Abstract
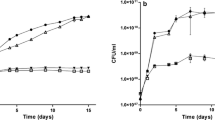
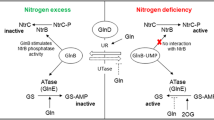
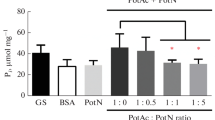
The nitrogen-fixing, root-associated strain Pseudomonas stutzeri A1501 carries a single gene encoding a protein from the PII family, designated glnK. The glnK gene is co-transcribed with two distantly related copies of amtB genes encoding putative ammonium channels. Transcription of glnK was decreased in the presence of ammonia and was partly dependent on NtrC and RpoN under nitrogen-limiting conditions. Inactivation of glnK led to a mutant strain devoid of nitrogenase activity, auxotrophic for glutamine and unable to deadenylylate glutamine synthetase, while inactivation of amtB 1 led to a prototrophic and Nif+ mutant strain. RT-PCR analysis showed that nifA transcription was abolished in the glnK mutant, while glnA remained transcribed. Using the yeast two-hybrid system, an interaction between GlnK and the C-terminal domain of NifL was observed, suggesting GlnK-dependent control of NifA activity by NifL. Introduction of a plasmid that expressed nifA from a constitutive promoter restored nitrogen fixation to the glnK mutant, and nitrogenase activity was observed even in the presence of ammonia. GlnK signalling appears to be a key regulatory element in control of ammonia assimilation, of nifA expression and in modulation of NifA activity by NifL.


Similar content being viewed by others
References
Alexander F, Yakunin P, Hallenbeck PC (2002) AmtB is necessary for NH4 +-induced nitrogenase switch-off and ADP-ribosylation in Rhodobacter capsulatus. J Bacteriol 184:4081–4088
Allen LN, Hanson RS (1985) Construction of broad-host-range cosmid cloning vectors: identification of genes necessary for growth of Methylobacterium organophilum on methanol. J Bacteriol 161:955–962
Arcondéguy T, Jack R, Merrick M (2001) The PII signal transduction proteins, pivotal players in microbial nitrogen control. Microbiol Mol Biol Rev 65:80–105
Arsène F, kaminski pa, Elmerich C (1996) Modulation of NifA activity by PII in Azospirillum brasilense: evidence for a regulatory role of the NifA N-terminal domain. J Bacteriol 178:4830–4838
Bender RA, Janssen KA, Resnick AD, Blumenberg M, Foor F, Magasanik B (1977) Biochemical parameters of glutamine synthetase from Klebsiella aerogenes. J Bacteriol 129:1001–1009
Blatny JM, Brautaset T, Winter-Larsen H, Haugan K, Valla S (1997) Construction and use of a versatile set of broad-host-range cloning and expression vectors based on RK2 replicon. Appl Environ Microbiol 63:370–379
Bonatto AC, Souza EM, Pedrosa FO, Yates MG, Benelli EM (2005) Effect of T- and C-loop mutations on the Herbaspirillum seropedicae GlnB protein in nitrogen signaling. Res Microbiol 156:634–640
Chen S, Liu L, Zhou X, Elmerich C, Li JL (2005) Functional analysis of the GAF domain of NifA in Azospirillum brasilense: effects of Tyr-Phe mutations on NifA and its interaction with GlnB. Mol Genet Genomics 273:415–422
Coutts G, Thomas G, Blakey D, Merrick M (2002) Membrane sequestration of the signal transduction protein GlnK by the ammonium transporter AmtB. EMBO J 21:536–545
de Zamaroczy M (1998) Structural homologues PII and Pz of Azospirillum brasilense provide intracellular signaling for selective regulation of various nitrogen-dependent functions. Mol Microbiol 29:449–463
Desnoues N, Lin M, Guo X, Ma L, Carreño-Lopez R , Elmerich C (2003) Nitrogen fixation genetics and regulation in a Pseudomonas stutzeri strain associated with rice. Microbiology 149:2251–2262
He L, Soupène E, Ninfa A, Kustu S (1998) Physiological role for the GlnK protein of enteric bacteria: relief of NifL inhibition under nitrogen-limiting conditions. J Bacteriol 180:6661–6667
Holtel A, Merrick M (1989) The Klebsiella pneumoniae PII protein (glnB gene product) is not absolutely required for nitrogen regulation and is not involved in NifL-mediated nif gene regulation. Mol Gen Genet 217:474–480
Jack R, De Zamaroczy M, Merrick M (1999) The signal transduction protein GlnK is required for NifL-dependent nitrogen control of nif gene expression in Klebsiella pneumoniae. J Bacteriol 181:1156–1162
James P, Halladay J, Craig EA (1996) Genomic libraries and a host strain designed for highly efficient two-hybrid selection in yeast. Genetics 144:1425–1436
Jiang P, Peliska JA, Ninfa AJ (1998) The regulation of Escherichia coli glutamine synthetase revisited: role of 2-ketoglutarate in the regulation of glutamine synthetase adenylylation state. Biochemistry 37:12802–12810
Klopperogge K, Grabbe R, Hoppert M, Schmitz RA (2002) Membrane association of Klebsiella pneumoniae NifL is affected by molecular oxygen and combined nitrogen. Arch Microbiol 173:223–234
Lalucat J, Bennasar A, Bosch R, García-Valdés E, Palleroni NJ (2006) Biology of Pseudomonas stutzeri. Microbiol Mol Biol Rev 70:510–547
Lin M, Smalla K, Heuer H, van Elsas JD (2000) Effect of an Alcaligenes faecalis inoculant strain on bacterial communities in flooded microcosms planted with rice seedlings. Appl Soil Ecol 15:211–225
Little R, Colombo V, Leech A, Dixon R (2002) Direct interaction of the NifL regulatory protein with the GlnK signal transducer enables the Azotobacter vinelandii NifL-NifA regulatory system to respond to conditions replete for nitrogen. J Biol Chem 277:15472–15481
Martin DE, Hurek T, Reinhold-Hurek B (2000) Occurrence of three PII-like signal transmitter proteins in the diazotrophic proteobacterium Azoarcus sp.BH72. Mol Microbiol 38:276–288
Martínez-Argudo I, Little R, Shearer N, Johnson P, Dixon R (2004) The NifL-NifA system: a multidomain transcriptional regulatory complex that integrates environmental signals. J Bacteriol 186:601–610
Meletzus D, Rudnick P, Doetsch N, Green A, Kennedy C (1998) Characterization of the glnK-amtB operon of Azotobacter vinelandii. J Bacteriol 180:3260–3264
Merrick M (2004) Regulation of nitrogen fixation in free-living diazotrophs. In: Klipp BMJPG W, Newton WE (eds) Genetics and regulation of nitrogen fixation in free-living bacteria. Kluwer, Netherlands, pp 197–233
Miller JB (1972) Assay for β-galactosidase. In: Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor, pp 352–355
Pedrosa FO, Elmerich C (2007) Regulation of nitrogen fixation and ammonium assimilation in associative and endophytic nitrogen fixing bacteria. In: Elmerich C, Newton WE (eds) Associative and endophytic nitrogen fixing bacteria and cyanobacterial associations. Kluwer, The Netherlands, pp 47–71
Perlova O, Ureta A, Nordlund S, Meletzus D (2003) Identification of three genes encoding PII-like proteins in Gluconacetobacter diazotrophicus: studies of their role(s) in the control of nitrogen fixation. J Bacteriol 185:5854–5861
Rudnick P, Kunz C, Gunatilaka MK, Hines ER, Kennedy C (2002) Role of GlnK in NifL-mediated regulation of NifA activity in Azotobacter vinelandii. J Bacteriol 184:812–820
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory, New York
Shäfer A, Tauch A, Jager W, Kalinowski J, Thierbach G, Puehler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmid pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Simon R, Priefer U, Pühler A (1983) A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in Gram-negative bacteria. Biotechnology 1:784–791
Tang DJ, He YQ, Feng JX, He BR, Jiang BL, Lu GT, Chen B, Tang JL (2005) Xanthomonas campestris pv. Campestris posseses a single glucogenic pathway that is required for virulence. J Bacteriol 187:6231–6237
Vermeiren H, Willems A, Schoofs G, de Mot R, Keijers V, Hai W, Vanderleyden J (1999) The rice inoculant strain A15 is a nitrogen-fixing Pseudomonas stutzeri strain. Syst Appl Microbiol 22:215–224
Vermeiren H, Keijers V, Vanderleyden J (2002) Isolation and sequence analysis of the glnKamtB1amtB2 gene cluster, encoding a PII homologue and two putative ammonium transporters, from Pseudomonas stutzeri A15. DNA Seq 13:67–74
Xie Z, Dou Y, Ping S, Chen M, Wang G, Elmerich C, Lin M (2006) Interaction between NifL and NifA in the nitrogen-fixing Pseudomonas stutzeri A1501. Microbiology 152:3535–3542
You CB, Song HX, Wang JP, Lin M, Hai WL (1991) Association of Alcaligenes faecalis with wetland rice. Plant Soil 137:81–85
Zhang Y, Pohlmann EL, Ludden PW, Roberts GP (2001) Functional characterization of three GlnB homologs in the photosynthetic bacterium Rhodospirillum rubrum: roles in sensing ammonium and energy status. J Bacteriol 183:6159–6168
Zhang Y, Pohlmann EL, Roberts GP (2004) Identification of critical residues in GlnB for its activation of NifA activity in the photosynthetic bacterium Rhodospirillum rubrum. Proc Natl Acad Sci 101:2782–2787
Acknowledgments
The authors wish to thank Mr. Dong-Jie Tang for suggesting to use the primer P18conF and Ms. Jerri Bram for reading the typescript. This work was supported by the Ministry of Science and Technology of China (National Basic Research Program 2001CB108904 and 2007CB707805, National High-Tech Program 2004AA21470 and 2006AA020101), and the National Natural Science Foundation of China (grant No. 30470047 and 30670050). Sheng He thanks College of Life Science and Technology of Guangxi University for support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jack Meeks.
S. He and M. Chen contributed equally to the work.
Rights and permissions
About this article
Cite this article
He, S., Chen, M., Xie, Z. et al. Involvement of GlnK, a PII protein, in control of nitrogen fixation and ammonia assimilation in Pseudomonas stutzeri A1501. Arch Microbiol 190, 1–10 (2008). https://doi.org/10.1007/s00203-008-0354-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-008-0354-x