Abstract
Summary
The study investigates an association between phalangeal bone mineral density (BMD) and self-reported passive smoking using data on 15,038 persons (aged 18–95 years), who underwent a BMD scan in the Danish KRAM study. BMD was significantly lower in persons exposed to long-term passive smoking in their home during adulthood.
Introduction
Smoking is associated with decreased bone mineral density (BMD) and increased risk of osteoporotic fractures. This study aimed to investigate a possible association between BMD at the phalangeal bones and self-reported passive smoking.
Methods
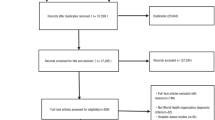
The study included a cohort of 15,544 men and women aged 18–95 years, who underwent a BMD scan in the Danish KRAM study. BMD scans of the middle phalanges of the second, third and fourth digits of the non-dominant hand were performed with a compact radiographic absorptiometry system (Alara MetriScan®). Also, height, weight and body fat percentage were measured and 96.7% (n = 15,038) of the participants answered a self-reported questionnaire with information on passive smoking, other lifestyle factors, education, etc. The association between passive smoking and BMD was examined using multiple linear regression analysis.
Results
A total of 39.1% (n = 5,829) of the participants had been exposed to passive smoking in adulthood at home. BMD was significantly lower in subjects exposed to passive smoking, 0.343 vs. 0.331 g/cm2; p < 0.01 (unadjusted) and 0.339 vs. 0.337 g/cm2; p < 0.05 (adjusted for age, gender, height and weight, and smoking). Multiple linear regression analysis showed that exposure to passive smoking for more than 20 years in adulthood at home was significantly related to BMD when adjusted for potential confounders (men, β = −4.4 × 10−3; r = −0.05; p < 0.01 and women, β = −2.3 × 10 −3; r = −0.03; p < 0.05). This relationship was also seen in the group of never smokers (β = −3.3 × 10−3; r = −0.03; p = 0.01).
Conclusion
Our study supports a potential negative effect of long-term passive smoking in adulthood at home on phalangeal BMD.

Similar content being viewed by others
References
World Health Organization (2007) Protection from exposure to second-hand tobacco smoke. Policy recommendations. World Health Organization
IARC (2002) International agency for reasearch on cancer. Tobacco smoke and involuntary smoking, vol 83. IARC, World Health Organization, Lyon
Barnoya J, Glantz SA (2005) Cardiovascular effects of secondhand smoke. Nearly as large as smoking. Circulation 111:2684–2698
Chan-Yeung M, Dimich-Ward H (2003) Respiratiry health effects of exposure to environmental tobacco smoke. Respirologu 8:131–139
World Health Organisation (2007) Assessment of osteoporosis at the primary health care level. World Health Organisation Scientific Group Technical Report
Johnell O, Kanis J (2005) Epidemiology of osteoporotic fractures. Osteoporos Int 16:3–7
Mossey JM, Mutran E, Knott K, Craik R (1989) Determinants of recovery 12 months after hip fracture: the importance of psychosocial factors. Am J Public Health 79(3):279–286
Vestergaard P, Rejnmark L, Mosekilde L (2007) Increased mortality in patients with a hip fracture-effect of pre-morbid conditions and post-fracture complications. Osteoporos Int 18:1583–1593
Brenneman SK, Barrett-Connor E, Sajjan S, Marksom LE, Siris ES (2006) Impact of recent fracture on health-related quality of life in postmenopausal women. J Bone Miner R 21(6):809–816
Oleksik A, Lips P, Dawsom A, Minshall ME, Shen W, Cooper S, Kanis J (2000) Health-related quality of life in postmenopausal women with low BMD or without prevalent vertebral fractures. J Bone Miner Res 15(7):1384–1392
Lunt M, Masaryk P, Scheidt-Nave C, Nijs J, Poor G, Pols H, Falch JA, Hammermeister G, Reid DM, Benevolenskaya L, Wener K, Cannata J, O’Niell TW, Felsenberg D, Silman AF, Reeve J (2001) The effects of lifestyle, dietary dairy intake and diabetes on bone density and vertebral deformity prevalence: the EVOS Study. Osteoporos Int 12:688–698
Law MR, Hackshaw AK (1997) A meta-analysis of cigarette smoking, bone mineral density and risk of hip fracture: recognition of a major effect. BMJ 315:841–846
Wong PKK, Christie JJ, Wark JD (2007) The effects of smoking on bone health. Clin Sci 113:233–241
Tziomalos K, Charsoulis F (2004) Endocrine effects of tobacco smoking. Clin Endocrinol 61:664–674
Høidrup S, Prescott E, Sørensen TIA, Gottschau A, Lauritzen JB, Schroll M, Grønbæk M (2000) Tobacco smoking and risk of hip fracture in men and women. Int J Epidemiol 29:253–259
MacInnis RJ, Cassar C, Nowson CA, Paton LM, Flicker L, Hopper JL, Larkins RG, Wark JD (2003) Determinats of bone density in 30- to 65-year-old women: a co-twin study. J Bone Miner Res 18(9):1650–1656
Hopper JL, Seeman E (1994) The bone density of female twins discordant for tobacco use. N Engl J Med 330(6):387–392
Kanis JA, Johnell O, Oden A, Johansson H, De Laet C, Eisman JA, Fujiwara S, Kroger H, McClosky EV, Mellstrom D, Melton LJ, Pols H, Reeve J, Silman A, Tenenhouse A (2005) Smoking and fracture risk: a meta-analysis. Osteoporos Int 16:155–162
Oncken C, Prestwood K, Kleppinger A, Wang Y, Cooney J, Raisz L (2006) Impact of smoking cessation on bone mineral density in postmenopausal women. J Women’s Health 15:1141–1150
Hollenbach KA, Barrett-Conner E, Edelstein SE, Holbrook T (1993) Cigarette smoking and bone mineral density in older men and women. Am J Public Health 83:1265–1270
Blum M, Harris SS, Must A, Phillips SM, Rand WM, Dawson-Hughes B (2002) Household tobacco smoke exposure is negativly associated with premenopausal bone mass. Osteoporos Int 13:663–668
Afghani A, Xie B, Wiswell RA, Gong J, Li Y, Johnson A (2003) Bone mass of Asian adolescents in China: influence of physical activity and smoking. Med Sci Sports Exerc 35(5):720–729
Altunbayrak O, Saridogan M, Ateser B, Akarirmak U (2009) Passive smoking and postmenopausal osteoporosis. Bone 44:s392, Abstract
Hsu YH, Bouxsein ML, Venners SA, Terwedow H, Feng Y, Rosen CJ, Xu X, Brain J (2006) Second-hand smoking increases the risk of osteoporosis and osteoporotic fractures in Chinese men and women. Osteoporos Int 17(Suppl 2):S144–S145, Abstract
Ajiro Y, Tokuhashi Y, Matsuzaki H, Nakajima S, Takashi T (2010) Impact of passive smoking on the bones of rats. Orthopedics 33(2):90–95
Matkovic V, Jelic T, Wardlaw GM, Ilich J, Goel PK, Wright JK, Andon MB, Smith KT, Heaney RP (1994) Timing of peak bone mass in Caucasian females and its implication for the prevention of osteoporosis. J Clin Invest 93:799–808
Boot AM, de Ridder AAJ, van der Sluis IM, van Slobbe I, Krenning EP, de Muinck Keizer-Schrama SMPF (2010) Peak bone mineral density, lean body mass and fractures. Bone 46:336–341
Lorentzon M, Mellström D, Ohlsson C (2005) Age of attainment of peak bone mass is site specific in Swedish men – The good study. J Bone Miner Res 20:1223–1227
Torgerson DJ, Campbell MK, Reid DM (1995) Life-style, environmental and medical factors influencing peak bone mass in women. Br J Rheumatol 34:620–624
Jones G, Riley M, Dwyer T (1999) Maternal smoking during pregnancy, growth, and bone mass in prepubertal children. J Bone Miner Res 14(1):146–151
U.S. Department of Health and Human Service (2006) The health consequences of involuntary exposure to tobacco smoke: a report of the Surgeon General. Atlanta (GA): U.S. Department of Health and Human Services, Centers for Disease Control and prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on smoking and Health. pp 17–23
Woodward A, Al-Delaimy W (1999) Measures of exposure to environmental tobacco smoke. Ann NY Acad Sci 895:156–172
Benson BW, Shulman JD (2005) Inclusion of tobacco exposure as a predictive factor for decreased bone mineral content. Nicotine Tob Res 7(5):719–724
Al-Delaimy WK, Willett WC (2008) Measurement of tobacco smoke exposure: comparison of toenail nicotine biomarkers and self-reports. Cancer Epidemiol Biomark Prev 17(5):1255–1261
Okoli CTC, Kelly T, Hahn EJ (2007) Secondhand smoke and nicotine exposure: a brief review. Addict Behav 32:1977–1988
Kanis JA (2002) Diagnosis of osteoporosis and assessment of fracture of risk. Lancet 359:1929–1936
Compston J, Cooper A, Cooper C, Francis R, Kanis JA, Marsh D et al (2009) Guidelines for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK. Maturitas 62(2):105–108
Hansen SJ, Nielsen MF, Ryg J, Wraae K, Andersen M, Brixen K (2009) Radiographic absorptiometry as a screening tool in male osteoporosis: results from the Odense Androgen study. Acta Radiolo 6:658–663
Boonen S, Nijs J, Borghs H, Peeters H, Vanderschueren D, Luyten FP (2005) Identifying postmenopausal women with osteoporosis by calcaneal ultrasound, metacarpal digital X-ray radiogrammetry and phalangeal radiographic absorptiometry: a comparative study. Osteoporos Int 16:93–100
Buch I, Qturai PS, Jensen LT (2010) Radiographic absorptiometry for pre-screening of osteoporosis in patients with low energy fractures. Scand J Clin Lab Invest 70:269–274
Thorpe JA, Steel SA (2008) The Alara Metriscan phalangeal densitometer: evaluation and triage thresholds. Br J Radiol 81:778–783
Abrahamsen B, Hansen TB, Jensen LB, Hermann AP, Eiken P (1997) Site of Osteodensitometry in perimenopausal women: correlation and limets and agreement between anatomic regions. J Bone Miner Res 12(9):1471–1479
Christensen AI, Severin M, Holmberg T, Eriksen L, Toftager M, Zachariassen A, Ekholm O, Tolstrup JS, Grønbæk M, Curtis T (2009) KRAM-undersøgelsen i tal og billeder [The KRAM-study in numbers and pictures]. The Danish Ministry of Health and Prevention and the TrygFoundation, Copenhagen
Statistics Denmark (2009) StatBank Denmark
Janson C, Künzli N, de Marco R, Chinn S, Jarvis D, Svanes C, Heinrich J, Jõgi R, Gislason T, Sunyer J, Ackermann-Liebrich U, Antó JM, Cerveri I, Karhof M, Leynaert B, Luczynska C, Neukirch VP, Wjst M, Burney P (2006) Changes in active and passive smoking in the European Community Respiratory Health Survey. Eur Resp J 27:517–524
U.S. Department of Health and Human Service (2006) The health consequences of involuntary exposure to tobacco smoke: a report of the Surgeon General. Atlanta (GA): U.S. Department of Health and Human Services, Centers for Disease Control and prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on smoking and Health. pp 127–158
Ekholm O, Kjøller M, Davidsen M, Hesse U, Eriksen L, Christensen AI, Grønbæk M (2006) Sundhed og sygelighed i Danmark 2005 & udviklingen siden 1987 [Health and morbidity in Denmark 2005]. National Institute of Public Health, Copenhagen
Fangel S, Linde PC, Thorsted BL (2007) Nye problemer med repræsentativitet i Surveys, som opregning med registre kan reducere [New problems with representativity which can be reduced with enumeration in registres]. Metode og Data 93:14–25
Acknowledgements
The KRAM study—the Danish Health Examination Survey 2007–2008 was funded by the Ministry of Health and Prevention and the TrygFoundation, Denmark. Thanks to the 13 participating municipalities and to all the participants who took part in the study by completing questionnaires and participating in health examinations. Finally, the data collection would not have been possible without the immense effort from all the masters and students from the National Institute of Public Health and local nurses and students from the 13 municipalities. The execution of this manuscript was funded by the Region of Southern Denmark.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holmberg, T., Bech, M., Curtis, T. et al. Association between passive smoking in adulthood and phalangeal bone mineral density: results from the KRAM study—the Danish Health Examination Survey 2007–2008. Osteoporos Int 22, 2989–2999 (2011). https://doi.org/10.1007/s00198-010-1506-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1506-9