Abstract
I estimate the effects of screening “low-risk” women for gestational diabetes using a regression discontinuity design and exploiting exogenous variation in testing at the overweight threshold of the body mass index in Finland. I find that screening low-risk mothers just above the overweight threshold increases the number of mothers diagnosed with gestational diabetes. There is a 1.5 percentage point, or 27%, increase in the probability of being diagnosed with gestational diabetes at the threshold, which translates into a 10.7 percentage point local average treatment effect given the 14.0 percentage point jump in the screening rates. The estimates on the effect on insulin treatment are, however, small and imprecise, suggesting that screening low-risk mothers did not result in diagnoses needing insulin treatment. The cost estimates in the existing literature suggest that the policy is cost-effective. The results also suggest that universal screening could decrease health disparities between mothers with low and high levels of education, given that gestational diabetes is treated if diagnosed. The effect on the probability of having an abnormal test result is over twice as great for the less educated mothers compared to the more educated mothers. Large effects of around 10–20% on adverse birth outcomes (low birth weight, macrosomia, metabolic testing, and C-section) can be ruled out.



Similar content being viewed by others
Data Availability
The dataset generated during the current study is not publicly available as it contains proprietary information that the authors acquired through a license. Information on how to obtain it and reproduce the analysis is available from the corresponding author upon request.
Code Availability
The do-file used for analysis is collected in the electronic supplementary material of this article.
Notes
Gestational diabetes used from here on. The American Diabetes Association defines gestational diabetes as follows: “any degree of glucose intolerance with onset or first recognized during pregnancy. The definition applies regardless of whether insulin or only diet modification is used for treatment or whether the condition persists after pregnancy. It does not exclude the possibility that unrecognized glucose intolerance may have antedated or begun concomitantly with the pregnancy.”
In Finland, the main rule is that all pregnant women should be screened for gestational diabetes with the oral glucose tolerance test; the few exceptions to this rule are presented in this paper.
That is, low-risk for developing gestational diabetes.
The Current Care Guidelines (Käypä hoito) are national, independent, evidence-based clinical practice guidelines that cover important issues related to Finnish health, medical treatment and prevention of disease. The guidelines are intended as a basis for treatment decisions and can be used by physicians, health care professionals and citizens. They were developed by the Finnish Medical Society Duodecim in association with various medical specialist societies.
When blood glucose levels in the body are too high (> 10 mmol/L), excess glucose can end up in the urine which can indicate gestational diabetes. Urine screening is conducted on all visits, even if the mother has tested negative for gestational diabetes with the OGTT.
In the analysis, I look at all instances of the OGTT regardless of the gestational week it was conducted. The data lack information on the week in which the test was conducted.
The cost of the test is from Terveystalo’s, a Finnish private health clinic, laboratory price list in 2015, the last year of the study period.
A descriptive population-based register study was conducted in Finland comparing women giving birth before the new guidelines were introduced in 2006 to women giving birth in 2010 after the new guidelines were implemented (Koivunen et al. 2015). The results suggest that the change in guidelines from a risk factor-driven approach to a comprehensive policy led to a significant increase—from 9.1% to 11.3%—in the prevalence of gestational diabetes, which was due to the increased number of mothers who could be treated with diet. Both the proportion and total number of insulin-treated women decreased significantly from 21.8% to 13.3%, suggesting that wider screening did not perform better in diagnosing women needing insulin treatment.
BMI or Quetelet index is a measure of relative size based on the mass and height of an individual; it is body mass divided by the square of height and always reported in \(kg/m^{2}\). A BMI from 18.5 to 25 may indicate optimal weight, while a BMI under 18.5 is considered underweight, and a BMI of over 25 is considered overweight. A person with a BMI of 30 or greater is considered obese.
The BMI is usually checked from a chart, where the y-axis represents the height of the person and the x-axis the weight. For example, a person who weighs 62 kg and is 158 cm tall would have a BMI of 25 according to the chart, while her actual BMI is 24.8 (62/(1.58*1.58) according to the formula. Hence, according to the chart, she is overweight, but according to the BMI formula, she is not. Similarly, if she weighed 61.5 kg, her weight would be rounded to 62, and hence, her BMI would be 25 according to the chart and 24.6 according to the formula.
16.7% (10.7 + 6) prevalence of positive test results for those who were screened due to crossing the overweight threshold versus 19% for those who were screened just under the threshold and are at risk.
The one-hour oral glucose challenge test (GCT) is a screening test for gestational diabetes that measures serum glucose concentration one hour after a 50 g oral glucose drink.
Estimated sample sizes for a two-sample proportions test (Pearson’s chi-squared test.)
References
Alalouf M, Miller S, Wherry LR (2019) What difference does a diagnosis make? Evidence from marginal patients. Working paper (26363)
Almond D, Doyle JJ, Kowalski AE, Williams H (2010) Estimating marginal returns to medical care: evidence from at-risk newborns. Q J Econ 125(2):591–634
Almond D, Lee A, Schwartz AE (2016) Impacts of classifying New York City students as overweight. Proc Natl Acad Sci 113(13):3488–3491
Angrist JD, Pischke JS (2008) Mostly harmless econometrics: an empiricist’s companion. Princeton University Press, Princeton
Barreca AI, Guldi M, Lindo JM, Waddell GR (2011) Saving babies? Revisiting the effect of very low birth weight classification. Q J Econ 126(4):2117–2123
Burkhauser RV, Cawley J (2008) Beyond bmi: the value of more accurate measures of fatness and obesity in social science research. J Health Econ 27(2):519–529
Calonico S, Cattaneo MD, Titiunik R (2014) Robust nonparametric confidence intervals for regression-discontinuity designs. Econometrica 82(6):2295–2326
Chen Y, Quick WW, Yang W, Zhang Y, Baldwin A, Moran J, Moore V, Sahai N, Dall TM (2009) Cost of gestational diabetes mellitus in the United States in 2007. Popul Health Manag 12(3):165–174
Crowther CA, Hiller JE, Moss JR, McPhee AJ, Jeffries WS, Robinson JS (2005) Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med 352(24):2477–2486
Currie J, Stabile M (2003) Socioeconomic status and child health: Why is the relationship stronger for older children? Am Econ Rev 93(5):1813–1823
Cutler DM, Lleras-Muney A (2010) Understanding differences in health behaviors by education. J Health Econ 29(1):1–28
Cutler DM, Vogl T (2011). Socioeconomic status and health: dimensions and mechanisms, Oxford handbook of health economics
Cygan-Rehm K, Karbownik K (2022) The effects of incentivizing early prenatal care on infant health. J Health Econ 83:102612
DeSisto CL (2014) Prevalence estimates of gestational diabetes mellitus in the United States, pregnancy risk assessment monitoring system (PRAMS), 2007–2010. Preventing chronic disease 11
Evans WN, Lien DS (2005) The benefits of prenatal care: evidence from the pat bus strike. J Econ 125(1–2):207–239
Farrar D, Duley L, Dowswell T, Lawlor DA (2017) Different strategies for diagnosing gestational diabetes to improve maternal and infant health. Cochrane Database of Syst Rev. No. 8
Ferrara A (2007) Increasing prevalence of gestational diabetes mellitus a public health perspective. Diabetes Care 30(Supplement 2):S141–S146
Gaggero A, Gil J, Jiménez-Rubio D, Zucchelli E (2021) Health information and lifestyle behaviours: the impact of a diabetes diagnosis. IZA Discussion Papers (No. 14106)
Galama TJ, Van Kippersluis H (2019) A theory of socio-economic disparities in health over the life cycle. Econ J 129(617):338–374
Gelman A, Imbens G (2014) Why high-order polynomials should not be used in regression discontinuity designs. Technical report, National Bureau of Economic Research
Goldman DP, Smith JP (2002) Can patient self-management help explain the SES health gradient? Proc Natl Acad Sci 99(16):10929–10934
Hahn J, Todd P, Van der Klaauw W (2001) Identification and estimation of treatment effects with a regression-discontinuity design. Econometrica 69(1):201–209
Heude B, Thiébaugeorges O, Goua V, Forhan A, Kaminski M, Foliguet B, Schweitzer M, Magnin G, Charles MA, Group EMCCS et al (2012) Pre-pregnancy body mass index and weight gain during pregnancy: relations with gestational diabetes and hypertension, and birth outcomes. Matern Child Health J 16(2):355–363
Imbens G, Kalyanaraman K (2012) Optimal bandwidth choice for the regression discontinuity estimator. Rev Econ Stud 79(3):933–959
Jolly MC, Sebire NJ, Harris JP, Regan L, Robinson S (2003) Risk factors for macrosomia and its clinical consequences: a study of 350,311 pregnancies. Eur J Obstet Gynecol Reprod Biol 111(1):9–14
Kim C, Newton KM, Knopp RH (2002) Gestational diabetes and the incidence of type 2 diabetes a systematic review. Diabetes Care 25(10):1862–1868
Kjos SL, Buchanan TA (1999) Gestational diabetes mellitus. N Engl J Med 341(23):1749–1756
Koivunen S, Kajantie E, Torkki A, Bloigu A, Gissler M, Pouta A, Vääräsmäki M (2015) The changing face of gestational diabetes: the effect of the shift from risk factor-based to comprehensive screening. Eur J Endocrinol 173(5):623–632
Kolu P, Raitanen J, Rissanen P, Luoto R (2012) Health care costs associated with gestational diabetes mellitus among high-risk women: results from a randomised trial. BMC Pregnancy Childbirth 12(1):71
Lamberg S, Raitanen J, Rissanen P, Luoto R (2012) Prevalence and regional differences of gestational diabetes mellitus and oral glucose tolerance tests in Finland. Eur J Public Health 22(2):278–280
Langer O, Yogev Y, Most O, Xenakis EM (2005) Gestational diabetes: the consequences of not treating. Am J Obstet Gynecol 192(4):989–997
Lee DS, Lemieux T (2010) Regression discontinuity designs in economics. J Econ Lit 48:281–355
Luoto R, Kinnunen TI, Aittasalo M, Kolu P, Raitanen J, Ojala K, Mansikkamäki K, Lamberg S, Vasankari T, Komulainen T et al (2011) Primary prevention of gestational diabetes mellitus and large-for-gestational-age newborns by lifestyle counseling: a cluster-randomized controlled trial. PLoS Med 8(5):e1001036
Mission JF, Ohno MS, Cheng YW, Caughey AB (2012) Gestational diabetes screening with the new iadpsg guidelines: a cost-effectiveness analysis. Am J Obstet Gynecol 207(4):326
Mulder EJ, De Medina PR, Huizink AC, Van den Bergh BR, Buitelaar JK, Visser GH (2002) Prenatal maternal stress: effects on pregnancy and the (unborn) child. Early Human Dev 70(1–2):3–14
Nicholson WK, Fleisher LA, Fox HE, Powe NR (2005) Screening for gestational diabetes mellitus: a decision and cost-effectiveness analysis of four screening strategies. Diabetes Care 28(6):1482–1484
Nilofer AR, Raju V, Dakshayini B, Zaki SA (2012) Screening in high-risk group of gestational diabetes mellitus with its maternal and fetal outcomes. Indian J Endocrinol Metab 16(Suppl1):S74
Round J, Jacklin P, Fraser R, Hughes R, Mugglestone M, Holt R (2011) Screening for gestational diabetes mellitus: cost-utility of different screening strategies based on a woman’s individual risk of disease. Diabetologia 54(2):256–263
Sacks KN, Friger M, Shoham-Vardi I, Abokaf H, Spiegel E, Sergienko R, Landau D, Sheiner E (2016) Prenatal exposure to gestational diabetes mellitus as an independent risk factor for long-term neuropsychiatric morbidity of the offspring. Am J Obstet Gynecol 215(3):380-e1
Schwartz J, Phillips W, Aghebat-Khairy B (1990) Revision of the oral glucose tolerance test: a pilot study. Clin Chem 36(1):125–128
Scott D, Loveman E, McIntyre L, Waugh N (2002) Screening for gestational diabetes: a systematic review and economic evaluation. Database of Abstracts of Reviews of Effects (DARE): Quality-assessed Reviews [Internet]
Song D, Hurley JC, Lia M (2020) Estimated treatment effects of tight glycaemic targets in mild gestational diabetes mellitus: a multiple cut-off regression discontinuity study design. Int J Environ Res Public Health 17(21):7725
Stommel M, Schoenborn CA (2010) Variations in BMI and prevalence of health risks in diverse racial and ethnic populations. Obesity 18(9):1821–1826
Tennant P, Doxford-Hook E, Flynn L, Kershaw K, Goddard J, Stacey T (2022) Fasting plasma glucose, diagnosis of gestational diabetes and the risk of large for gestational age: a regression discontinuity analysis of routine data. BJOG Int J Obstetr Gynaecol 129(1):82–89
Tieu J, McPhee AJ, Crowther CA, Middleton P, Shepherd E (2017) Screening for gestational diabetes mellitus based on different risk profiles and settings for improving maternal and infant health. Cochrane Database Syst Rev (8)
Torloni M, Betran A, Horta B, Nakamura M, Atallah A, Moron A, Valente O (2009) Prepregnancy bmi and the risk of gestational diabetes: a systematic review of the literature with meta-analysis. Obes Rev 10(2):194–203
Yan J (2017) The effects of prenatal care utilization on maternal health and health behaviors. Health Econ 26(8):1001–1018
Acknowledgements
I thank Kristiina Huttunen, Manuel Bagues, Marko Terviö, Kaisa Kotakorpi, Matti Sarvimäki, Markku Siikanen, Katrine Løken and Libertad Gonzalez for valuable suggestions and comments. The paper has also benefited from comments of seminar participants at Helsinki Center of Economic Research (HECER) and the 11th Nordic Summer Institute in Labour Economics. I extend my gratitude to the journal editor, Professor van Soest, and the anonymous reviewers for their constructive and helpful comments. I thank the OP Group Research Foundation (Grant nos. 201600040, 201500016 & 201300023) and the Yrjö Jahnsson Foundation for funding.
Funding
This study was funded by the OP Group Research Foundation and the Yrjö Jahnsson Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Krista Riukula declares that she has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by the author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Appendices
Appendix A Additional tables and figures
Mother’s characteristics. See Fig. 2 for figure notes
Mother’s height and weight. See Fig. 2 for figure notes
First-time mothers over age 25. See Fig. 2 for figure notes. First-time mothers over age 25 should be screened regardless of their BMI
Appendix B Effect of screening on birth outcomes
The results suggest that the expansion of screening increased the number of cases that could be treated with dietary advice. A natural question is whether screening affects birth outcomes. However, it is not clear how screening might affect birth outcomes and the mechanism behind any such effects. Mothers are already given dietary advice at the beginning of their pregnancy, as Fig. 1 shows, and I could not find an effect on insulin treatment. Furthermore, there might be some unanticipated consequences if a mother has normal test results while having a “bad lifestyle” or diet or in the case of abnormal test results causing stress, both of which might cancel out the positive effects of treatment. It has been shown that maternal psychological factors like stress may significantly contribute to pregnancy complications and unfavorable development of the (unborn) child (see, e.g., Mulder et al. 2002, for a review).
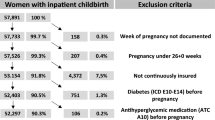
A larger problem concerns issues of power. As I do not expect to see large effects of screening on many birth outcomes, and as adverse birth outcomes are rather rare in today’s Finland, the sample sizes needed to detect the effects would be enormous. Table 9 shows the population sizes needed to detect different effect sizes for multiple birth outcomes with the power of 0.8. The estimated sample sizes are calculated using Pearson’s chi-squared test and are estimations for a two-sample proportions test. In order to detect a 1% effect on low birth weight, I would need 10 million observations. With the current sample size of roughly 70,000 births varying with the bandwidth used, I am able to detect with high certainty an effect size of 7% to 20% depending on the outcome variable. Hence, it is highly unlikely that significant estimates would be found, given the small sample and expected effect sizes.
Table 10 provides estimates from the local linear regressions using a triangular kernel for birth outcomes. The estimates from the first-stage regression (i.e., the jump in the treatment) varies from 12.1 to 13.3 percentage points, depending on the bandwidth used. The optimal bandwidths obtained with the CCT bandwidth selector are now considerably lower (1.40 to 1.97 BMI units) than in the previous analysis. The intention-to-treat effects for adverse birth outcomes are small, ranging from −0.002 to 0.007 and imprecise due to the low fraction of adverse birth outcomes and insufficient mass around the cutoff. The IV estimates range from −0.012 to 0.054. Hence, I cannot rule out that there is no effect or a larger effect. For most of the outcomes, however, I can rule out effects larger than 10–20%. However, for C-section, which is the most frequent outcome, I am able to detect an effect of 7% with a power of 0.8 and a sample size of roughly 55,000, or 27,500 on both sides,Footnote 13 as shown in Table 9. Hence, the chances that the effect is larger than 7% are very small because the sample size is around 52,000.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Riukula, K. The effects of screening for gestational diabetes. Empir Econ 65, 1931–1964 (2023). https://doi.org/10.1007/s00181-023-02397-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00181-023-02397-8