Abstract
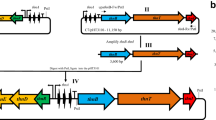
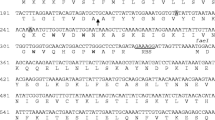
Thurincin H is a bacteriocin produced by Bacillus thuringiensis, it is encoded in a group of ten genes, most of which have been characterized experimentally or by homology. However, the activity of the thnI gene encoding a 95 amino acid ORF remains unknown. In this work, the thnI gene was cloned under the regulation of two promoters and transformed into a sensitive strain to determine if it acts as an immunity protein. In addition, a deletion mutant without the thnI gene was used to test whether thnI is required or not for the biosynthesis of thurincin H. It was concluded that thnI does not provide immunity and is not required to produce thurincin H.


Similar content being viewed by others
References
Casados-Vázquez LE, Bideshi DK, Barboza-Corona JE (2018) Regulator ThnR and the ThnDE ABC transporter proteins confer autoimmunity to thurincin H in Bacillus thuringiensis. Antonie Van Leeuwenhoek 111(12):2349–2360
Lee H, Churey JJ, Worobo RW (2009) Biosynthesis and transcriptional analysis of thurincin H, a tandem repeated bacteriocin genetic locus, produced by Bacillus thuringiensis SF361. FEMS Microbiol Lett 299(2):205–213
Mathur H, O’Connor PM, Cotter PD, Hill C, Ross RP (2014) Heterologous expression of thuricin CD immunity genes in Listeria monocytogenes. Antimicrob Agents Chemother 58(6):3421–3428
Mathur H, Rea MC, Cotter PD, Hill C, Ross RP (2015) The sactibiotic subclass of bacteriocins: an update. Curr Protein Pept Sci 16(6):549–558
Oros-Flores ZS, Casados-Vázquez LE, Bideshi DK, Salcedo-Hernández R, Barboza-Corona JE (2018) Co-synthesis of kenyacin 404 and heterologous thurincin H enhances the antibacterial activity of Bacillus thuringiensis. Biotech Lett 40(11):1531–1540
Sit CS, van Belkum MJ, McKay RT, Worobo RW, Vederas JC (2011) The 3D solution structure of thurincin H, a bacteriocin with four sulfur to α-carbon crosslinks. Angew Chem 123(37):8877–8880
Stein T, Heinzmann S, Solovieva I, Entian KD (2003) Function of Lactococcus lactis nisin immunity genes nisI and nisFEG after coordinated expression in the surrogate host Bacillus subtilis. J Biol Chem 278(1):89–94
Wieckowski BM, Hegemann JD, Mielcarek A, Boss L, Burghaus O, Marahiel MA (2015) The PqqD homologous domain of the radical SAM enzyme ThnB is required for thioether bond formation during thurincin H maturation. FEBS Lett 589(15):1802–1806
Zheng G, Hehn R, Zuber P (2000) Mutational analysis of the sbo-alb locus of Bacillus subtilis: identification of genes required for subtilosin A production and immunity. J Bacteriol 182(11):3266–3273
Funding
Luz E. Casados-Vázquez is a Young Associate Research supported by “Consejo Nacional de Ciencia y Tecnología” (CONACYT), México (Grant 269). This research was supported by Grant “Problemas Nacionales 2016” CONACyT (3664) and M. A. Núñez-Valle is a Bioscience graduate student supported by CONACyT. We thank to the Laboratorio Nacional PlanTECC, México, for the financial support granted in the project ‘Mantenimiento de la infraestructura del Laboratorio Nacional PlanTECC’.
Author information
Authors and Affiliations
Contributions
Núñez-Valle and Casados-Vázquez carried out the experiment. Casados-Vázquez and Massange-Sánchez wrote the manuscript with support from Barboza-Corona. Casados-Vázquez, Massange-Sánchez and Barboza-Corona supervised the project. Casados-Vázquez conceived the original idea.
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Massange-Sánchez, J.A., Núñez-Valle, M.A., Barboza-Corona, J.E. et al. The thnI gene is not required for thurincin H biosynthesis or immunity. Arch Microbiol 204, 344 (2022). https://doi.org/10.1007/s00203-022-02938-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-02938-2