Abstract
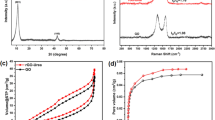
This study aims to investigate the electrochemical reduction effects on graphene oxide (GO) powders with various bias voltages and treatment times. Phosphate-buffered saline solution was used as the electrolyte in the electrochemical reduction process. The experimental results showed that the GO powders were reduced to produce the best reduced GO (rGO) powders as using a bias of −17.5 V for 2 h. After the analysis of Raman spectra for GO and rGO powders, the intensity ratios of the D and G bands increased from 0.85 to 1.08, respectively. The carbon to oxygen ratios increased from 0.4 to 1.79 measured by an X-ray photoelectron spectroscopy. Moreover, the electrical conductivity obviously increased from 7.92 × 10−4 to 4.16 × 10−1 S/cm. Fourier transform infrared spectra revealed the disappearance of oxygen-containing functional groups in rGO powders. According to the cyclic voltammetry analysis, the specific capacitance of the rGO powders could reach 183 F/g at the scan rate of 100 mV/S in 1 M KCl electrolyte solution. This specific capacitance value was 16 times higher than that obtained with the GO powders. The results indicated that the produced high-quality rGO powders could be used for high-performance supercapacitors.











Similar content being viewed by others
References
Wang Y, Shi Z, Huang Y, Ma Y, Wang C, Chen M, Chen Y (2009) Supercapitor devices based on graphene materials. J Phys Chem C 113:13103–13107
Iqbal MF, Ashiq MN, Hassan M-U, Nawaz R, Masood A, Razaq A (2018) Excellent electrochemical behavior of graphene oxide based aluminum sulfide nanowalls for supercapacitor applications. Energy 159:151–159
Yanik MO, Yigit EA, Akansu YE, Sahmetlioglu E (2017) Magnetic conductive polymer-graphene nanocomposites based supercapacitors for energy storage. Energy 138:883–889
Khalaj M, Sedghi A, Miankushki HN, Golkhatmi SZ (2019) Synthesis of novel graphene/Co3O4/polypyrrole ternary nanocomposites as electrochemically enhanced supercapacitor electrodes. Energy 188:116088
Kumar R, Joanni E, Savu R, Pereira MS, Singh RK, Constantino CJL, Kubota LT, Matsuda A, Moshkalev SA (2019) Fabrication and electrochemical evaluation of micro supercapacitors prepared by direct laser writing on free-standing graphite oxide paper. Energy 179:676–684
Li X, Zhu H, Wang K, Cao A, Wei J, Li C, Jia Y, Li Z, Li X, Wu D (2010) Graphene-on-silicon schottky junction solar cells. Adv Mater 22:2743–2748
Yue G, Wu J, Xiao Y, Lin J, Huang M, Lan Z, Fan L (2013) Functionalized graphene/poly(3,4-ethylenedioxythiophene):polystyrenesulfonate as counter electrode catalyst for dye-sensitized solar cells. Energy 54:315–321
Pei S, Cheng HM (2012) The reduction of graphene oxide. Carbon 50:3210–3228
Luo D, Zhang G, Liu J, Sun X (2011) Evaluation criteria for reduced graphene oxide. J Phys Chem C 115:11327–11335
Kang S, Zhuang J, Kang S, Peng Y, Guan S (2019) Synthesis of high-quality graphene with enhanced electrochemical properties by two-step reduction method. Ceram Int 45(18):23954–23965
Ismail Z (2019) Green reduction of graphene oxide by plant extracts: A short review. Ceram Int 45(18):23857–23868
Ma Q, Song J, Jin C, Li Z, Liu J, Meng S, Zhao J, Guo Y (2013) A rapid and easy approach for the reduction of graphene oxide by formamidinesulfinic acid. Carbon 54:36–41
Song NJ, Chen CM, Lu C, Liu Z, Kong QQ, Cai R (2014) Thermally reduced graphene oxide films as flexible lateral heat spreaders. J Mater Chem A 2:16563–16568
Zhang P, Shen Y, Wu W, Li J, Zhou Z (2018) Enhanced photocatalytic performance of KNbO3(100)/reduced graphene oxide nanocomposites investigated using first-principles calculations: RGO reductivity effect. Appl Surf Sci 434:932–939
Low CTJ, Walsh FC, Chakrabarti MH, Hashim MA, Hussain MA (2013) Electrochemical approaches to the production of graphene flakes and their potential applications. Carbon 54:1–21
Inamdar AI, Jo Y, Kim J, Han J, Pawar SM, Kalubarme RS, Park CJ, Hong JP, Park YS, Jung W, Kim H, Im H (2015) Synthesis and enhanced electrochemical supercapacitive properties of manganese oxide nanoflake electrodes. Energy 83:532–538
Liu S, Wang J, Zeng J, Ou J, Li Z, Liu X, Yang S (2010) Green electrochemical synthesis of Pt/ graphene sheet nanocomposite film and its electrocatalytic property. J Power Sources 195:4628–4633
Zhou M, Wang Y, Zhai Y, Zhai J, Ren W, Wang F, Dong S (2009) Controlled synthesis of large-area and patterned electrochemically reduced graphene oxide films. Chem Eur J 15:6116–6120
Guo HL, Wang XF, Qian QY, Wang FB, Xia XH (2009) A green approach to the synthesis of graphene nanosheets. ACS Nano 3:2653–2659
Ramachandran R, Felix S, Joshi GM, Raghupathy BPC, Jeong SK, Grace AN (2013) Synthesis of graphene platelets by chemical and electrochemical route. Mater Res Bull 48:3834–3842
Wang M, Duong LD, Oh JS, Mai NT, Kim S, Hong S, Hwang T, Lee Y, Nam JD (2014) Large-area conductive and flexible reduced graphene oxide (RGO) membrane fabricated by electrophoretic deposition (EPD). ACS Appl Mater Interfaces 6:1747–1753
Lin DC, Liu YY, Lee HW, Sun J, Wang HT, Yan K, Xie J, Cui Y (2016) Layered reduced graphene oxide with nanoscale interlayer gaps as a stable host or lithium metal anodes. Nat Nanotechnol 11:626–632
XSabina D, Roksana M, Agnieszka S, Tadeusz P, Michalina KM, Maciej S (2016) Studies of reduced graphene oxide and graphite oxide in the aspect of their possible application in gas sensors. Sensors 16:103–118
Beatiriz MG, Yu B, Mirko P, Davide S, Roman K, Gerasimos K, Iwan M (2018) Reduction of moisture sensitivity of PbS quantum dot solar cells by incorporation of reduced graphene oxide. Sol Energy Mater Sol Cells 183:1–7
Mani V, Devadas B, Chen SM (2013) Direct electrochemistry of glucose oxidase at electrochemically reduced graphene oxide-multiwalled carbon nanotubes hybrid material modified electrode for glucose biosensor. Biosens Bioelectron 41:309–315
Jeon JW, Kwon SR, Lutkenhaus JL (2015) Polyaniline nanofiber/ electrochemically reduced graphene oxide layer-by-layer electrodes for electrochemical energy storage. J Mater Chem A 3:3757–3767
Liu X, Qi X, Zhang Z, Ren L, Hao G, Liu Y, Wang Y, Huang K, Wei X, Huang Z, Zhong J (2014) Electrochemically reduced graphene oxide with porous structure as binder-free electrode for high-rate supercapacitors. RSC Adv 4:13673–13679
Aized T, Khan MB, Raza H, Ilyas M (2017) Production routes, electromechanical properties and potential application of layered nanomaterials and 2D nanopolymeric composites–a review. Int J Adv Manuf Technol 93:3449–3459
Li M, Liu J, Zhang X, Tian Y, Jiang K (2018) Fabrication of graphene/ nickel composite microcomponents using electroforming. Int J Adv Manuf Technol 96:3191–3196
Hanif M, Wasim A, Shah AH, Noor S, Sajid M, Mujtaba N (2019) Optimization of process parameters using grpahene-based dielectric in electric discharge machining of AISI D2 steel. Int J Adv Manuf Technol 103:3735–3749
Wang X, Yi S, Guo H, Li CJ, Ding SL (2020) Erosion characteristics of electrical discharge machining using graphene powder in deionized water as dielectric. Int J Adv Manuf Technol 108:357–368
Purkait T, Singh G, Kuamr D, Singh M, Dey RS (2018) High-performance flexible supercapacitors based on electrochemically tailored three-dimensional reduced graphene oxide networks. Sci Rep 8:640–652
Hqaque AMJ, Park H, Sung D, Jon S, Choi SY, Kim K (2012) An electrochemically reduced graphene oxide-based electrochemical immunosensing platform for ultrasensitive antigen detection. Anal Chem 84:1871–1878
Khan MMI, Haque AMJ, Kim K (2013) Electrochemical determination of uric acid in the presence of ascorbic acid on electrochemically reduced graphene oxide modified electrode. J Electroanal Chem 700:54–59
Ghanashyam G, Jeong HK (2018) Thermally reduced graphite oxide-titanium dioxide composites for supercapacitors. Chem Phys Lett 706:421–425
Ramadoss A, Saravanakumar B, Kim SJ (2015) Thermally reduced graphene oxide-coated fabrics for flexible supercapacitors and self-powered systems. Nano Energy 15:587–597
Zhao B, Liu P, Jiang Y, Pan D, Tao H, Song J, Fang T, Xu W (2012) Supercapacitor performances of thermally reduced graphene oxide. J Power Sources 198:423–427
Zhang K, Mao L, Zhang LL, Chan HSO, Zhao XS, Wu J (2011) Surfactant-intercalated, chemically reduced graphene oxide for high performance supercapacitor electrodes. J Mater Chem 21:7302–7307
Fang Z, Zhao Q, Li T, Yan J, Ren Y, Feng J, Wei T (2012) Easy synthesis of porous graphene nanosheets and their use in supercapacitors. Carbon 50:1699–1712
Peng XY, Liu XX, Diamond D, Lau KT (2011) Synthesis of electrochemically-reduced graphene oxide film with controllable size and thickness and its use in supercapacitor. Carbon 49:3488–3496
Yang J, Gunasekaran S (2013) Electrochemically reduced graphene oxide sheets for use in high performance supercapacitors. Carbon 51:36–44
Yang Q, Pang SK, Yung KC (2016) Electrochemically reduced graphene oxide/carbon nanotubes composites as binder-free supercapacitor electrodes. J Power Sources 311:144–152
Hung YF, Cheng C, Huang CK, Yang CR (2018) A facile method for batch preparation of electrochemically reduced graphene oxide. Nanomaterials 9:376–391
Bruzzone AAA, Costa HL, Lonardo PM, Lucca DA (2008) Advances in engineered surfaces for functional performance. CIRP Ann Manuf Technol 57:750–769
Ribeiro FSF, Lopes JC, Bianchi EC, Sanchez LE (2020) Applications of texturization techniques on cutting tools —a survey. Int J Adv Manuf Technol 109:1117–1135
Koshy P, Tovey J (2011) Performance of electrical discharge textured cutting tools. CIRP Ann Manuf Technol 60:153–156
Escudero ML, Llorente I, Pérez-Maceda BT, José-Pinilla SS, Sánchez-López L, Lozano RM, Aguado-Henche S, Clemente de Arriba C, Alobera-Gracia MA, García-Alonso MC (2020) Electrochemically reduced graphene oxide on CoCr biomedical alloy: Characterization, macrophage biocompatibility and hemocompatibility in rats with graphene and graphene oxide. Mater Sci Eng C 109:110522
Hong BJ, Compton OC, An Z, Eryazici I, Nguyen ST (2012) Successful stabilization of graphene oxide in electrolyte solutions: enhancement of biofunctionalization and cellular uptake. ACS Nano 6:63–73
Rahman JU, Du NV, Nam WH, Shin WH, Lee KH, Seo W-S, Kim MH, Lee S (2019) Grain boundary interfaces controlled by reduced graphene oxide in nonstoichiometric SrTiO3-δ thermoelectrics. Sci Rep 9:8624
Yang S, Yue W, Huang D, Chen C, Lin H, Yang X (2012) A facile green strategy for rapid reduction of graphene oxide by metallic zinc. RSC Adv 2:8827–8832
Wang Y, Shi Z, Yin J (2011) Facile synthesis of soluble graphene via a green reduction of graphene oxide in tea solution and its biocomposites. ACS Appl Mater Interfaces 3:1127–1133
Thakur S, Karak N (2012) Green reduction of graphene oxide by aqueous phytoextracts. Carbon 50:5331–5339
Ding JN, Liu YB, Yuan NY, Ding GQ, Fan Y, Yu CT (2012) The influence of temperature, time and concentration on the dispersion of reduced graphene oxide prepared by hydrothermal reduction. Diam Relat Mater 21:11–15
Park S, An JH, Jung IW, Piner RD, An SJ, Li XS, Velamakanni A, Ruoff RS (2009) Colloidal suspensions of highly reduced graphene oxide in a wide variety of organic solvents. Nano Lett 9:1593–1597
Allahbakhsh A, Sharif F, Mazinani S, Kalaee MR (2014) Synthesis and characterization of graphene oxide in suspension and powder forms by chemical exfoliation method. Int J Nano Nano Dimens 5:11–20
Kaniyoor A, Baby TT, Ramaprabhu S (2010) Graphene synthesis via hydrogen induced low temperature exfoliation of graphite oxide. J Mater Chem 20:8467–8469
You JM, Kim D, Kim SK, Kim MS, Han HS, Jeon S (2013) Novel determination of hydrogen peroxide by electrochemically reduced graphene oxide grafted with aminothiophenol-Pd nanoparticles. Sens. Actuators B Chem 178:450–457
Li M, Liu Z, Ruan J, Chen X, Xu F, Chen X, Lu X, Yang S (2014) Noncovalently grafting sulfonic acid onto graphene oxide for improved hole transport in polymer solar cells. RSC Adv 4:53999–54006
Zhang Y, Yuan S, Zhou W, Xu J, Li Y (2007) Spectroscopic evidence and molecular simulation investigation of the pi-pi interaction between pyrene molecules and carbon nanotubes. J Nanosci Nanotechnol 7:2366–2375
Zhang J, Yang H, Shen G, Cheng P, Zhang J, Guo S (2010) Reduction of graphene oxide via L-ascorbic acid. Chem Commun 46:1112–1114
Bartlam C, Morsch S, Heard KWJ, Quayle P, Yeates SG, Vijayaraghvan A (2018) Nanoscale infrared identification and mapping of chemical functional groups on graphene. Carbon 139:317–324
Chua CK, Pumera M (2014) Chemical reduction of graphene oxide: a synthetic chemistry viewpoint. Chem Soc Rev 43:291–312
Kumar A, Khandelwal M (2014) Amino acid mediated functionalization and reduction of graphene oxide—synthesis and the formation mechanism of nitrogen-doped graphene. N J Chem 38:3457–3467
Anwar AW, Majeed A, Iqbal N, Ullah W, Shuaib A, Ilyas U, Bibi F, Rafique HM (2015) Specific capacitance and cyclic stability of graphene based metal/metal oxide nanocomposites: a review. J Mater Sci Technol 31:699–707
Harima Y, Setodoi S, Imae I, Komaguchi K, Ooyama Y, Ohshita J, Mizota H, Yano J (2011) Electrochemical reduction of graphene oxide in organic solvents. Electrochim Acta 56:5363–5368
Ke Q, Liu Y, Liu H, Zhang Y, Hu Y, Wang J (2014) Surfactant-modified chemically reduced graphene oxide for electrochemical supercapacitors. RSC Adv 4:26398–26406
Acknowledgments
The authors gratefully acknowledge the financial support for this study provided by Ministry of Science and Technology (MOST), Taiwan under projects MOST 107-2622-E-003-005-CC3 and MOST 108-2622-E-027-019-CC3.
Funding
This study received financial support from Ministry of Science and Technology (MOST), Taiwan, under projects MOST 107-2622-E-003-005-CC3 and MOST 108-2622-E-027-019-CC3.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hung, YF., Cheng, C., Huang, CK. et al. Investigation of electrochemical reduction effects on graphene oxide powders for high-performance supercapacitors. Int J Adv Manuf Technol 113, 1203–1213 (2021). https://doi.org/10.1007/s00170-020-06578-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00170-020-06578-y