Abstract
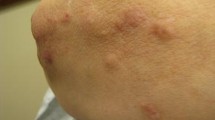
Zoledronic acid, an amino-bisphosphonate, is an antiresorptive drug given intravenously. It is commonly used for the treatment of severe postmenopausal osteoporosis, and it is also used for other types of osteoporosis, management of bone metastasis, multiple myeloma, hypercalcemia, and Paget’s disease. The use varies according to these diseases. The drug is usually well-tolerated with the most common side effects being acute-phase reactions with weakness and musculoskeletal pain as well as gastrointestinal effects. However, the increase in usage of zoledronic acid has produced rare reports of side effects, such as atrial fibrillation, atypical fracture, delayed fracture healing, osteonecrosis of the jaw, or buccal or genital mucosa. Less common cutaneous side effects associated with zoledronic acid include fever rash, maculopapular lesions, keratitis, dermatitis, B-cell pseudolymphoma, skin erythema multiform, and vasculitis. Although these cutaneous reactions are generally mild and self-limiting, others can be severe such as toxic epidermal necrolysis and pancytopenia. We hereby describe a case of diffuse skin adverse reactions, a type of confluent erythematous macules in the trunk and arms with extended petechial macules along the left thigh and leg, induced 2 days after the administration of zoledronic acid for primary osteoporosis in a 53-year-old woman, without any allergic context or other associated drugs taken.


Similar content being viewed by others
References
Cremers SC, Pillai G, Papapoulos SE (2005) Pharmacokinetics/pharmacodynamics of bisphosphonates: use for optimisation of intermittent therapy for osteoporosis. Clin Pharmacokinet 44(6):551–570
Briot K, Roux C, Thomas T, Blain H, Buchon D, Chapurlat R et al (2018) 2018 Update of French recommendations on the management of postmenopausal osteoporosis. Joint Bone Spine. https://doi.org/10.1016/j.jbspin.2018.02.009
Ruggiero SL, Mebrotra B, Rosenberg TJ et al (2004) Osteonecrosis of the jaws associated with the use of biphosphonates. A review of 63 cases. J Oral Maxillofac Facial Surg 62:527–534
Andreadis D, Mauroudis S, Poulopoulos A, Markopoulos A, Epivatianos A (2012) Lip ulceration associated with intravenous administration of zoledronic acid: report of a case. Head Neck Pathol 6:275–278
Succaria F, Collier M, Mahalingam M (2015) Zoledronic acid–induced interface dermatitis. Am J Dermatopathol 37:933–935
Scheper MA, Chaisuparat R, Cullen KJ, Meiller TF (2010) A novel soft tissue in vitro model for biphosphonate-associated osteonecrosis. Fibrogenesis Tissue Repair 3:6–17
Walter C, Klein MO, Pabst A, Al-Nawas B, Duschner H, Ziebart H (2010) Influence of bisphosphonates on endothelial cells, fibroblasts, and osteogenic cells. Clin Oral Investig 14:35–41
Kory H, Kitagawa MD (2011) and Marcelle Grassi. Zoledronate acid-induced cutaneous B-Cell pseudolymphoma. J Am Acad Dermatol 65(6):1238–1240
European Medicines Agency (2008) Zoledronic acid. Summary of product characteristics. http://www.emea.europa.eu. Cited 4 Jan 2010
Rizos EC, Milionis HJ, Elisaf MS (2006) Fever with rash following zoledronate acid administration. Clin Exp Rheumatol 24(4):455
Rizzoli R, Reginster J-Y, Boonen S, Breart G, Felsenberg ADD, Kaufman J-M et al (2011) Adverse reactions and drug–drug interactions in the management of women with postmenopausal osteoporosis. Calcif Tissue Int 89:91–104
Musette P, Brandi M, Cacoub J, Kaufman M, Rizzoli R, Reginster J-Y (2010) Treatment of osteoporosis: recognizing and managing cutaneous adverse reactions and drug-induced hypersensitivity. Osteoporos Int 21:723–732
Author information
Authors and Affiliations
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nassar, K., Janani, S. Diffuse adverse cutaneous reactions induced by zoledronic acid administration: a case report. Osteoporos Int 32, 2583–2586 (2021). https://doi.org/10.1007/s00198-021-06021-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-06021-2