Abstract
Purpose
The purpose of this study was to investigate the learning curve associated with robotic assisted knee arthroplasty (RAS KA). Therefore, the evaluation of the influence of an experienced surgeon on the overall team performance of three surgeons regarding the learning curve in RAS KA was investigated. It was hypothesized that the presence of an experienced surgeon flattens the learning curve and that there was no inflection point for the learning curve of the surgical team.
Methods
Fifty-five cases consisting of 31 total knee arthroplasties (TKA) and 24 unicompartmental arthroplasties (UKA) performed by three surgeons during 2021 were prospectively investigated. Single surgeon and team performance for operation time learning curve and inflection points were investigated using cumulative sum analysis (CUSUM).
Results
A downward trend line for individual surgeons and the team performance regarding the operation time learning curve was observed. No inflexion point was observed for the overall team performance regarding TKA and UKA. The surgeon that performed all cases with the assistance of the experienced surgeon had significantly shorter surgical times than the surgeon that only occasionally received assistance from the experienced surgeon (p = 0.004 TKA; p = 0.002 UKA).
Conclusion
The presence of an experienced surgeon in robotically assisted knee arthroplasty can flatten the learning curve of the surgical team formerly unexperienced in robotic assisted systems. Manufacturers should provide expanded support during initial cases in centres without previous experience to robotic assisted knee arthroplasty.
Level of evidence
III.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Learning curves are well known in medical education and surgical training [9]. Robotic assisted surgery (RAS) is becoming increasingly popular in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty (UKA) [4]. RAS has demonstrated more accurate implant positioning in vivo and in vitro when compared to the conventional instruments [1, 4, 8].
For both TKA and UKA, RAS has shown no learning curve with the implant positioning but a significant learning curve with the workflow and instrumentation [3, 4, 12]. Surgical time flattens out effect after 5–10 cases and, in most studies, reaches a steady value after 8 cases in image-based RAS for UKA and TKA [4, 6, 11, 17, 18, 20, 22]. RAS offers a good opportunity for low-volume arthroplasty surgeons to achieve high levels of implant position accuracy in TKA and UKA [10].
Literature to date mainly reflects single surgeon performance, or a group's performance where the whole group goes through the learning curve simultaneously [19, 20]. Individual surgeon characteristics, such as experience with arthroplasty or experience with digital technology as are, however, difficult to quantify and some studies show a learning curve up to 40 cases [21]. Since each centre and thus, surgeons within that centre, start with RAS simultaneously, it remains unclear if the presence of a RAS-experienced surgeon influences the learning curve of the surgical team including RAS-inexperienced surgeons. Therefore, this study sought to answer the following questions: (1) What is the influence of a RAS experienced surgeon on the learning curve for RAS TKA and UKA of the surgical team? and (2) Is there an inflection point in the learning curve of the surgical team? It was hypothesized that the presence of an experienced surgeon flattens the learning curve and that there was no inflection point for the learning curve of the surgical team.
Methods
This study was performed at a tertiary arthroplasty referral centre. Ethics board approval was obtained prior to commencement of the study (1146/2021). Consecutive patients with end stage knee osteoarthritis undergoing primary RAS UKA and RAS TKA between June 2021 and October 2021 were included in this study. Indications for RAS UKA were based on the criteria by Hamilton et al. [7], whereas all other patients received a RAS TKA. Age, gender, BMI and ASA were collected. The indications for UKA and TKA remained unchanged with the commencement of the study. The standard implants prior to implementation of RAS were CI (conventionally instrumented) Oxford Phase 3 mobile-bearing UKA (Zimmer Biomet, Warsaw, IN, U.S.) and CI Persona TKA (Zimmer Biomet). Three surgeons were included in the study and have completed the same 2-day clinical application certification: first year post residency surgeon with no prior exposure to CAS (computer assisted surgery) or RAS (Surgeon 1, PP); an 11 years post residency surgeon with no prior exposure to CAS or RAS (Surgeon 2, TG); a dual-fellowship trained surgeon with significant prior exposure to both CAS and RAS (Surgeon 3, AK).
RAS UKA (MAKO Rio, Stryker, Kalamazoo, MI, U.S.) was performed using a standardized work-flow [16] with the application of both the saw and the burr, using proprietary implants (Restoris, Stryker). RAS TKA was performed using a standardized work-flow [16]. Implants (cemented Triathlon, Stryker) were positioned by applying functional alignment strategy [2] aiming to respect the anatomical position of the femur as much as possible. Surgeon 3 routinely resurfaced the patella, Surgeon 2 did not resurface, Surgeon 1 selectively resurfaced.
Surgeons 1 and 3 immediately switched all knee arthroplasty to RAS, whereas surgeon 2 switched to 20% caseload. Surgeons 1 and 3 performed all cases together, surgeon 2 performed 50% of the cases with surgeon 1 and 50% with surgeon 3. Six scrub nurses were involved in the implementation phase, without being controlled for in the present study. A total of 55 cases were performed during the study period, 31 TKAs and 24 UKAs. There were no differences in demographic data between surgeons Table 1.
Outcome measures
The primary outcome measure was surgical time, measured from initial incision until final wound closure. The surgical time has been further broken down into following parts of the procedure: surgical approach, bone referencing, balancing, bone preparation, trialling, final implementation and closure. Secondary outcome measure was the accuracy of implant positioning. Patients in both treatment groups underwent pre- and postoperative anteroposterior knee, lateral knee and full leg-length standing radiographs. Two independent observers (CS and ML) determined the accuracy of implant positioning by comparing the value achieved intraoperatively to the planned value in the corresponding postoperative radiograph. Measurements were performed using MediCad (Medicad Hectec, Altdorf, Germany). Agreement between the observers was investigated by interclass correlation coefficient (ICC). Femoral and tibial axes were used as reference markers to assess accuracy of all positioning and measure alignment in degrees [1], with a tolerance of 1.0°. Coronal [13, 15] and sagittal alignment [5] were measured on AP and lateral views, Fig. 1A–D.
A–D The details for implant positioning are shown for each TKA (A, B) and UKA (C, D) in AP and lateral view x rays. mLDFA mechanical lateral distal femoral angel, mMPTA mechanical medial proximal tibial angle, JLCA joint line conversion angle, FSA-mTA femoral shift axis-mechanical tibial axis, mFA-mTA mechanical femoral axis-mechanical tibial axis
Finally, all patients were followed-up at week 2, 6 and 12 postoperatively by the independent observers for potential complications and adverse events.
Statistical analysis
Normality distribution was analyzed using the Shapiro–Wilk test. Normally distributed continuous data are presented with mean ± standard deviation (SD), non-normally distributed data with median [IQR]. Continuous data were compared using ANOVA and Mann–Whitney U Test, depending on the distribution. CUSUM (cumulative sum) analysis was performed as previously described [12]. Due to the variability of previous experience between surgeons, surgeon 3 data was used as a baseline and comparator for surgical time, but was not used as the target time in the CUSUM analysis. A prospective analysis of consecutive cases has been performed. A post-hoc power analysis with a beta of 0.9 and an alpha of 0.05 revealed 4 cases per surgeon to detect a difference in surgical times for TKA and 6 cases per surgeon to detect a difference for UKA. IBM SPSS statistics v27 (Armonk, NY, U.S.) was used for statistical analysis. Significance was set at p < 0.05.
Results
Surgical time
Only surgeon 2 had longer mean surgical time than surgeons 1 and 3, Table 2.
CUSUM analysis of TKA cases, Fig. 2, demonstrated no inflection point for surgeons 2 and 3 and three peak values in the surgical time learning curve for surgeon 1. A maximum CUSUM value of 35 and a downward trendline for all surgeons was observed.
CUSUM analysis of UKA cases, Fig. 3, demonstrated an inflection point at case 5 for surgeon 1, but only with a maximum CUSUM value of 35.
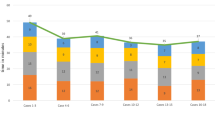
Mean surgical times of TKA and UKA, Figs. 4 and 5, reveal a downward slope from the first case.
Implant positioning
There were no outliers in implant positioning between the intraoperative values and postoperative values obtained from radiographs. ICC for radiographic measurements was 0.94.
Two patients had an adverse event. One female after TKA sustained a fall at week 8, rupturing the patellar tendon and puncturing the skin horizontally, thus exposing the joint. After an unsuccessful DAIR (Debridement, Antibiotics and Implant retention) and patellar tendon reconstruction, a first stage revision with patellar tendon re-reconstruction was performed. There were no issues at follow-up at week 2 and 6. Second patient fell during week 3, opening up her scar proximally. A DAIR has been performed, at 12 weeks after revision surgery, there were no further issues.
Discussion
The most important finding of this study was that the presence of an RAS experienced surgeon flattens the learning curve of an RAS unexperienced surgical team.
Kayani et al. previously demonstrated that six cases were needed until an inflexion point is achieved [12]. In the present study, an inflection point could be observed at 5 cases for UKA and at 9 cases for TKA respectively. There were major differences for the 2 surgeons, with one of the different variables being the presence of the experienced surgeon providing assistance. As Vermue et al. previously noted [21], some surgeon specific variables are difficult to quantify, but a significant variable here was the RAS-experienced surgeon providing assistance. This study nevertheless does demonstrate a clear learning effect reflected in surgical time reduction, but these are lower than in comparative studies [4, 6, 11, 12]. Learning curves are observed in other new technologies, not just RAS. Gharaibeh et al. [6] analyzed intercompartmental pressure differences after TKA between the lateral and the medial compartment using a newly introduced sensor-guided assessment technique for soft tissue balancing. They compared 2 groups of each 45 cases displaying the balancing for the first 45 against the second 45 cases. In the first group 10 cases were identified as unbalanced whereas every case in the second group was well balanced. A number needed to treat of 30 was identified at which the inflection point occurred for the learning curve for proper balancing with this technique whereas no effect was displayed for the surgical time [6]. The described effect of improvement comparing starting cases to end stage cases is also known from other works [14]. Even longer learning curve effects compared to these findings have been observed. Vermue et al. [21] report up to 35 and 43 cases until an inflection point was reached. The authors believe that inconsistencies in the surgical team are responsible for these differences [21]. This effect was not found in the present study, although a total of 6 scrub nurses were included. No inflection points occurred in the analysis of the team performance. This demonstrates the beneficiary effect of the experienced surgeon´s presence on the overall team performance. The importance of the presence of an experienced surgeon is also underlined by the findings of Vermue et al. [21]. Although consistency of the surgical team was not assured for the scrub nurse in our setting, the described negative effect on the surgical time performance was not observed in the present study [21]. Thus, the presence of an experienced surgeon can neutralize a potential negative effect of team inconsistency on the surgical time performance. Interestingly, the downward slope for RAS UKA was flatter than for RAS TKA. This might be connected to the fact that one of the 2 RAS-unexperienced surgeons has had significant experience in classic jig-based UKA. Similar effect was observed by Kayani et al. since different pre-existing grades of experience for classic jig-based KA may lead to different learning curves for RAS KA [12, 20]. Similarly, Vermue et al. indicate that different learning curves depend on the individual, pre-existing surgical experience in classic jig-based TKA [21]. No outliers in alignment were observed in this study, which corresponds to previous results of both RAS-TKA and RAS-UKA [4, 12].
The primary limitation is a small sample size of 55 cases, divided between 3 surgeons, as well as the absence of a STAI score evaluation. The results, however, demonstrate the investigated effect even with this sample size. The individual surgical characteristics, such as previous surgical experience including number of cases performed have not been quantified. As pointed out by Vermue et al., in a study with six surgeons, quantification of all potential variables is almost impossible and is therefore disregarded in the present study. One surgeon only performed 5 UKA, which is not enough to assess the learning curve, which might take as long as 40 cases, as per Vermue, however, the initial times are significantly longer than for the other inexperienced surgeon that always had the experienced surgeon as the assistant. Patella resurfacing philosophy between surgeons in the present study has been different, however, the changes in time would not alter the results observed, since there is no learning curve for the conventional patella and the less experienced RAS surgeons either did not use the patella or only occasionally used the patella. Furthermore, no RAS currently offers a patella application, which could be assessed as a part of the learning curve. The follow-up for a clinical study is short, 90 days, but is longer than in comparable studies. Another limitation finally is found in the circumstances set by the COVID-19 pandemic probably inherent to every elective surgical discipline: Reduced elective operative capacity and surgical cases may not only have led to a significantly reduced case load but also to a disturbance of the development of the learning curve for RAS KA in this case series. Finally, a control group is missing in this study so that the learning curve behaviour can only be described in the manner of tendencies, although a true control group would almost be impossible because each surgeon can only undergo one learning curve, in this case 1 surgeon with assistance of an experienced surgeon and 1 surgeon with occasional assistance, with different results. Patient reported outcomes are not reported, since the study only has a short follow-up and clinical outcomes are not the primary outcome of the study. Lack of adverse events and radiographic evidence of excellent implant position serve as a proxy for outcomes.
Conclusion
The presence of an experienced surgeon in robotically assisted knee arthroplasty can flatten learning curve of the surgical team formerly unexperienced in robotic assisted systems. Manufacturers should provide expanded support during initial cases in centres without previous experience to robotic assisted knee arthroplasty.
Abbreviations
- RAS KA:
-
Robotic assisted knee arthroplasty
- TKA:
-
Total knee arthroplasties
- UKA:
-
Unicompartmental arthroplasties
- CUSUM:
-
Cumulative sum analysis
- THA:
-
Total hip arthroplasty
- CAS:
-
Computer-assisted surgery
- CI:
-
Conventionally instrumented
- ICC:
-
Interclass correlation coefficient
- SD:
-
Standard deviation
- IQR:
-
Interquartile range
References
Bell SW, Anthony I, Jones B, MacLean A, Rowe P, Blyth M (2016) Improved accuracy of component positioning with robotic-assisted unicompartmental knee arthroplasty: data from a prospective, randomized controlled study. J Bone Jt Surg Am 98:627–635
Chang JS, Kayani B, Wallace C, Haddad FS (2021) Functional alignment achieves soft-tissue balance in total knee arthroplasty as measured with quantitative sensor-guided technology. Bone Joint J 103:507–514
Chinnappa J, Chen DB, Harris IA, MacDessi SJ (2015) Total knee arthroplasty using patient-specific guides: is there a learning curve? Knee 22:613–617
Clement ND, Al-Zibari M, Afzal I, Deehan DJ, Kader D (2020) A systematic review of imageless hand-held robotic-assisted knee arthroplasty: learning curve, accuracy, functional outcome and survivorship. EFORT Open Rev 5:319–326
Gaudiani MA, Nwachukwu BU, Baviskar JV, Sharma M, Ranawat AS (2017) Optimization of sagittal and coronal planes with robotic-assisted unicompartmental knee arthroplasty. Knee 24:837–843
Gharaibeh MA, Chen DB, MacDessi SJ (2018) Soft tissue balancing in total knee arthroplasty using sensor-guided assessment: is there a learning curve? ANZ J Surg 88:497–501
Hamilton TW, Pandit HG, Lombardi AV, Adams JB, Oosthuizen CR, Clavé A et al (2016) Radiological decision aid to determine suitability for medial unicompartmental knee arthroplasty: development and preliminary validation. Bone Joint J 98:3–10
Hampp EL, Chughtai M, Scholl LY, Sodhi N, Bhowmik-Stoker M, Jacofsky DJ et al (2019) Robotic-arm assisted total knee arthroplasty demonstrated greater accuracy and precision to plan compared with manual techniques. J Knee Surg 32:239–250
Hopper AN, Jamison MH, Lewis WG (2007) Learning curves in surgical practice. Postgrad Med J 83:777–779
Kayani B, Haddad FS (2019) Robotic unicompartmental knee arthroplasty: current challenges and future perspectives. Bone Joint Res 8:228–231
Kayani B, Konan S, Huq SS, Ibrahim MS, Ayuob A, Haddad FS (2021) The learning curve of robotic-arm assisted acetabular cup positioning during total hip arthroplasty. Hip Int 31:311–319
Kayani B, Konan S, Pietrzak JRT, Huq SS, Tahmassebi J, Haddad FS (2018) The learning curve associated with robotic-arm assisted unicompartmental knee arthroplasty: a prospective cohort study. Bone Joint J 100:1033–1042
Koh IJ, Kim JH, Jang SW, Kim MS, Kim C, In Y (2016) Are the Oxford(®) medial unicompartmental knee arthroplasty new instruments reducing the bearing dislocation risk while improving components relationships? A case control study. Orthop Traumatol Surg Res 102:183–187
Mahure SA, Teo GM, Kissin YD, Stulberg BN, Kreuzer S, Long WJ (2021) Learning curve for active robotic total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-021-06452-8
Moon YW, Ha CW, Do KH, Kim CY, Han JH, Na SE et al (2012) Comparison of robot-assisted and conventional total knee arthroplasty: a controlled cadaver study using multiparameter quantitative three-dimensional CT assessment of alignment. Comput Aided Surg 17:86–95
Roche M (2021) The MAKO robotic-arm knee arthroplasty system. Arch Orthop Trauma Surg 141:2043–2047
Tay ML, Carter M, Bolam SM, Zeng N, Young SW (2022) Robotic-arm assisted unicompartmental knee arthroplasty system has a learning curve of 11 cases and increased operating time. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-021-06814-2
Thiengwittayaporn S, Uthaitas P, Senwiruch C, Hongku N, Tunyasuwanakul R (2021) Imageless robotic-assisted total knee arthroplasty accurately restores the radiological alignment with a short learning curve: a randomized controlled trial. Int Orthop. https://doi.org/10.1007/s00264-021-05179-y
Thiengwittayaporn S, Uthaitas P, Senwiruch C, Hongku N, Tunyasuwanakul R (2021) Imageless robotic-assisted total knee arthroplasty accurately restores the radiological alignment with a short learning curve: a randomized controlled trial. Int Orthop 45:2851–2858
Vermue H, Lambrechts J, Tampere T, Arnout N, Auvinet E, Victor J (2020) How should we evaluate robotics in the operating theatre? Bone Joint J 102:407–413
Vermue H, Luyckx T, Winnock de Grave P, Ryckaert A, Cools AS, Himpe N, et al. (2020) Robot-assisted total knee arthroplasty is associated with a learning curve for surgical time but not for component alignment, limb alignment and gap balancing. Knee Surg Sports Traumatol Arthrosc
Zhang J, Ndou WS, Ng N, Gaston P, Simpson PM, Macpherson GJ et al (2021) Robotic-arm assisted total knee arthroplasty is associated with improved accuracy and patient reported outcomes: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-021-06464-4
Funding
Open access funding provided by Johannes Kepler University Linz. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
CS: wrote the manuscript, acquisition of data, interpretation of the data. PP: revised the manuscript, interpretation of the data. ML: revised the manuscript, interpretation of the data. TG: revised the manuscript. AK: conceived the study, edited the manuscript, interpretation of the data, performed the statistical analysis, acquisition of data.
Corresponding author
Ethics declarations
Conflict of interest
We report personal fees paid to our institution during the conduct of the study from Zimmer Biomet, Europe and from Depuy Synthes Orthopädie Gmbh, Peter Brehm Gmbh outside the submitted work. Additionally, one co-author (T.G) received personal fees as a consultant for ZimmerBiomet outside the submitted work. All other co-authors certify that there are no funding or commercial associations (consultancies, stock ownership, equity interest, patent/licensing arrangements, etc.) that might pose a conflict of interest in connection with the submitted article related to the author or any immediate family members.
Ethical approval
This study received ethical approval from the local institutional review board (EK-No.: 1146/2021) of the “Ethikkommission JKU Linz” of the Johannes Kepler University Linz (JKU Linz).
Informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained by all participating patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schopper, C., Proier, P., Luger, M. et al. The learning curve in robotic assisted knee arthroplasty is flattened by the presence of a surgeon experienced with robotic assisted surgery. Knee Surg Sports Traumatol Arthrosc 31, 760–767 (2023). https://doi.org/10.1007/s00167-022-07048-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-022-07048-6