Abstract
Purpose
Frailty is common in critically ill patients but the timing and optimal method of frailty ascertainment, trajectory and relationship with care processes remain uncertain. We sought to elucidate the trajectory and care processes of frailty in critically ill patients as measured by the Clinical Frailty Scale (CFS) and Frailty Index (FI).
Methods
This is a multi-centre prospective cohort study enrolling patients ≥ 50 years old receiving life support > 24 h. Frailty severity was assessed with a CFS, and a FI based on the elements of a comprehensive geriatric assessment (CGA) at intensive care unit (ICU) admission, hospital discharge and 6 months. For the primary outcome of frailty prevalence, it was a priori dichotomously defined as a CFS ≥ 5 or FI ≥ 0.2. Processes of care, adverse events were collected during ICU and ward stays while outcomes were determined for ICU, hospital, and 6 months.
Results
In 687 patients, whose age (mean ± standard deviation) was 68.8 ± 9.2 years, frailty prevalence was higher when measured with the FI (CFS, FI %): ICU admission (29.8, 44.8), hospital discharge (54.6, 67.9), 6 months (34.1, 42.6). Compared to ICU admission, aggregate frailty severity increased to hospital discharge but improved by 6 months; individually, CFS and FI were higher in 45.3% and 50.6% patients, respectively at 6 months. Compared to hospital discharge, 18.7% (CFS) and 20% (FI) were higher at 6 months. Mortality was higher in frail patients. Processes of care and adverse events were similar except for worse ICU/ward mobility and more frequent delirium in frail patients.
Conclusions
Frailty severity was dynamic, can be measured during recovery from critical illness using the CFS and FI which were both associated with worse outcomes. Although the CFS is a global measure, a CGA FI based may have advantages of being able to measure frailty levels, identify deficits, and potential targets for intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Both the Clinical Frailty Scale and Frailty Index can be used for frailty ascertainment on admission and during recovery from critical illness although the Frailty Index identifies more patients as frail. | |
Frailty is associated with severity dependent mortality increases using both the Clinical Frailty Scale and Frailty Index. | |
Processes of care were similar between frail and non-frail patients with the exception of decreased mobility and more frequent delirium in frail patients. | |
In survivors, frailty during recovery is dynamic with many patients transitioning to increased frailty states. Further research on how to improve the recovery of critically ill frail patients is required. |
Introduction
As the population ages, a greater number of persons are living with functional limitation and frailty [1]. Frailty is a state of increased vulnerability resulting from a decline in reserve across multiple physiological systems, increasing susceptibility to adverse events [2]. Vulnerable older individuals are overrepresented among critically ill patients whereby 30–40% of individuals admitted to intensive care units (ICUs) are frail [3, 4]. Frailty is associated with worse outcomes from critical illness both during the index hospitalization and over long-term follow-up [5]. These negative outcomes include longer hospital stay, higher mortality, reduced health related quality of life (QoL), risk of disability and admission to long-term care [5, 6]. There are significant knowledge gaps regarding frailty in critically ill patients including the optimal timing and method of frailty assessment, the impact of ICU care processes on the course of frailty, and the interplay between critical illness and the trajectory of frailty.
Several tools have been developed and validated for frailty assessment [7]. In the ICU, the clinical frailty scale (CFS) [8] is the most commonly used [9], likely due to its simplicity and ease of use. Although the CFS was initially validated in community dwelling patients older than 65, it is commonly used in those over the age of 50 in critically ill populations [10, 11]. The CFS was designed as a screening tool and should prompt further assessment such as a comprehensive geriatric assessment (CGA), the reference standard for frailty identification and assessment [12]. Deficit models of frailty quantified by the Frailty Index (FI) [13] based on CGA elements may have the advantage of informing on the individual factors contributing to frailty. There are few studies on the FI in critically ill and only one study has reported on an ICU admission FI based on a modified CGA [14]. In addition, the optimal timing of frailty assessment in critically ill patients is uncertain, with the vast majority of studies screening for frailty upon ICU admission [5]. Moreover, it is unclear how critical illness and attendant processes of care modify the degree and trajectory of frailty.
To begin addressing these knowledge gaps, we conducted a multicentre prospective observational study of critically ill patients ≥ 50 years old, measuring frailty using the CFS and CGA based FI at ICU admission, hospital discharge and at 6-month follow-up. Our objectives were to: (1) ascertain the prevalence of frailty using the CFS and FI at these time points; (2) determine the trajectory of frailty as measured with CFS or FI. (3) outline processes of care; (4) determine the association with patient outcomes with frailty as measured with CFS or FI. Findings from this multi-centre study will provide a framework for developing interventional studies designed to improve outcomes in critically ill frail patients. A portion of this work was presented as an abstract to the international meeting of the European Society of Intensive Care Medicine in October 2023 [15].
Methods
Frailty, Outcomes, Recovery and Care Steps of Critically Ill Patients (FORECAST) was a prospective multi-centre observational study conducted in 14 tertiary medical-surgical ICUs across Canada. We enrolled patients between September 2019 and November 2021 with final patient follow-up completed in June 2022. The study protocol was approved by Queen’s University Health Sciences Research Ethics Board (REB), Clinical Trials Ontario (CTO 1750) and the REBs at each participating site. Consent was obtained from the patient or substitute decision maker (SDM) prior to any data collection. The study was registered at clinicaltrials.gov (NCT05509036), is reported as per STROBE guidelines for observational studies [16] (see electronic supplementary material, ESM) and we indicate study modifications in response to the coronavirus disease 2019 (COVID-19) pandemic in the manuscript [17].
We included critically ill patients at least 50 years old receiving one or more of the following life support interventions for greater than 24 h; (1) invasive mechanical ventilation (IMV) or non-invasive ventilation (NIV); high flow oxygen was acceptable if the fraction of inspired oxygen (FiO2) was greater than 0.5 after 24 h; (2) receipt of intravenous vasopressors or inotropes; (3) acute renal replacement therapy. Patients were excluded for the following: need for life support interventions < 24 h; were > 5 days after ICU admission; had no family or caregivers to provide collateral history; patient or caregivers not proficient in English or French; projected inability to complete 6-month follow-up by telephone or in-person; or expected to survive < 72 h.
Data collection
After ICU admission, defined as when the ICU clinical team commenced care of the patient, trained research coordinators collected baseline demographics including age, sex, Charlson Comorbidity Index [18], acute physiology and chronic health evaluation (APACHE) II [19], Katz Index of Activities of Daily Living (ADL)[20], Palliative Performance Scale (PPS) [21], the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) [22] and health-related quality of life (HRQoL) with the EQ5D5L instrument [23]. During the ICU and hospital ward stay processes of care and adverse events were collected by chart review. Severity of illness for the first 24 h of ICU stay was quantified with APACHE II and during the ICU stay with Sequential Organ Failure Assessment (SOFA) [24]. ICU processes of care included nutritional support and adequacy defined as the proportion of prescribed calories and protein received on ICU days receiving enteral feeding and prior to discharge or death, sedative and analgesic medications received, mobility levels achieved using the ICU Mobility Scale [25] and involvement of physiotherapy and/or occupational therapy. Adverse events during the ICU stay included data on possible nosocomial infection including positive cultures and antibiotic-free days and development of delirium. Delirium was abstracted from that recorded as part of usual care with the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU)[26] or the Intensive Care Delirium Screening Checklist (ICDSC) [27] dependent on institution. During the hospital ward stay, data on sedative and analgesic medications received, mobility as defined by the highest observed level of activity observed on random direct observation by research coordinators on ward visits [28], and delirium occurrence as measured with CAM [29] were collected. Six-month outcomes were assessed through in-person evaluation or by telephone; due to the COVID-19 pandemic, in-person follow-up was terminated in March 2020. Outcomes collected at 6 months included vital status, Katz ADL, PPS, hospitalizations, outpatient clinic visits, health-related quality of life (EQ-5D-5L) and place of residence.
Frailty
Frailty was assessed on ICU admission, hospital discharge and at 6 months post discharge using the CFS (ESM, eFigure 1) and FI consisting of 42 potential deficits based on the elements of a CGA [14] (ESM, eTables 1 and 2). At all-time points, frailty was assessed directly with the participant, the SDM or both depending on participants’ ability. The frailty assessment was conducted by trained research coordinators using a standardized questionnaire. Where possible both SDM and participant were utilized to obtain the most rigorous data possible. The baseline CFS was anchored to 2 weeks prior to admission [30] and collected either in-person where possible or by a telephone interview [31]. Data collection tools were developed in a pilot study [32].
Outcomes and statistical analysis
For the primary outcome of frailty prevalence, frailty was a priori defined as CFS ≥ 5 and for the FI ≥ 0.2 [33, 34]. The FI was calculated as a proportion of deficits identified divided by total number of potential deficits measured as previously described [35]. For missing FI constituent items, any patients with 20% or more missing values had the FI set to missing [36]. The relationship between the CFS and FI at ICU admission, hospital discharge and at 6 months was assessed via Pearson’s correlation coefficient. To determine the impact of frailty on outcomes, outcomes were compared between those identified as frail using the CFS and FI (CFS ≥ 5 and FI ≥ 0.20) separately, and those not identified as frail (CFS < 5 and FI < 0.2). Survival status over time according to frailty status group is presented via Kaplan–Meier curves. Aggregate frailty levels after ICU admission only include survivors with no correction for death. Changes in frailty level between time points amongst survivors were assessed via paired t-tests. Alluvial plots were constructed as per Brummel et al. to outline the trajectory of frailty [37]. Ventilator free days and antibiotic free days were defined as the number of days alive and not on IMV or on antibiotics in the 28 days after ICU admission with 28-day decedents assigned 0 free days [38]. Multivariable logistic regression models with 6-month mortality as the dependent variable and a priori selected predictors of interest of ICU admission, CFS ≥ 5 or FI ≥ 0.2 or hospital discharge CFS ≥ 5 or FI ≥ 0.2 as predictors of interest while adjusting for a priori cofounders of age, sex, admission severity of illness (APACHE II) and Charlson co-morbidity were used to provide adjusted odds ratios comparing mortality by frailty status.
Continuously measured variables are reported as mean ± standard deviation (SD) for normally distributed data, and median with interquartile range (IQR) for skewed data. Continuous variables were compared between groups via the Wilcoxon rank-sum test. Categorical variables are reported as counts and percentages and compared between groups via a Chi-squared test. For tests of statistical significance, a p value less than 0.05 was considered significant without adjustment for multicity due to the exploratory nature of this study. All analyses are complete case and imputation for missing values was not performed. Data analysis was conducted using SAS Version 9.4 (SAS Institute Inc., Cary, NC, USA) and the alluvial plots were created with R (version 4.2.2, ggalluvial package).
Sample size
In our pilot study, 30–40% of critically ill patients were classified as frail on ICU admission and approximately 50–60% were classified as frail at hospital discharge by the FI or CFS, respectively; 6-month mortality was 35% [32]. Considering both mortality and follow-up rates, we estimated that 55–65% of the final cohort would provide data on long-term frailty outcomes. Using an event-driven sample size and to allow for the ability to include sufficient baseline variable in multivariable models, we estimated that we would need to enrol 630 patients. This was later increased to 700 to account for potential increased loss to follow-up during the COVID-19 pandemic.
Results
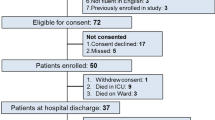
A total of 700 patients were enrolled and data from 687 patients was available for analysis (CONSORT diagram, Fig. 1). For the overall cohort, the mean (SD) age was 68.8 (9.2) years, 293 (43%) were female, APACHE II score was 23.3 (8.1), and the Charlson Co-morbidity Index was 1.9 (2.1). Receipt of organ support interventions on ICU admission included mechanical ventilation (both IMV and NIV) in 618 (90%), vasoactive medications in 506 (74%) and acute renal replacement therapy in 35 (5%).
Frailty prevalence
Frailty at ICU admission, hospital discharge and at 6 months was more common when assessed with the FI vs. the CFS at all three time points: ICU admission—307 (44.8%) vs. 205 (29.8%), p < 0.0001; hospital discharge—303 (67.9%) vs. 246 (54.6%), p < 0.0001; 6 months—155 (42.6%) vs. 125 (34.1%), p = < 0.0001, respectively. The Pearson correlation (95% confidence interval [CI], p-value) between CFS and FI was 0.79 (0.76, 0.82), p < 0.0001) at ICU admission, 0.7 (0.65, 0.75, p < 0.0001) at hospital discharge, and 0.79 (0.75, 0.83, p < 0.0001) at 6 months. The characteristics of patients according to ICU admission frailty levels are outlined in supplementary eTables 3 and 4 (CFS and FI). Frail patients (as measured by CFS or FI) were older, had higher severity of illness, higher number of co-morbidities, lower QoL, reduced ADLs and reduced function as measured by the PPS compared to those non-frail. Frailty prevalence by age decade did not vary significantly in those under 80 but rose in those over the age of 80 where 42/86 (48.8%) had CFS > 5 and 53/85 (62%) had a FI > 0.2 (ESM, eTable 5). Sensitivity analysis of different frailty thresholds for the CFS and FI are reported in supplementary Material eTable 6.
Frailty trajectory
From ICU admission, aggregate frailty levels in survivors increased at hospital discharge and then decreased at 6 months (ESM, eFigure 2). Among patients remaining alive, the proportion of those with a CFS ≥ 5 rose from 205 (29.8%) on ICU admission to 246 (54.6%) at hospital discharge and decreased to 125 (34.1%) at 6 months. The proportion of patients’ frail as per an FI ≥ 0.2 rose from 307 (44.8%) at ICU admission to 303 (67.9%) at hospital discharge and decreased to 155 (42.6%) at 6 months. In survivors, from hospital discharge to 6 months post discharge, CFS levels remained similar or worsened in 89 (26.6) and 63 (18.9%) respectively while the FI remained similar or worsened in 37 (11.1%) and 67 (20.1%) respectively. Frailty levels were significantly higher (p < 0.001) than ICU admission at hospital discharge (CFS n = 451, FI n = 445) and at 6-month follow-up (CFS n = 337, FI n = 333) but frailty levels at 6 months compared to hospital discharge were significantly lower (p < 0.001). Figure 2 shows frailty evolution over time with alluvial plots; among those with frailty at baseline, the majority were still frail or dead at hospital discharge and at 6 months with relatively few improving or transitioning between frail to non-frail by either definition (Table 1).
Processes of care
Processes of care during the ICU stay are outlined in Table 2. Whether frailty was measured with the CFS or FI, frail patients were more likely to have positive cultures on ICU admission, had higher SOFA scores during the ICU stay, had lower antibiotic free days and had lower levels of ICU mobility (ESM, eFigure 3). ICU delirium was more common in patients with frailty ascertained by the FI than when measured by the CFS. On the ward, frail patients (both by CFS and FI) were more likely to be delirious and less likely to be ambulatory on direct observation (ESM, eTable 7). Increases in limitations of life-sustaining treatment during the hospital stay were more common in frail patients vs. non frail patients respectively by CFS: 49 (24%) vs. 71 (15%), p = 0.004 or by FI: 72 (23%) vs. 47 (12%), p < 0.001.
Outcomes
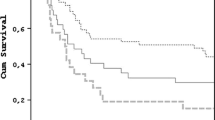
Patient outcomes by ICU admission frailty status and hospital discharge frailty status are outlined in Tables 3 and 4, respectively. Both the CFS and FI as measures of frailty were associated with worse outcomes. Mortality was higher for frail patients vs. non-frail patients both by admission CFS and FI at hospital discharge [72 (35%) vs. 92 (19%), p < 0.001 and 97 (32%) vs. 66 (18%), p < 0.001] and at 6-months [105 (51%) vs. 126 (26%), p < 0.0001 and 140 (46%) vs. 90 (24%), p < 0.0001]. Kaplan–Meier survival curves by frailty status are presented in Fig. 3. Further, the degree of frailty at ICU admission was associated with mortality in a dose–response manner with mortality rising from approximately 20% in those with CFS < 4 or FI < 0.1, to approximately 50% in those with CFS > 4 or FI > 0.3 (Fig. 4). Ventilator-free days and ICU-free days were also lower in frail patients. At 6 months, frail patients were less likely to have returned home and had lower levels of function as measured by ADL and PPS. HRQOL was also lower in those frail as measured by CFS and FI. Frailty measured at hospital discharge was associated with worse outcomes at 6 months including higher mortality, as well as reduced likelihood of living at home, and impaired ADLs, PPS and QoL. In multiple logistic regression, the adjusted odds ratios (95% CI) of 6-month mortality from ICU admission CFS ≥ 5 or FI ≥ 0.2 were 2.2 (1.6–3.2, p < 0.001, n = 687) and 2 (1.4–2.8, p = 0.0002, n = 684) respectively while the odds ratios of 6 month mortality from hospital discharge CFS ≥ 5 or FI ≥ 0.2 were 2.4 (1.2–5, p = 0.017, n = 451) and 2 (0.9, 4.7, p = 0.097, n = 446) respectively.
Discussion
In this cohort of critically ill patients 50 years or older who received life support interventions in the ICU, frailty whether measured by the CFS or FI was common at ICU admission, hospital discharge and at 6 months with more individuals identified as frail with the FI than the CFS at all time points. Frailty at ICU admission was associated with worse outcomes including mortality during the index hospitalization and worse outcomes at 6-month follow-up, including increased mortality, reduced functional status and reduced quality of life. Frailty measured at hospital discharge was also associated with worse outcomes. There were high rates of mortality in patients characterized as frail in a dose response manner. In survivors, frailty by both measures was dynamic and its severity fluctuated over the course of observation with increasing levels during hospitalization improving to six months although this was impacted by survival bias with higher mortality rates in those with higher frailty levels. Significant number of patients did not return to baseline frailty states with transitions in both directions although most of the transitions were to higher frailty states. The results of our study are in keeping with other studies of critically ill frail patients reporting similar longitudinal frailty transitions using the CFS [37, 39].
Both the CFS and FI performed similarly in identifying patients with worsened outcomes, were highly correlated and could be measured at different time points during the recovery from life threatening illness. Although the CFS is the most commonly used ICU frailty tool and can be administered by any ICU clinician [40, 41], it is a summative measure [8] used mainly as a descriptive screening tool to characterize populations, or as a prognostic tool [42,43,44]. The CFS does not provide insights on the causes of frailty but should lead to further assessment to identify and potentially remedy modifiable factors. Since frailty may have multi-domain contributors including physical, mental and social elements, a CGA should be used to identify its’ determinants [45]. As far as we are aware, this has not been done in those requiring critical care.
The CGA has been shown to improve the likelihood of discharge to home, improve QoL and reduce caregiver burden in hospitalized patients [46, 47]. Critical elements of CGA include tailored treatment plans, multi-disciplinary input and involvement of patients, and caregivers in goal setting [46]. The utility of FI based on the elements of CGA over the CFS may lie in the examination of the individual deficits identified to help understand the transition between frailty states or progression of frailty and may allow for the identification of deficits amenable to intervention, need for optimization of therapy or need for secondary prevention. Examples include identification of possible depression or anxiety disorders, incontinence, fall risk, congestive heart failure or myocardial infarction. Further, the FI may be more suitable as a repeatable test as it able to identify accumulating deficits in a granular manner upstream of any manifestation in loss of function as measured by the CFS. The FI can be constructed with data collected by the members of the clinical team as part of their routine care [48], and thus the identification of frailty with a CGA based FI as in our study may guide the implementation of these elements in a patient’s treatment plan. The dynamic nature of frailty we documented in this cohort during hospitalization and recovery may reflect the impact of underlying chronic health conditions and superimposed new or residual acute conditions highlighting the need to design and evaluate interventions that may favourably modify the severity and trajectory of frailty.
The poor outcomes observed and mortality gradient with frailty severity can inform shared decision making with patients and families. In addition, the differences observed in process of care between frail and non-frail patients may point to potential targets for intervention including physical rehabilitation, better infection control and delirium prevention. Adoption of a multi-faceted intervention such as the ABCDEF bundle [49] during the ICU stay may be particularly relevant to frail patients and it may more effectively improve outcomes in frail patients than in non-frail patients. Further, interventions may need to be continued throughout the ward stay and after hospital discharge in the community with a focus on rehabilitation, exercise, nutrition, medication optimization and social connection [50]. Multi-disciplinary ICU follow-up clinics could be used to facilitate this. Any interventions will need to be rigorously piloted and tested in further studies for their effect on patient, caregiver centred outcomes.
Strengths of this study include the prospective, multicentre, longitudinal design using two different methods of frailty assessment, evaluating frailty and other key outcomes periodically over the trajectory of critical illness beyond hospital discharge. The cohort was assembled with broad inclusion criteria reflective of patients treated in ICUs although the exclusion criteria, requiring follow-up at 6 months may have selected out patients with better supports and more stable housing situations and may not have been representative of the whole cohort eligible. Other limitations of this study include the observational design which precludes causal inferences. Statistical tests were not adjusted for multiplicity which could have resulted in some isolated type I errors. However, the consistency and strength of associations suggests the overall results are robust to type I errors, and adjustments for multiplicity need to be weighed against increased risk of type II errors. Our reliance on patients who had family members or caregivers to provide information for the frailty assessment may have introduced selection bias and increased the likelihood of discharge home with caregiver support. Finally, in part, our study was conducted during the COVID-19 pandemic which may have impacted the results observed.
Conclusion
Frailty can be measured with both the CFS and FI over the course of critical illness, is dynamic and fluctuates during recovery. Whether ascertained by the CFS or FI, frailty on ICU admission and hospital discharge is associated with worse outcomes. Research is required to study the advantages of care directed to treating or mitigating the deficits identified in a FI based on a CGA. Frailty is common in the critically ill, likely to increase with aging populations and there is a need to develop and study care pathways that can improve outcomes.
Data availability
De-identified data collected during this study will be made available upon reasonable request after all secondary analysis are complete and on upon approval of pertinent research ethics boards.
References
Ofori-Asenso R, Chin KL, Mazidi M, Zomer E, Ilomaki J, Zullo AR, Gasevic D, Ademi Z, Korhonen MJ, LoGiudice D, Bell JS, Liew D (2019) Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw Open 2:e198398
Xue QL (2011) The frailty syndrome: definition and natural history. Clin Geriatr Med 27:1–15
Flaatten H, De Lange DW, Morandi A, Andersen FH, Artigas A, Bertolini G, Boumendil A, Cecconi M, Christensen S, Faraldi L, Fjølner J, Jung C, Marsh B, Moreno R, Oeyen S, Öhman CA, Pinto BB, Soliman IW, Szczeklik W, Valentin A, Watson X, Zaferidis T, Guidet B, on behalf of the VIPsg, (2017) The impact of frailty on ICU and 30-day mortality and the level of care in very elderly patients (≥ 80 years). Intensive Care Med 43: 1820–1828
Bagshaw SM, Stelfox HT, McDermid RC, Rolfson DB, Tsuyuki RT, Baig N, Artiuch B, Ibrahim Q, Stollery DE, Rokosh E, Majumdar SR (2014) Association between frailty and short- and long-term outcomes among critically ill patients: a multicentre prospective cohort study. CMAJ 186:E95-102
Muscedere J, Waters B, Varambally A, Bagshaw SM, Boyd JG, Maslove D, Sibley S, Rockwood K (2017) The impact of frailty on intensive care unit outcomes: a systematic review and meta-analysis. Intensive Care Med 43:1105–1122
Bagshaw SM, Stelfox HT, Johnson JA, McDermid RC, Rolfson DB, Tsuyuki RT, Ibrahim Q, Majumdar SR (2015) Long-term association between frailty and health-related quality of life among survivors of critical illness: a prospective multicenter cohort study. Crit Care Med 43:973–982
Buta BJ, Walston JD, Godino JG, Park M, Kalyani RR, Xue QL, Bandeen-Roche K, Varadhan R (2016) Frailty assessment instruments: systematic characterization of the uses and contexts of highly-cited instruments. Ageing Res Rev 26:53–61
Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, Mitnitski A (2005) A global clinical measure of fitness and frailty in elderly people. CMAJ 173:489–495
Bertschi D, Waskowski J, Schilling M, Donatsch C, Schefold JC, Pfortmueller CA (2022) Methods of assessing frailty in the critically ill: a systematic review of the current literature. Gerontology 68:1321–1349
Bruno RR, Wernly B, Bagshaw SM, van den Boogaard M, Darvall JN, De Geer L, de Gopegui Miguelena PR, Heyland DK, Hewitt D, Hope AA, Langlais E, Le Maguet P, Montgomery CL, Papageorgiou D, Seguin P, Geense WW, Silva-Obregon JA, Wolff G, Polzin A, Dannenberg L, Kelm M, Flaatten H, Beil M, Franz M, Sviri S, Leaver S, Guidet B, Boumendil A, Jung C (2023) The clinical frailty scale for mortality prediction of old acutely admitted intensive care patients: a meta-analysis of individual patient-level data. Ann Intensive Care 13:37
Bagshaw M, Majumdar SR, Rolfson DB, Ibrahim Q, McDermid RC, Stelfox HT (2016) A prospective multicenter cohort study of frailty in younger critically ill patients. Crit Care 20:175
Turner G, Clegg A (2014) Best practice guidelines for the management of frailty: a British Geriatrics Society, Age UK and Royal College of General Practitioners report. Age Ageing 43:744–747
Rockwood K, Mitnitski A (2007) Frailty in relation to the accumulation of deficits. J Gerontol Ser A 62:722–727
Heyland DK, Garland A, Bagshaw SM, Cook D, Rockwood K, Stelfox HT, Dodek P, Fowler RA, Turgeon AF, Burns K (2015) Recovery after critical illness in patients aged 80 years or older: a multi-center prospective observational cohort study. Intensive Care Med 41:1911–1920
Muscedere JAP, Bagshaw S, Ball I, Boyd JG, Cook D, Day A, Kho M, Loubani O, Martin A, Mehta S, Mele T, Norman P, Rewa O, Sarti A, Shahin J, Sibley S, Wang H (2023) Frailty, outcomes, recovery and care steps of critically ill patients (FORECAST): a prospective observational multi centre study. Intensive Care Med Exp 11:72
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP (2007) Strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335:806–808
Perlis RH, Haneuse SJPA, Rubenfeld GD, Fihn SD, Rivara FP (2021) Reporting clinical studies affected by the COVID-19 pandemic: guidelines for authors. JAMA Netw Open 4:e2036155–e2036155
Charlson ME, Pompei P, Ales KL, McKen-zie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chron Dis 40:373–383
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Katz S (1983) Assessing self-maintenance: activities of daily living, mobility, and instrumental activities of daily living. J Am Geriatr Soc 31:721–727
Anderson F, Downing GM, Hill J, Casorso L, Lerch N (1996) Palliative performance scale (PPS): a new tool. J Palliat Care 12:5–11
Jorm AF, Jacomb PA (1989) The informant questionnaire on cognitive decline in the elderly (IQCODE): socio-demographic correlates, reliability, validity and some norms. Psychol Med 19:1015–1022
Rabin R, de Charro F (2001) EQ-5D: a measure of health status from the EuroQol Group. Ann Med 33:337–343
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the european society of intensive care medicine. Intensive Care Med 22:707–710
Hodgson C, Needham D, Haines K, Bailey M, Ward A, Harrold M, Young P, Zanni J, Buhr H, Higgins A, Presneill J, Berney S (2014) Feasibility and inter-rater reliability of the ICU mobility scale. Heart Lung 43:19–24
Ely EW, Inouye SK, Bernard GR, Gordon S, Francis J, May L, Truman B, Speroff T, Gautam S, Margolin R, Hart RP, Dittus R (2001) Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA 286:2703–2710
Gusmao-Flores D, Salluh JIF, Chalhub RÁ, Quarantini LC (2012) The confusion assessment method for the intensive care unit (CAM-ICU) and intensive care delirium screening checklist (ICDSC) for the diagnosis of delirium: a systematic review and meta-analysis of clinical studies. Crit Care 16:R115
Liu B, Moore JE, Almaawiy U, Chan WH, Khan S, Ewusie J, Hamid JS, Straus SE, Collaboration MO (2018) Outcomes of mobilisation of vulnerable elders in ontario (MOVE ON): a multisite interrupted time series evaluation of an implementation intervention to increase patient mobilisation. Age Ageing 47:112–119
Inouye SK, van Dyck CH, Alessi CA, Balkin S, Siegal AP, Horwitz RI (1990) Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med 113:941–948
Dykes L, K R CFS Guidance and Training. In: Editor (ed) Book CFS Guidance and Training. Dalhousie University, City
Chan DC, Tsou HH, Chen CY, Chen CY (2010) Validation of the Chinese-Canadian study of health and aging clinical frailty scale (CSHA-CFS) telephone version. Arch Gerontol Geriatr 50:e74-80
Muscedere J, Bagshaw SM, Boyd G, Sibley S, Norman P, Day A, Hunt M, Rolfson D (2022) The frailty, outcomes, recovery and care steps of critically ill patients (FORECAST) study: pilot study results. Intensive Care Med Exp 10:23
Gordon EH, Reid N, Khetani IS, Hubbard RE (2021) How frail is frail? A systematic scoping review and synthesis of high impact studies. BMC Geriatr 21:719
Hoover M, Rotermann M, Sanmartin C, Bernier J (2013) Validation of an index to estimate the prevalence of frailty among community-dwelling seniors. Health Rep 24:10–17
Mitnitski AB, Mogilner AJ, Rockwood K (2001) Accumulation of deficits as a proxy measure of aging. Sci World J 1:323–336
Theou O, Brothers TD, Mitnitski A, Rockwood K (2013) Operationalization of frailty using eight commonly used scales and comparison of their ability to predict all-cause mortality. J Am Geriatr Soc 61:1537–1551
Brummel NE, Girard TD, Pandharipande PP, Thompson JL, Jarrett RT, Raman R, Hughes CG, Patel MB, Morandi A, Gill TM, Ely EW (2020) Prevalence and course of frailty in survivors of critical illness. Crit Care Med 48:1419–1426
Havey TC, Fowler RA, Pinto R, Elligsen M, Daneman N (2013) Duration of antibiotic therapy for critically ill patients with bloodstream infections: a retrospective cohort study. The Canadian journal of infectious diseases and medical microbiology. J Canadien des Maladies Infectieuses et de la Microbiologie Medicale 24: 129–137
Geense W, Zegers M, Dieperink P, Vermeulen H, van der Hoeven J, van den Boogaard M (2020) Changes in frailty among ICU survivors and associated factors: results of a one-year prospective cohort study using the Dutch Clinical Frailty Scale. J Crit Care 55:184–193
Shears M, Takaoka A, Rochwerg B, Bagshaw SM, Johnstone J, Holding A, Tharmalingam S, Millen T, Clarke F, Rockwood K, Li G, Thabane L, Muscedere J, Stelfox HT, Cook DJ (2018) Assessing frailty in the intensive care unit: a reliability and validity study. J Crit Care 45:197–203
Flaatten H, Guidet B, Andersen FH, Artigas A, Cecconi M, Boumendil A, Elhadi M, Fjølner J, Joannidis M, Jung C, Leaver S, Marsh B, Moreno R, Oeyen S, Nalapko Y, Schefold JC, Szczeklik W, Walther S, Watson X, Zafeiridis T, de Lange DW, the VIPSG, (2021) Reliability of the clinical frailty Scale in very elderly ICU patients: a prospective European study. Ann Intensive Care 11:22
Theou O, Searle SD (2018) Using frailty tools as prognostic markers in patients who are acutely ill. CMAJ 190:E182–E183
Rockwood K, Theou O (2020) Using the clinical frailty scale in allocating scarce health care resources. Can Geriatr J 23:210–215
Guidet B, de Lange DW, Boumendil A, Leaver S, Watson X, Boulanger C, Szczeklik W, Artigas A, Morandi A, Andersen F, Zafeiridis T, Jung C, Moreno R, Walther S, Oeyen S, Schefold JC, Cecconi M, Marsh B, Joannidis M, Nalapko Y, Elhadi M, Fjølner J, Flaatten H (2020) The contribution of frailty, cognition, activity of daily life and comorbidities on outcome in acutely admitted patients over 80 years in European ICUs: the VIP2 study. Intensive Care Med 46:57–69
Lee H, Lee E, Jang IY (2020) Frailty and comprehensive geriatric assessment. J Korean Med Sci 35:e16
Ellis G, Gardner M, Tsiachristas A, Langhorne P, Burke O, Harwood RH, Conroy SP, Kircher T, Somme D, Saltvedt I, Wald H, O'Neill D, Robinson D, Shepperd S (2017) Comprehensive geriatric assessment for older adults admitted to hospital. Cochrane Database Syst Rev 9: CD006211
Chen Z, Ding Z, Chen C, Sun Y, Jiang Y, Liu F, Wang S (2021) Effectiveness of comprehensive geriatric assessment intervention on quality of life, caregiver burden and length of hospital stay: a systematic review and meta-analysis of randomised controlled trials. BMC Geriatr 21:377
Darvall JN, Greentree K, Loth J, Bose T, De Silva A, Braat S, Lim WK, Story DA (2020) Development of a frailty index from routine hospital data in perioperative and critical care. J Am Geriatr Soc 68:2831–2838
Marra A, Ely EW, Pandharipande PP, Patel MB (2017) The ABCDEF bundle in critical care. Crit Care Clin 33:225–243
Rasiah J, Prorok JC, Adekpedjou R, Barrie C, Basualdo C, Burns R, De Paul V, Donnelly C, Doyle A, Frank C, Dolsen S, Giguère A, Hsiung S, Kim P, McDonald EG, O’Grady H, Patey A, Puxty J, Racey M, Resin J, Sims-Gould J, Stewart S, Theou O, Webster S, Muscedere J (2022) Enabling healthy aging to AVOID frailty in community dwelling older canadians. Can Geriat J 25:202–211
Acknowledgements
We would like to acknowledge the following individuals for their contributions toward the implementation of this study at the participating centres: Tracy Boyd and Michaela Hanley from Kingston Health Sciences Centre, Kingston, ON; Irene Watpool, Rebecca Porteous, Jessica Haines and Sydney Mietzitis from The Ottawa Hospital, Ottawa, ON; Eileen Campbell, Athena Ovsenek and Loretta Baratta from London Health Sciences Centre—Victoria Hospital, London, ON; Laurel Kelly, Heather O’Grady and Abby Hurd from St. Joseph’s Healthcare, Hamilton, ON; Nadia Baig and Lily Guan from University of Alberta, Calgary, AB; Valerie Barrette and Laura Lee Magennis, from Nova Scotia Health Authority, Halifax, NS; Danae Tassy, Vincent Cheung and Sam-Ang Toun from Maisonneuve Rosemont, Montreal, QC; Estel Deblois, Lise Lavoie, Julien Le Beller, Annick Jutras, Monia Noël-Hunter and Emilie Côté from Hotel Dieu De Levis, Levis, QC, Josie Campisi, Raham Rahgoshai and Aasmine Kaur from University of McGill, Montreal, QC; Virginie Williams, Sabrina Araujo De Franca and Julia Lainer Palacios from Sacre Coeur Hospital, Montreal, QC; Sumesh Shah from Mount Sinai Hospital, Toronto, ON; Tracey Bentall, Courtney Hooper and Chadia ElKhatib from London Health Sciences Centre—University Hospital, London, ON.
We would like to acknowledge Karla Krewulak for review of the manuscript.
Funding
Funding for the study was received from the Canadian Frailty Network which is funded by the Government of Canada. The funder did not have any role in the design, study conduct, data analysis or writing of the manuscript.
SMB is supported by a Canada Research Chair in Critical Care Outcomes and Systems Evaluation. PMA is supported by a Fonds de recherche du Québec—Santé Clinical Scholar Award. MK was funded by a Canada Research Chair in Critical Care Rehabilitation and Knowledge Translation during this study.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
JM is the Scientific Director for the Canadian Frailty Network. SMB reports receiving fees for scientific advisory work from Baxter, BioPorto, Novartis, Sea Star Medical, Sphingotec (all unrelated to the submitted work). JGB receives a stipend from Ontario Health-Trillium Gift of Life for his role as Regional Medical Lead. He also received an in-kind contribution from Edwards Lifesciences who have provided cerebral oximeters for his program of research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The Canadian Critical Care Trials Group members are listed in the Acknowledgement section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Muscedere, J., Bagshaw, S.M., Kho, M. et al. Frailty, Outcomes, Recovery and Care Steps of Critically Ill Patients (FORECAST): a prospective, multi-centre, cohort study. Intensive Care Med 50, 1064–1074 (2024). https://doi.org/10.1007/s00134-024-07404-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-024-07404-9