Abstract
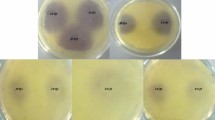
Acinetobacter species remain alive in hospitals on various surfaces, both dry and moist, forming an important source of hospital infections. These bacteria are naturally resistant to many antibiotic classes. Although the role of the quorum sensing system in regulating the virulence factors of Acinetobacter species has not been fully elucidated, it has been reported that they play a role in bacterial biofilm formation. The biofilm formation helps them to survive under unfavorable growth conditions and antimicrobial treatments. It is based on the accumulation of bacterial communication signal molecules in the area. In this study, we compared the bacterial signal molecules of 50 nosocomial Acinetobacter baumannii strain and 20 A. baumannii strain isolated from soil. The signal molecules were detected by the biosensor bacteria (Chromobacterium violaceum 026, Agrobacterium tumefaciens A136, and Agrobacterium tumefaciens NTL1) and their separation was determined by thin-layer chromatography. As a result, it has been found that soil-borne isolates can produce 3-oxo-C8-AHL and C8-AHL, whereas nosocomial-derived isolates can produce long-chain signals such as C10-AHL, C12-AHL, C14-AHL and C16-AHL. According to these results, it is possible to understand that these signal molecules are found in the infection caused by A. baumannii. The inhibition of this signaling molecules in a communication could use to prevent multiple antibiotic resistance of these bacteria.


Similar content being viewed by others
References
Baumann P (1968) Isolation of Acinetobacter from soil and water. J Bacteriol 96:39–42
Bhargava N, Sharma P, Capalash N (2015) Quorum sensing in Acinetobacter baumannii. Quorum sensing vs quorum quenching: a battle with no end in sight. Springer, India, pp 101–113
Boşgelmez-Tınaz G, Ulusoy S, Arıdoğan B, Eroğlu F, Kaya S (2005) N-butanoyl-l-homoserine lactone (BHL) deficient Pseudomonas aeruginosa isolates from an intensive care unit. Microbiol Res 160:399–403. doi:10.1016/j.micres.2005.03.005
Bozkurt F, Kaya S, Tekin R, Gulsun S, Deveci O, Dayan S, Hosoglu S (2014) Analysis of antimicrobial consumption and cost in a teaching hospital. J Infect Public Health 7:161–169. doi:10.1016/j.jiph.2013.09.007
Castillo-Juarez I et al. (2017) Exploiting quorum sensing inhibition for the control of Pseudomonas aeruginosa and Acinetobacter baumannii biofilms. Curr Top Med Chem 7. PMID:28056745
Chan K-GG et al (2011) Characterization of N-acylhomoserine lactone-degrading bacteria associated with the Zingiber officinale (ginger) rhizosphere: co-existence of quorum quenching and quorum sensing in Acinetobacter and Burkholderia. BMC Microbiol 11:51. doi:10.1186/1471-2180-11-51
Chan K-G, Cheng HJ, Chen JW, Yin W-F, Ngeow YF (2014) Tandem mass spectrometry detection of quorum sensing activity in multidrug resistant clinical isolate Acinetobacter baumannii. Sci World J. doi:10.1155/2014/891041
Chen H, Fujita M, Feng Q, Clardy J, Fink GR (2004) Tyrosol is a quorum-sensing molecule in Candida albicans. PNAS 101:5048–5052. doi:10.1073/pnas.0401416101
Chow JY, Yang Y, Tay SB, Chua KL, Yew WS (2014) Disruption of biofilm formation by the human pathogen Acinetobacter baumannii using engineered quorum-quenching lactonases. Antimicrob Agents Chemother 58:1802–1805. doi:10.1128/AAC.02410-13
de Kievit TR, Iglewski BH (2000) Bacterial quorum sensing in pathogenic relationships. Infect Immun 68:4839–4849. doi:10.1128/iai.68.9.4839-4849.2000
Ertek M (2008) Hastane Enfeksiyonları: Türkiye Verileri. İÜ Cerrahpaşa Tıp Fakültesi Sürekli Tıp Eğitimi Etkinlikleri 60:9–14
Fuqua WC, Winans SC, Greenberg EP (1994) Quorum sensing in bacteria: the LuxR–LuxI family of cell density-responsive transcriptional regulators. J Bacteriol 176:269–275 (PMC205046)
González RH, Nusblat A, Nudel BC (2001) Detection and characterization of quorum sensing signal molecules in Acinetobacter strains. Microbiol Res 155:271–277. doi:10.1016/S0944-5013(01)80004-5
González RH, Dijkshoorn L, Van den Barselaar M, Nudel C (2009) Quorum sensing signal profile of Acinetobacter strains from nosocomial and environmental sources. Rev Argent Microbiol 41:73–78 PMID:19623895
Kalia VC (2013) Quorum sensing inhibitors: an overview. Biotechnol Adv 31:224–245. doi:10.1016/j.biotechadv.2012.10.004
Kostoulias X et al (2016) Impact of a cross-kingdom signaling molecule of Candida albicans on Acinetobacter baumannii physiology. Antimicrob Agents Chemother 60:161–167. doi:10.1128/AAC.01540-15
Koul S, Prakash J, Mishra A, Kalia VC (2016) Potential emergence of multi-quorum sensing inhibitor resistant (MQSIR) bacteria. Indian J Microbiol 56:1–18. doi:10.1007/s12088-015-0558-0
McClean KH et al (1997) Quorum sensing and Chromobacterium violaceum: exploitation of violacein production and inhibition for the detection of N-acylhomoserine lactones. Microbiology (Reading, England) 143(Pt 12):3703–3711
McLean RJ, Pierson LS, Fuqua C (2004) A simple screening protocol for the identification of quorum signal antagonists. J Microbiol Methods 58:351–360. doi:10.1016/j.mimet.2004.04.016
Miller J (1972) Experiments in molecular genetics. Cold Springs Harbor University Press, Cold Spring Harbor
Modarresi F, Azizi O, Shakibaie MR, Motamedifar M, Mansouri S (2016) Cloning and expression of quorum sensing N-acyl-homoserine synthase (LuxI) gene detected in Acinetobacter baumannii. Iran J Microbiol 8:139 (PMC4906721)
Morohoshi T, Kato M, Fukamachi K, Kato N, Ikeda T (2008) N-acylhomoserine lactone regulates violacein production in Chromobacterium violaceum type strain ATCC 12472. FEMS Microbiol Lett 279:124–130. doi:10.1111/j.1574-6968.2007.01016.x
Pina PGP, Grosbuis S, Guyot L, Ghnassia JC, Allouch PY (1998) An Acinetobacter baumanii outbreak at the Versailles Hospital Center. Pathol Biol 46:385–394 PMID:9769866
Priya K, Yin W-FF, Chan K-GG (2013) Anti-quorum sensing activity of the traditional Chinese herb, Phyllanthus amarus. Sensors (Basel, Switzerland) 13:14558–14569. doi:10.3390/s131114558
Shaw PD, Ping G, Daly SL, Cha C, Cronan JE, Rinehart KL, Farrand SK (1997) Detecting and characterizing N-acyl-homoserine lactone signal molecules by thin-layer chromatography. PNAS 94:6036–6041. doi:10.1073/pnas.94.12.6036
Sio CF et al (2006) Quorum quenching by an N-acyl-homoserine lactone acylase from Pseudomonas aeruginosa PAO1. Infect Immun 74:1673–1682. doi:10.1128/IAI.74.3.1673-1682.2006
Stacy DM, Welsh MA, Rather PN, Blackwell HE (2012) Attenuation of quorum sensing in the pathogen Acinetobacter baumannii using non-native N-Acyl homoserine lactones. ACS Chem Biol 7:1719–1728. doi:10.1021/cb300351x
Tariq TM (2014) Bacteriologic profile and antibiogram of blood culture isolates from a Children’s Hospital in Kabul. J Coll Physicians Surg Pak 24:3396–3399 PMID:24953929
Waheed H, Hashmi I, Khan SJ, Kim SR, Arshad M, Nasir H (2016) Microbial population dynamics and profiling of quorum sensing agents in membrane bioreactor. Int Biodeterior Biodegrad 113:66–73. doi:10.1016/j.ibiod.2015.12.014
Zahin M, Hasan S, Aqil F, Khan MS, Husain FM, Ahmad I (2010) Screening of certain medicinal plants from India for their anti-quorum sensing activity. Indian J Exp Biol 48:1219–1224 PMID:21250604
Acknowledgements
The data of this study have been excerpted from the Ph.D. thesis of the author. This work was financed by Hacettepe University Scientific Research Project grant BAP 013BDYP604002.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All of the authors declare they have no competing interest.
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Erdönmez, D., Rad, A.Y. & Aksöz, N. Quorum sensing molecules production by nosocomial and soil isolates Acinetobacter baumannii . Arch Microbiol 199, 1325–1334 (2017). https://doi.org/10.1007/s00203-017-1408-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-017-1408-8