Abstract
Purpose
Content validity is the most important property of PROMs. The COSMIN initiative has published guidelines for evaluating the content validity of PROMs, but they have only sparsely been applied to relevant PROMs for musculoskeletal conditions. The aim of this study was to use the COSMIN Risk of Bias checklist to evaluate the content validity of five PROMs, that are highly relevant in musculoskeletal research and used by the arthroscopic surgery community: the modified Harris’ Hip Score (mHHS), the Copenhagen Hip and Groin Outcome Score (HAGOS), the International Knee Documentation Committee Subjective Knee evaluation Form (IKDC-SKF), the Knee injury and Osteoarthritis Outcome Score (KOOS) and the Knee Numeric-Entity Evaluation Score ACL (KNEES-ACL).
Methods
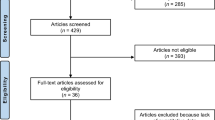
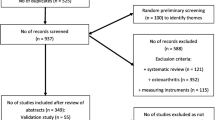
The development articles for the five PROMs were identified through searches in PubMed and SCOPUS. A literature search was performed to identify additional studies assessing content validity of the PROMs. Additional information, necessary for the assessments, was obtained from the PROM developers after direct request. To evaluate the quality of the development studies and rate the content validity, the COSMIN Risk of Bias checklist was applied to all studies.
Results
All five development studies were identified. Three subsequent content validity studies were identified, all evaluating KOOS and one also IKDC. One content validity study was of inadequate quality and excluded from further analysis. The development of mHHS, IKDC-SKF, and KOOS was rated inadequate and possess insufficient content validity for their target populations. Due to the irrelevance of multiple items, KOOS was in particular inappropriate to evaluate patients with an ACL injury. The development of HAGOS was also rated inadequate, although the insufficiency aspects can be regarded as minor. KNEES-ACL possessed sufficient content validity.
Conclusion
Out of five PROMs, only KNEES-ACL possessed sufficient content validity. Particularly, KOOS should not be used as an outcome for patients with an ACL injury. There is an urgent need for condition-specific PROMs for musculoskeletal conditions, developed with adequate methods.
Level of evidence
III.

Similar content being viewed by others
Abbreviations
- ACL:
-
Anterior cruciate ligament
- ADL:
-
Activities of Daily Living
- COSMIN:
-
COnsensus-based Standards for the selection of health Measurement Instruments
- FAIS:
-
Femoroacetabular impingement syndrome
- FAOS:
-
Foot and Ankle Outcome Score
- HAGOS:
-
Copenhagen Hip and Groin Outcome Score
- HOOS:
-
Hip disability and Osteoarthritis Outcome Score
- IKDC-SKF:
-
International Knee Documentation Committee Subjective Knee Evaluation Form
- KNEES-ACL:
-
Knee Numeric-Entity Evaluation Score ACL
- KOOS:
-
Knee injury and Osteoarthritis Outcome Score
- mHHS:
-
Modified Harris’ Hip Score
- PROMs:
-
Patient reported outcome measures
- SEFAS:
-
Self-reported Foot and Ankle Score
- WOMAC:
-
Western Ontario and McMaster Universities Osteoarthritis Index
References
Bellamy N, Buchanan WW (1986) A preliminary evaluation of the dimensionality and clinical importance of pain and disability in osteoarthritis of the hip and knee. Clin Rheumatol 5:231–241
Brodersen JDL, Thorsen H, McKenna S (2013) Writing health-related items for Rasch models—patient-reported outcome scales for health sciences: from medical paternalism to patient autonomy. Rasch Models Health. https://doi.org/10.1002/9781118574454.ch15
Byrd JW, Jones KS (2000) Prospective analysis of hip arthroscopy with 2-year follow-up. Arthroscopy 16:578–587
Collins NJ, Prinsen CA, Christensen R, Bartels EM, Terwee CB, Roos EM (2016) Knee Injury and Osteoarthritis Outcome Score (KOOS): systematic review and meta-analysis of measurement properties. Osteoarthritis Cartilage 24:1317–1329
Comins JD, Brodersen J, Siersma V, Jensen J, Hansen CF, Krogsgaard MR (2021) How to develop a condition-specific PROM. Scand J Med Sci Sports 31:1216–1224
Comins JD, Krogsgaard MR, Brodersen J (2013) Development of the Knee Numeric-Entity Evaluation Score (KNEES-ACL): a condition-specific questionnaire. Scand J Med Sci Sports 23:e293-301
Comins JD, Siersma VD, Lind M, Jakobsen BW, Krogsgaard MR (2018) KNEES-ACL has superior responsiveness compared to the most commonly used patient-reported outcome measures for anterior cruciate ligament injury. Knee Surg Sports Traumatol Arthrosc 26:2438–2446
Engelhart L, Nelson L, Lewis S, Mordin M, Demuro-Mercon C, Uddin S et al (2012) Validation of the Knee Injury and Osteoarthritis Outcome Score subscales for patients with articular cartilage lesions of the knee. Am J Sports Med 40:2264–2272
Gagnier JJ, Shen Y, Huang H (2018) Psychometric properties of patient-reported outcome measures for use in patients with anterior cruciate ligament injuries: a systematic review. JBJS Rev 6:e5
Grevnerts HT, Terwee CB, Kvist J (2015) The measurement properties of the IKDC-subjective knee form. Knee Surg Sports Traumatol Arthrosc 23:3698–3706
Hansen CF, Jensen J, Brodersen J, Siersma V, Comins JD, Krogsgaard MR (2021) Are adequate PROMs used as outcomes in randomized controlled trials? an analysis of 54 trials. Scand J Med Sci Sports 31:972–981
Hansen CF, Jensen J, Siersma V, Brodersen J, Comins JD, Krogsgaard MR (2021) A catalogue of PROMs in sports science—Quality assessment of PROM development and validation. Article 7 in a series of 10. Scand J Med Sci Sports. https://doi.org/10.1111/sms.13923
Huang H, Nagao M, Arita H, Shiozawa J, Nishio H, Kobayashi Y et al (2019) Reproducibility, responsiveness and validation of the Tampa Scale for Kinesiophobia in patients with ACL injuries. Health Qual Life Outcomes 17:150
Hung M, Hon SD, Cheng C, Franklin JD, Aoki SK, Anderson MB et al (2014) Psychometric evaluation of the lower extremity computerized adaptive test, the modified harris hip score, and the hip outcome score. Orthop J Sports Med 2:2325967114562191
Impellizzeri FM, Jones DM, Griffin D, Harris-Hayes M, Thorborg K, Crossley KM et al (2020) Patient-reported outcome measures for hip-related pain: a review of the available evidence and a consensus statement from the International Hip-related Pain Research Network, Zurich 2018. Br J Sports Med 54:848–857
Irrgang JJ, Anderson AF, Boland AL, Harner CD, Kurosaka M, Neyret P et al (2001) Development and validation of the international knee documentation committee subjective knee form. Am J Sports Med 29:600–613
Jacobs CA, Peabody MR, Lattermann C, Vega JF, Huston LJ, Spindler KP et al (2018) Development of the KOOSglobal platform to measure patient-reported outcomes after anterior cruciate ligament reconstruction. Am J Sports Med 46:2915–2921
Kemp JL, Collins NJ, Roos EM, Crossley KM (2013) Psychometric properties of patient-reported outcome measures for hip arthroscopic surgery. Am J Sports Med 41:2065–2073
Mokkink LB, de Vet HCW, Prinsen CAC, Patrick DL, Alonso J, Bouter LM et al (2018) COSMIN Risk of Bias checklist for systematic reviews of Patient-Reported Outcome Measures. Qual Life Res 27:1171–1179
Mokkink LB, Terwee CB, Knol DL, Stratford PW, Alonso J, Patrick DL et al (2010) The COSMIN checklist for evaluating the methodological quality of studies on measurement properties: a clarification of its content. BMC Med Res Methodol 10:22
Mokkink LB, Terwee CB, Patrick DL, Alonso J, Stratford PW, Knol DL et al (2010) The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J Clin Epidemiol 63:737–745
Moller A, Bissenbakker KH, Arreskov AB, Brodersen J (2020) Specific measures of quality of life in patients with multimorbidity in primary healthcare: a systematic review on patient-reported outcome measures’ adequacy of measurement. Patient Relat Outcome Meas 11:1–10
Ohlin A, Ahlden M, Lindman I, Jonasson P, Desai N, Baranto A et al (2020) Good 5-year outcomes after arthroscopic treatment for femoroacetabular impingement syndrome. Knee Surg Sports Traumatol Arthrosc 28:1311–1316
Patrick DL, Burke LB, Gwaltney CJ, Leidy NK, Martin ML, Molsen E et al (2011) Content validity–establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO good research practices task force report: part 1–eliciting concepts for a new PRO instrument. Value Health 14:967–977
Patrick DL, Burke LB, Gwaltney CJ, Leidy NK, Martin ML, Molsen E et al (2011) Content validity–establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO Good Research Practices Task Force report: part 2–assessing respondent understanding. Value Health 14:978–988
Patrick DL, Burke LB, Powers JH, Scott JA, Rock EP, Dawisha S et al (2007) Patient-reported outcomes to support medical product labeling claims: FDA perspective. Value Health 10(Suppl 2):S125-137
Pellekooren S, Ostelo R, Pool A, van Tulder M, Jansma E, Chiarotto A (2021) Content validity of patient-reported outcome measures of satisfaction with primary care for musculoskeletal complaints: a systematic review. J Orthop Sports Phys Ther 51:94–102
Prinsen CAC, Mokkink LB, Bouter LM, Alonso J, Patrick DL, de Vet HCW et al (2018) COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual Life Res 27:1147–1157
Roos EM, Roos HP, Lohmander LS, Ekdahl C, Beynnon BD (1998) Knee Injury and Osteoarthritis Outcome Score (KOOS)–development of a self-administered outcome measure. J Orthop Sports Phys Ther 28:88–96
Roos EM, Toksvig-Larsen S (2003) Knee injury and Osteoarthritis Outcome Score (KOOS)—validation and comparison to the WOMAC in total knee replacement. Health Qual Life Outcomes 1:17
Streiner DL, Norman GR, Cairney J (2015) Selecting the items. Health Measurement Scales—A practical guide to their development and use. Fifth edition. 5:74–99
Terwee CB, Jansma EP, Riphagen II, de Vet HC (2009) Development of a methodological PubMed search filter for finding studies on measurement properties of measurement instruments. Qual Life Res 18:1115–1123
Terwee CB, Prinsen CAC, Chiarotto A, Westerman MJ, Patrick DL, Alonso J et al (2018) COSMIN methodology for evaluating the content validity of patient-reported outcome measures: a Delphi study. Qual Life Res 27:1159–1170
Thorborg K, Holmich P, Christensen R, Petersen J, Roos EM (2011) The Copenhagen Hip and Groin Outcome Score (HAGOS): development and validation according to the COSMIN checklist. Br J Sports Med 45:478–491
van der Zwaard BC, Terwee CB, Roddy E, Terluin B, van der Horst HE, Elders PJ (2014) Evaluation of the measurement properties of the Manchester foot pain and disability index. BMC Musculoskelet Disord 15:276
van Meer BL, Meuffels DE, Vissers MM, Bierma-Zeinstra SM, Verhaar JA, Terwee CB et al (2013) Knee injury and Osteoarthritis Outcome Score or International Knee Documentation Committee Subjective Knee Form: which questionnaire is most useful to monitor patients with an anterior cruciate ligament rupture in the short term? Arthroscopy 29:701–715
Wiering B, de Boer D, Delnoij D (2017) Patient involvement in the development of patient-reported outcome measures: a scoping review. Health Expect 20:11–23
Funding
No particular funding was provided for this research.
Author information
Authors and Affiliations
Contributions
CFH, JJ, VS, JB, JDC and MRK conceived and planned the project. AO was invited as an external assessor. CFH and JJ collected data for the project, and CFH, AO and VS completed data analyses. All authors participated in writing the manuscript. CFH and MRK directed the project.
Corresponding author
Ethics declarations
Conflict of interest
One author (JDC) is the main developer of the KNEES-ACL, and two authors (MRK and JB) participated in the development of KNEES-ACL. These three authors did not participate in the assessment of the PROMs or the discussions related to consensus in the assessments. There are no financial conflicts.
Ethical approval
No approval required.
Informed consent
Not relevant.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
167_2021_6761_MOESM1_ESM.pdf
Supplementary file1 Additional file 1 Presents the correspondences with the original PROM developers regarding data that could not be obtained directly from the published development studies (PDF 110 KB)
167_2021_6761_MOESM2_ESM.docx
Supplementary file2 Additional file 2 Presents a more detailed description of the domains and scoring system of the COSMIN Risk of Bias checklist (DOCX 22 KB)
Rights and permissions
About this article
Cite this article
Hansen, C.F., Jensen, J., Odgaard, A. et al. Four of five frequently used orthopedic PROMs possess inadequate content validity: a COSMIN evaluation of the mHHS, HAGOS, IKDC-SKF, KOOS and KNEES-ACL. Knee Surg Sports Traumatol Arthrosc 30, 3602–3615 (2022). https://doi.org/10.1007/s00167-021-06761-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-021-06761-y