfur: purification and properties of a sulfur oxygenase reductase from the facultatively anaerobic archaebacterium desulfurolobus ambivalens. J Bacteriol 171(3):1638–43. https://doi.org/10.1128/jb.171.3.1638-1643.1989
Lamed RJ, Zeikus JG (1981) Novel NADP-linked alcohol–aldehyde/ketone oxidoreductase in thermophilic ethanologenic bacteria. Biochem J 195(1):183–190. https://doi.org/10.1042/bj1950183
Larkin MA, Blackshields G, Brown NP, Chenna R, Mcgettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X Version 2.0. Bioinformatics 23(21):2947–2948. https://doi.org/10.1093/bioinformatics/btm404
Lee LS, Goh KM, Chan CS, Tan GYA, Yin WF, Chong CS, Chan KG (2018) Microbial diversity of thermophiles with biomass deconstruction potential in a foliage-rich hot spring. MicrobiologyOpen 7(6):1–13. https://doi.org/10.1002/mbo3.615
Li M, Chen Z, Zhang P, Pan X, Jiang C, An X, Liu S, Chang W (2008) Crystal structure studies on sulfur oxygenase reductase from acidianus tengchongensis. Biochem Biophys Res Commun 369(3):919–923. https://doi.org/10.1016/j.bbrc.2008.02.131
Liu S-J (2008) Archaeal and bacterial sulfur oxygenase-reductases: genetic diversity and physiological function. Microb Sulfur Metab. https://doi.org/10.1007/978-3-540-72682-1_17
Panda SK, Jyoti V, Bhadra B, Nayak KC, Shivaji S, Rainey FA, Das SK (2009) Thiomonas bhubaneswarensis sp. nov., an obligately mixotrophic, moderately thermophilic, thiosulfate-oxidizing bacterium. Int J Syst Evolut Microbiol 59(9):2171–2175. https://doi.org/10.1099/ijs.0.007120-0
Pelletier N, Leroy G, Guiral M, Giudici-Orticoni MT, Aubert C (2008) First characterisation of the active oligomer form of sulfur oxygenase reductase from the bacterium aquifex aeolicus. Extremophiles 12(2):205–215. https://doi.org/10.1007/s00792-007-0119-5
Rühl P, Kletzin A (2017a) The sulfur oxygenase reductase activity assay: catalyzing a reaction with elemental sulfur as substrate at high temperatures. Bio-Protoc 7(14):1–10. https://doi.org/10.21769/bioprotoc.2403
Rühl P, Kletzin A (2017b) The sulfur oxygenase reductase activity assay: catalyzing a reaction with elemental sulfur as substrate at high temperatures. Bio-Protoc. https://doi.org/10.21769/bioprotoc.2403
Rui W, Lin JQ, Liu XM, Pang X, Zhang CJ, Yang CL, Gao XY, Lin CM, Li YQ, Li Y, Lin JQ, Chen LX (2019) Sulfur oxidation in the acidophilic autotrophic acidithiobacillus Spp. Front Microbiol. https://doi.org/10.3389/fmicb.2018.03290
Sadiq FA, Li Y, Liu TJ, Flint S, Zhang G, Yuan L, Pei Z, He GQ (2016) The heat resistance and spoilage potential of aerobic mesophilic and thermophilic spore forming bacteria isolated from chinese milk powders. Int J Food Microbiol 238:193–201. https://doi.org/10.1016/j.ijfoodmicro.2016.09.009
Sato Y, Yabuki T, Adachi N, Moriya T, Arakawa T, Kawasaki M, Wakagi T (2020) Crystallographic and cryogenic electron microscopic structures and enzymatic characterization of sulfur oxygenase reductase from Sulfurisphaera tokodaii. J Struct Biol 4:100030. https://doi.org/10.1016/j.yjsbx.2020.100030
Sun CW, Chen ZW, He ZG, Zhou PJ, Liu SJ (2003) Purification and properties of the sulfur oxygenase/reductase from the acidothermophilic archaeon, acidianus strain S5. Extremophiles 7(2):131–134. https://doi.org/10.1007/s00792-002-0304-5
Tian T, He H, Liu XQ (2014) The SufBCD protein complex is the scaffold for iron-sulfur cluster assembly in thermus thermophiles HB8. Biochem Biophys Res Commun 443(2):376–381. https://doi.org/10.1016/j.bbrc.2013.11.131
Tikhonova TV, Slutsky A, Antipov AN, Boyko KM, Polyakov KM, Sorokin DY, Zvyagilskaya RA, Popov VO (2006) Molecular and catalytic properties of a novel cytochrome c nitrite reductase from nitrate-reducing haloalkaliphilic sulfur-oxidizing bacterium thioalkalivibrio nitratireducens. Biochim Biophys Acta 1764(4):715–723. https://doi.org/10.1016/j.bbapap.2005.12.021
Urich T, Bandeiras TM, Leal SS, Rachel R, Albrecht T, Zimmermann P, Scholz C, Teixeira M, Gomes CM, Kletzin A (2004) The sulfur oxygenase reductase from acidianus ambivalens is a multimeric protein containing a low-potential mononuclear non-haem iron centre. Biochem J 381(1):137–146. https://doi.org/10.1042/BJ20040003
Urich T, Kroke A, Bauer C, Seyfarth K, Reuff M, Kletzin A (2005) Identification of core active site residues of the sulfur oxygenase reductase from acidianus ambivalens by site-directed mutagenesis. FEMS Microbiol Lett 248(2):171–176. https://doi.org/10.1016/j.femsle.2005.05.031
Urich T, Gomes CM, Kletzin A, Frazão C (2006) X-ray structure of a self-compartmentalizing sulfur cycle metalloenzyme. Science 311(5763):996–1000. https://doi.org/10.1126/science.1120306
Veith A, Urich T, Seyfarth K, Protze J, Frazão C, Kletzin A (2011) Substrate pathways and mechanisms of inhibition in the sulfur oxygenase reductase of acidianus ambivalens. Front Microbiol. https://doi.org/10.3389/fmicb.2011.00037
Wu ZM, Zheng RC, Tang XL, Zheng YG (2017) Identification and characterization of a thermostable and cobalt-dependent amidase from burkholderia phytofirmans ZJB-15079 for efficient synthesis of (R)-3,3,3-trifluoro-2-hydroxy-2-methylpropionic acid. Appl Microbiol Biotechnol 101(5):1953–1964. https://doi.org/10.1007/s00253-016-7921-x
Funding
This mini-review received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
"Authors (1) and (4) contributed the majority part for the publication of this manuscript by doing the literature review, conceptualizing the idea, the data analysis and writing the manuscript. Author (2) and (3) performed the in-silico data analysis and its interpretations. All authors reviewed the manuscript."
Corresponding author
Ethics declarations
Conflict of interest
The authors certify that there is no actual or potential conflict of interest in relation to this review article.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pal, N., Sinha, S., Shivani et al. A review on bacterial and archaeal thermostable sulfur oxidoreductases (SORS)-an insight into the biochemical, molecular and in-silico structural comparative analysis of a neglected thermostable enzyme of industrial significance. Arch Microbiol 204, 655 (2022). https://doi.org/10.1007/s00203-022-03256-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03256-3